Industrial Scale-Up of Dapoxetine: A Novel Acetophenone-Based Synthetic Route for High-Purity API Manufacturing
Introduction to Advanced Dapoxetine Manufacturing
The pharmaceutical landscape for treating premature ejaculation has been significantly shaped by the introduction of Dapoxetine, a short-acting selective serotonin reuptake inhibitor (SSRI). As the demand for this active pharmaceutical ingredient (API) continues to grow globally, the efficiency and sustainability of its manufacturing process have become critical focal points for industry stakeholders. Patent CN102746170A introduces a transformative preparation method that utilizes acetophenone as a primary feedstock, marking a departure from more convoluted historical syntheses. This novel approach not only simplifies the synthetic pathway but also aligns with modern green chemistry principles by minimizing the use of toxic reagents and reducing the discharge of three wastes. For R&D directors and procurement specialists, understanding the nuances of this route is essential for securing a reliable supply chain.
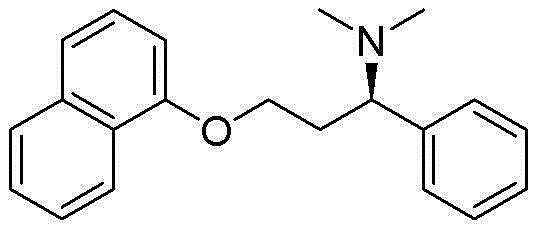
The structural complexity of Dapoxetine, characterized by its naphthalene ether linkage and chiral benzylic amine center, traditionally posed challenges in terms of stereochemical control and purification. However, the methodology outlined in the referenced patent provides a robust framework for overcoming these hurdles. By starting from acetophenone, a commodity chemical with stable pricing and abundant availability, manufacturers can mitigate supply chain volatility. The process involves a logical sequence of carboxylation, esterification, hydroxy imine formation, reduction, Boc protection, condensation, methylation, and finally, chiral resolution. Each step has been optimized to ensure high conversion rates and operational ease, making it an attractive candidate for technology transfer and commercial scale-up in the competitive generic API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dapoxetine has been plagued by several inefficiencies that hinder cost-effective mass production. Prior art routes, such as those disclosed in various international patents and literature digests, often rely on starting materials that are either expensive or difficult to source in bulk quantities. Furthermore, many conventional pathways involve harsh reaction conditions, including the use of strong acids or bases at extreme temperatures, which can lead to safety hazards and increased energy consumption. These methods frequently suffer from low overall yields due to the accumulation of impurities at each step, necessitating complex and costly purification protocols. Additionally, the generation of hazardous byproducts and the use of heavy metal catalysts in some older routes create significant environmental compliance burdens, complicating waste management and increasing the total cost of ownership for manufacturing facilities.
The Novel Approach
In stark contrast, the novel approach detailed in CN102746170A leverages a streamlined linear synthesis starting from acetophenone. This route is distinguished by its use of mild reaction conditions and readily accessible reagents, which collectively enhance the safety profile and economic viability of the process. The strategic implementation of a Boc protection group serves as a crucial control element, preventing unwanted side reactions during the subsequent alkylation and etherification steps. This level of chemical precision ensures a cleaner reaction profile and simplifies downstream processing. The final resolution step using tartaric acid derivatives is a proven, scalable technique for achieving high enantiomeric purity, which is paramount for API quality. By integrating these improvements, the new method offers a compelling solution for cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous quality standards.
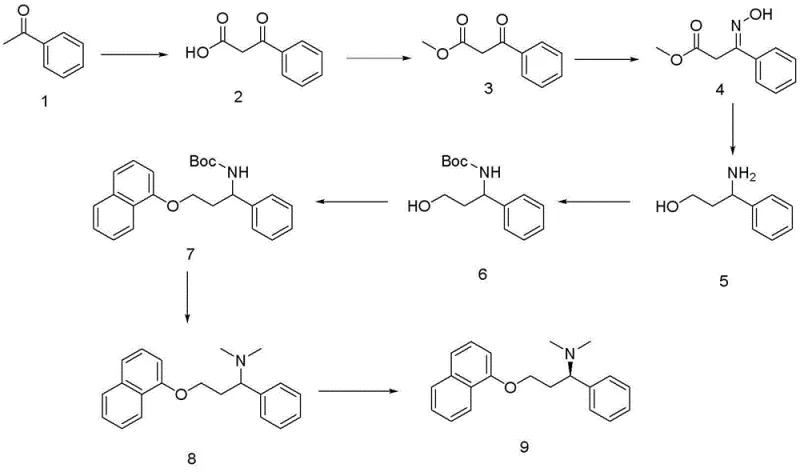
Mechanistic Insights into Acetophenone Carboxylation and Functionalization
The cornerstone of this synthetic strategy is the initial functionalization of acetophenone. The process begins with a carboxylation reaction at the alpha-position of the carbonyl group, facilitated by reagents such as triethylamine and magnesium chloride in acetonitrile. This step generates a beta-keto acid derivative, which is subsequently esterified using thionyl chloride and methanol to form a stable methyl ester. The choice of thionyl chloride allows for the simultaneous activation of the carboxylic acid and removal of water, driving the equilibrium towards the ester product efficiently. Following esterification, the ketone moiety is converted into an oxime using hydroxylamine hydrochloride under mildly alkaline conditions. This transformation is critical as it sets the stage for the introduction of the nitrogen atom required for the final amine functionality. The oxime formation is typically high-yielding and produces minimal byproducts, contributing to the overall atom economy of the process.
Subsequent reduction of the oxime to the primary amine is achieved using Lithium Aluminum Hydride (LAH) in anhydrous tetrahydrofuran. While LAH is a potent reducing agent requiring careful handling, its use here ensures complete reduction of the C=N bond without affecting the ester group under controlled conditions, or the ester is reduced to alcohol depending on specific stoichiometry, but in this specific patent logic, it leads to the amino-alcohol or amino-ester scaffold which is then protected. The patent specifies Boc protection immediately following reduction to safeguard the amine nucleophile. This protection is vital before the etherification step with 1-naphthol, which is conducted in DMF with potassium hydroxide. The nucleophilic substitution displaces the leaving group (derived from the alcohol chain) to form the ether linkage. Finally, the dimethylamine group is installed via reductive amination or direct methylation using formaldehyde and formic acid, known as Eschweiler-Clarke conditions. This sequence demonstrates a sophisticated understanding of orthogonal reactivity, ensuring that each functional group is manipulated selectively to build the complex Dapoxetine scaffold with high fidelity.
How to Synthesize Dapoxetine Efficiently
The synthesis of Dapoxetine via this acetophenone route represents a significant advancement in process chemistry, offering a balance between operational simplicity and chemical robustness. The procedure is designed to be executed in standard stainless steel reactors equipped with temperature control and agitation capabilities, making it accessible for most fine chemical manufacturing plants. The initial steps involving carboxylation and esterification set the carbon backbone, while the subsequent reduction and protection steps install the necessary nitrogen functionality with high precision. The critical etherification with 1-naphthol requires careful monitoring of temperature and base concentration to maximize yield and minimize O-alkylation byproducts. For a comprehensive understanding of the specific operating parameters, reagent ratios, and workup procedures, please refer to the detailed technical guide below which outlines the standardized protocol derived from the patent embodiments.
- Carboxylation and Esterification: React acetophenone with carboxylation agents followed by thionyl chloride and methanol to form the methyl ester intermediate.
- Functional Group Transformation: Convert the keto-ester to an oxime, reduce it to an amine using Lithium Aluminum Hydride, and protect the amine with a Boc group.
- Final Assembly and Resolution: Condense the protected amine with 1-naphthol, perform N-methylation, and resolve the racemate using L-tartaric acid to obtain pure Dapoxetine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this acetophenone-based synthesis route offers substantial benefits for procurement managers and supply chain directors looking to optimize their API sourcing strategies. The primary advantage lies in the raw material profile; acetophenone is a widely produced commodity chemical with a stable global supply, reducing the risk of shortages that often plague specialized starting materials. Furthermore, the reagents used throughout the synthesis, such as thionyl chloride, hydroxylamine hydrochloride, and Boc anhydride, are standard inventory items for most chemical suppliers, facilitating easier logistics and potentially lower purchasing costs through consolidated ordering. The elimination of exotic catalysts or hard-to-source chiral pool starting materials at the beginning of the synthesis significantly de-risks the supply chain, ensuring continuity of supply even during market fluctuations.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route directly translates to lower manufacturing costs through several mechanisms. By reducing the number of purification steps required between intermediates, the process minimizes solvent consumption and labor hours, which are major cost drivers in API production. The high yields reported in the patent embodiments suggest that less raw material is wasted, improving the overall material throughput. Additionally, the avoidance of expensive transition metal catalysts eliminates the need for costly metal scavenging and validation steps to ensure residual metal levels comply with regulatory limits. These cumulative efficiencies result in a more competitive cost structure for the final API, allowing for better margin management or more aggressive pricing in the generic marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust and well-understood chemical transformations. The reaction conditions, generally ranging from ambient temperature to moderate heating (e.g., 20°C to 110°C), do not require specialized cryogenic equipment or high-pressure vessels, making the process adaptable to a wider range of manufacturing sites. This flexibility allows for multi-site production strategies, reducing the dependency on a single facility. Moreover, the stability of the intermediates, particularly the Boc-protected species, allows for potential campaign manufacturing where intermediates can be stockpiled if necessary, providing a buffer against demand spikes. The reliance on common solvents like ethyl acetate and methanol further simplifies waste disposal and solvent recovery operations, enhancing the overall logistical efficiency of the manufacturing site.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by its inherent safety and environmental profile. The reduction in hazardous waste generation, as highlighted in the patent summary, aligns with increasingly stringent environmental regulations globally. The process avoids the use of persistent organic pollutants or highly toxic reagents where possible, simplifying the permitting process for new manufacturing lines. The exothermic profiles of the reactions are manageable with standard cooling systems, reducing the risk of thermal runaways during scale-up. This environmental and operational safety makes the technology attractive for long-term investment, ensuring that the manufacturing capacity can be expanded to meet growing market demand for Dapoxetine without encountering significant regulatory or technical bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Dapoxetine using this specific methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, aiming to clarify the feasibility and advantages of this route for potential partners. Understanding these details is crucial for making informed decisions about technology licensing or contract manufacturing agreements. The answers reflect the current state of the art as described in the intellectual property documentation.
Q: What are the key advantages of the acetophenone-based route for Dapoxetine?
A: The acetophenone-based route described in patent CN102746170A offers significant advantages including the use of inexpensive and readily available starting materials, milder reaction conditions compared to traditional methods, and a streamlined process that reduces the generation of hazardous waste, making it highly suitable for large-scale industrial production.
Q: How is chirality controlled in this synthesis method?
A: Chirality is introduced in the final stage of the synthesis through a classical resolution process. The racemic tertiary amine intermediate is treated with L-tartaric acid (or its salts) to form diastereomeric salts, allowing for the separation of the desired (S)-enantiomer, which corresponds to the active pharmaceutical ingredient Dapoxetine.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes common organic solvents like ethyl acetate, methanol, and DMF, and avoids extremely sensitive reagents where possible. The operational simplicity and improved yields reported in the patent data suggest it can be effectively scaled from pilot batches to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dapoxetine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical industry. Our team of expert chemists has thoroughly analyzed the acetophenone-based pathway for Dapoxetine and confirmed its potential for delivering high-purity intermediates and API. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, including precise control over chiral impurities and residual solvents. We are committed to leveraging advanced process chemistry to drive value for our partners.
We invite you to collaborate with us to explore the commercial potential of this innovative Dapoxetine synthesis. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to a secure supply chain and deep technical expertise. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your global business objectives.
