Optimizing Oxcarbazepine Production via Novel Dibenzoazepine Intermediate Synthesis
Optimizing Oxcarbazepine Production via Novel Dibenzoazepine Intermediate Synthesis
The pharmaceutical landscape for anticonvulsant agents continues to evolve, driven by the demand for more efficient and cost-effective manufacturing processes for established drugs like Oxcarbazepine. Patent CN1721409A introduces a groundbreaking methodology for synthesizing novel Dibenzo[b,f]azepine derivatives, which serve as critical precursors in the production of this vital epilepsy medication. The core innovation lies in the stabilization and functionalization of the dibenzoazepine scaffold, specifically addressing the historical challenges associated with the instability of key amine intermediates. By redefining the synthetic route, this technology offers a pathway to higher purity intermediates with significantly reduced impurity profiles. ![General structure of Dibenzo[b,f]azepine derivatives (Formula I) used as Oxcarbazepine intermediates](/insights/img/dibenzoazepine-intermediates-oxcarbazepine-synthesis-supplier-20260315084931-01.png) As illustrated in the general structure above, the versatility of the R1 and R2 substituents allows for fine-tuning the physicochemical properties of the intermediate, facilitating easier purification and handling in large-scale reactors. For R&D directors and process chemists, understanding the nuances of this scaffold is essential for implementing robust manufacturing protocols that meet stringent regulatory standards.
As illustrated in the general structure above, the versatility of the R1 and R2 substituents allows for fine-tuning the physicochemical properties of the intermediate, facilitating easier purification and handling in large-scale reactors. For R&D directors and process chemists, understanding the nuances of this scaffold is essential for implementing robust manufacturing protocols that meet stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Oxcarbazepine intermediates has been plagued by the inherent instability of the 5H-dibenzo[b,f]azepine core, particularly when attempting to introduce the carbamoyl functionality required for the final API. Traditional methods, such as those described in prior art like WO96/21649, often rely on harsh reaction conditions involving strong mineral acids or carboxylic acids in the presence of metal cyanate salts. These aggressive environments frequently lead to the degradation of the sensitive azepine ring, resulting in lower overall yields and a complex mixture of side products that are difficult to separate. Furthermore, the necessity for additional organic solvents in these conventional carbamylation reactions increases the operational complexity, requiring extensive solvent recovery systems and posing greater environmental hazards. For procurement managers, these inefficiencies translate directly into higher raw material consumption and increased waste disposal costs, eroding the profit margins of the final active pharmaceutical ingredient. The thermal instability of the intermediates under acidic conditions also necessitates precise temperature control, limiting the batch sizes and complicating the scale-up process for supply chain planners.
The Novel Approach
In stark contrast to the legacy methodologies, the process disclosed in CN1721409A utilizes a remarkably mild carbamylation strategy that leverages acetic acid as both the solvent and the reaction medium. This approach surprisingly eliminates the need for strong mineral acids, thereby preserving the integrity of the dibenzoazepine skeleton during the critical functionalization step. The invention demonstrates that by using an excess of metal cyanate salts, such as sodium cyanate or potassium cyanate, in glacial acetic acid, the reaction proceeds with exceptional efficiency and selectivity. 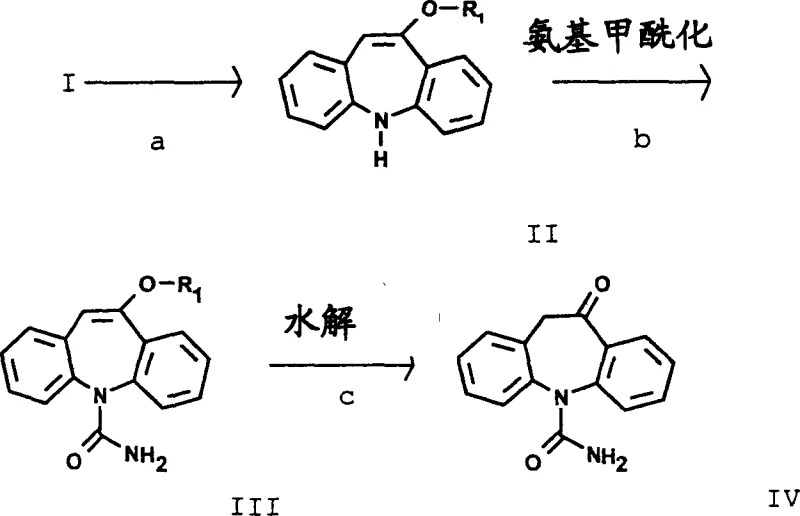 As depicted in the reaction scheme, the transformation from the amine precursor (Formula II) to the carbamoyl derivative (Formula III) is achieved under gentle conditions that prevent the formation of degradation by-products. This not only boosts the chemical yield significantly but also simplifies the downstream purification process, as the crude product obtained is of sufficiently high quality for subsequent hydrolysis steps. For technical teams, this represents a paradigm shift towards greener chemistry, reducing the reliance on hazardous reagents while enhancing the overall atom economy of the synthesis.
As depicted in the reaction scheme, the transformation from the amine precursor (Formula II) to the carbamoyl derivative (Formula III) is achieved under gentle conditions that prevent the formation of degradation by-products. This not only boosts the chemical yield significantly but also simplifies the downstream purification process, as the crude product obtained is of sufficiently high quality for subsequent hydrolysis steps. For technical teams, this represents a paradigm shift towards greener chemistry, reducing the reliance on hazardous reagents while enhancing the overall atom economy of the synthesis.
Mechanistic Insights into Acetic Acid-Mediated Carbamylation
The mechanistic superiority of this novel route stems from the unique role of acetic acid in modulating the reactivity of the cyanate ion towards the dibenzoazepine amine. Unlike strong mineral acids which fully protonate the amine and render it non-nucleophilic, acetic acid provides a buffered environment that maintains a sufficient concentration of the free amine species necessary for nucleophilic attack on the cyanate. This delicate balance ensures that the carbamylation occurs rapidly at the nitrogen center without triggering the acid-catalyzed hydrolysis or polymerization of the azepine ring. The absence of additional organic solvents further concentrates the reactants, driving the equilibrium towards the desired product according to Le Chatelier's principle. From a kinetic perspective, the reaction benefits from the high local concentration of reagents, allowing for shorter reaction times and reduced energy input. For quality control specialists, this mechanism implies a much cleaner reaction profile, minimizing the generation of structurally related impurities that could otherwise carry through to the final drug substance. The ability to conduct this reaction without external cooling or heating beyond moderate temperatures further underscores the thermodynamic favorability of this specific solvent-reagent combination.
Furthermore, the stability of the resulting carbamoyl intermediate (Formula III) is markedly improved compared to its predecessors, allowing for isolation and potential storage if necessary. The patent data indicates that the hydrolysis of this intermediate to the final ketone (Formula IV, Oxcarbazepine) can be performed with high efficiency, maintaining the gains achieved in the earlier steps. The structural rigidity imparted by the carbamoyl group protects the molecule during work-up, reducing the risk of epimerization or ring-opening side reactions. This robustness is critical for commercial manufacturing, where variations in processing times or minor deviations in pH can otherwise lead to batch failures. By understanding these mechanistic details, process engineers can design reactor setups that maximize throughput while maintaining the strict quality parameters required for pharmaceutical intermediates. The elimination of transition metals or exotic catalysts also removes the burden of heavy metal testing and clearance, streamlining the regulatory filing process for the final API.
How to Synthesize 10-Methoxy-dibenzo[b,f]azepine-5-carboxamide Efficiently
The practical implementation of this synthesis begins with the preparation of the 5H-dibenzo[b,f]azepine precursor, typically obtained through the hydrolysis of the corresponding enol ether derivative (Formula I) under controlled conditions. Once the free amine is generated, it is immediately subjected to the carbamylation protocol to prevent degradation. The detailed standardized synthesis steps involve suspending the amine in glacial acetic acid, adding a slight excess of sodium cyanate, and stirring at ambient temperature until conversion is complete. Following the reaction, the mixture is carefully neutralized to precipitate the product or extracted into an organic phase, depending on the specific substitution pattern. This streamlined workflow minimizes unit operations, reducing the total processing time and labor costs associated with the manufacture of these high-value intermediates.
- Prepare the dibenzo[b,f]azepine precursor (Formula II) by hydrolyzing the corresponding enol ether derivative under controlled acidic or basic conditions to expose the reactive amine site.
- Execute the carbamylation reaction by suspending the precursor in glacial acetic acid and adding an excess of metal cyanate salt (such as sodium cyanate) without additional organic solvents.
- Isolate the resulting carbamoyl intermediate (Formula III) through pH adjustment and extraction, followed by hydrolysis to yield the final 10-oxo-10,11-dihydro-dibenzo[b,f]azepine-5-carboxamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend far beyond simple chemical yield improvements. The elimination of strong mineral acids and additional organic solvents drastically simplifies the material sourcing strategy, reducing dependency on hazardous chemicals that require special handling and storage permits. This simplification translates into significant cost reductions in API manufacturing, as the expenses related to solvent recovery, waste treatment, and corrosion-resistant equipment maintenance are substantially lowered. The robustness of the process also enhances supply chain reliability, as the risk of batch failure due to sensitive reaction conditions is minimized, ensuring a consistent flow of intermediates to downstream formulation sites. Moreover, the higher purity of the crude intermediate reduces the burden on purification teams, allowing for faster turnaround times and shorter lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of expensive solvent systems and the reduction in reagent equivalents directly lower the variable costs per kilogram of production. By avoiding the use of strong acids, the lifespan of reactor vessels and piping is extended, deferring capital expenditure on equipment replacement. The higher yield means less raw material is wasted, optimizing the utilization of expensive starting materials and improving the overall gross margin of the production line.
- Enhanced Supply Chain Reliability: The mild reaction conditions reduce the safety risks associated with exothermic events or hazardous gas evolution, facilitating smoother operations in multi-purpose plants. This safety profile allows for larger batch sizes and more flexible scheduling, enabling suppliers to respond more agilely to fluctuations in market demand. The stability of the intermediates also permits longer storage windows, providing a buffer against supply disruptions and ensuring continuity of supply for critical medications.
- Scalability and Environmental Compliance: The solvent-free nature of the carbamylation step aligns perfectly with green chemistry principles, reducing the facility's environmental footprint and simplifying compliance with increasingly strict environmental regulations. The process is inherently scalable, moving seamlessly from laboratory benchtop to commercial scale-up of complex pharmaceutical intermediates without the need for specialized high-pressure or cryogenic equipment. This ease of scale-up accelerates time-to-market for generic versions of the drug, providing a competitive edge in the global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These insights are derived directly from the experimental data and process descriptions found within the patent documentation, providing a reliable foundation for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of the acetic acid carbamylation method over traditional mineral acid methods?
A: The use of acetic acid eliminates the need for strong mineral acids which can degrade the sensitive dibenzoazepine ring system. This mild condition significantly improves reaction yield and reduces the formation of degradation by-products, ensuring a cleaner impurity profile for downstream API processing.
Q: How does this novel process impact the scalability of Oxcarbazepine manufacturing?
A: By removing the requirement for additional organic solvents during the carbamylation step, the process simplifies work-up procedures and reduces solvent recovery costs. The robustness of the reaction under mild conditions allows for safer scale-up from pilot batches to multi-ton commercial production without compromising safety or quality.
Q: Are the intermediates described in CN1721409A stable enough for long-term storage?
A: While the free amine intermediates (Formula II) are relatively unstable, the patented process converts them rapidly into stable carbamoyl derivatives (Formula III). This immediate stabilization strategy prevents decomposition during storage and transport, ensuring consistent quality for pharmaceutical manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzoazepine Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust intermediate supply chains in the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1721409A are realized in practical, large-volume manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Dibenzo[b,f]azepine derivative meets the exacting standards required for API synthesis. Our commitment to quality ensures that our clients receive intermediates that facilitate smooth downstream processing, minimizing delays and maximizing final drug potency.
We invite global partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages specific to your production volume. We encourage you to contact us for specific COA data and route feasibility assessments to verify the compatibility of our intermediates with your current manufacturing processes. Let us collaborate to drive efficiency and innovation in the production of life-saving anticonvulsant therapies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
