Revolutionizing Eslicarbazepine Production: A Green Microbial Strategy for Commercial Scale-Up
Revolutionizing Eslicarbazepine Production: A Green Microbial Strategy for Commercial Scale-Up
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing, particularly for complex chiral intermediates like Eslicarbazepine, a next-generation antiepileptic drug. Patent CN102465159A introduces a groundbreaking microbial transformation method that utilizes Saccharomyces cerevisiae CGMCC No.2230 to achieve the asymmetric reduction of Oxcarbazepine. This biological approach addresses critical pain points in traditional synthesis, such as the use of toxic reagents, low atom economy, and difficult purification processes. By leveraging the intrinsic stereoselectivity of microbial enzymes, this technology offers a pathway to produce high-purity Eslicarbazepine with exceptional enantiomeric excess, positioning it as a superior alternative for reliable pharmaceutical intermediate suppliers seeking to optimize their production portfolios.
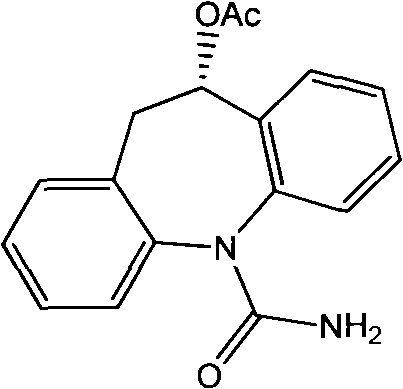
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Eslicarbazepine has relied heavily on chemical resolution or asymmetric hydrogenation, both of which present significant logistical and economic hurdles. Traditional resolution methods, such as those described in earlier Portela patents, involve reducing Oxcarbazepine to a racemic mixture followed by chiral separation. This process is inherently inefficient because it theoretically discards 50% of the synthesized material—the unwanted enantiomer—unless complex and costly recycling loops are implemented. Furthermore, asymmetric hydrogenation routes often require expensive transition metal catalysts, such as Ruthenium complexes, and high-pressure hydrogen sources, which introduce safety risks and necessitate rigorous removal of heavy metal residues to meet pharmacopeial standards.

The Novel Approach
In stark contrast, the novel microbial method described in CN102465159A bypasses these limitations by employing whole-cell biocatalysis. The process utilizes engineered yeast cells that act as self-contained factories, housing the necessary oxidoreductases and cofactor regeneration systems. This eliminates the need for external addition of expensive cofactors like NADPH, as the microorganism regenerates them in situ using simple cosubstrates like glucose. The reaction proceeds in an aqueous buffer system under mild temperatures (25-45°C), avoiding the harsh conditions and toxic solvents associated with chemical synthesis. This not only enhances the safety profile of the manufacturing process but also simplifies the downstream purification, as the product can be extracted directly from the fermentation broth with high stereochemical purity.
Mechanistic Insights into Microbial Asymmetric Reduction
The core of this technology lies in the stereospecific ketoreductase activity inherent to the Saccharomyces cerevisiae strain CGMCC No.2230. When exposed to the prochiral ketone substrate, Oxcarbazepine, the microbial enzymes selectively reduce the carbonyl group at the 10-position to form the (S)-hydroxy configuration, which corresponds to the therapeutically active Eslicarbazepine. The mechanism relies on a tightly coupled redox cycle where the oxidation of the added cosubstrate (glucose) drives the reduction of the drug precursor. This internal cofactor recycling is crucial for maintaining high turnover numbers and ensuring that the reaction proceeds to completion without stalling due to cofactor depletion, a common issue in isolated enzyme systems.
Furthermore, the cellular environment provides a protective matrix that stabilizes the enzymes, allowing for robust operation over extended reaction times (8-40 hours). The patent data indicates that by optimizing parameters such as pH (5.0-8.0) and biomass loading, the system can achieve conversion rates exceeding 99% with an enantiomeric excess (ee) of 100%. This level of control over the impurity profile is critical for R&D directors, as it minimizes the formation of the (R)-enantiomer and other side products, thereby reducing the burden on crystallization and chromatography steps during final API processing.
How to Synthesize Eslicarbazepine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this biocatalytic route in a pilot or production setting. It begins with the activation and fermentation of the yeast strain to generate sufficient biocatalyst mass, followed by the suspension of these cells in a buffered reaction medium containing the substrate. The process is designed to be modular, allowing for adjustments in substrate concentration and cosubstrate feeding to maximize yield. For detailed operational parameters and specific incubation conditions required to replicate these results, please refer to the standardized synthesis guide below.
- Cultivate Saccharomyces cerevisiae CGMCC No.2230 in a nutrient medium containing glucose and ammonium sulfate to generate enzyme-rich biomass.
- Perform the biotransformation by suspending the wet cell mass in phosphate buffer with Oxcarbazepine substrate, glucose cosubstrate, and ethanol.
- Incubate the reaction mixture at 25-45°C for 8-40 hours, followed by extraction with n-hexane and purification to isolate high-purity Eslicarbazepine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this microbial platform offers profound strategic benefits beyond mere technical feasibility. The shift from chemical catalysis to biocatalysis fundamentally alters the cost structure of manufacturing by removing dependency on precious metals and specialized chiral ligands. Additionally, the use of water as the primary reaction medium significantly lowers the costs associated with solvent purchase, recovery, and disposal. These factors combine to create a more resilient supply chain that is less vulnerable to fluctuations in the prices of petrochemical-derived reagents and rare earth metals.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and the avoidance of 50% material loss inherent in resolution processes lead to substantial raw material savings. By utilizing glucose as a cheap cosubstrate for cofactor regeneration, the process avoids the need for purchasing stoichiometric amounts of reducing agents. This streamlined material flow translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing strategies in the global API market without compromising on quality margins.
- Enhanced Supply Chain Reliability: Biological catalysts can be produced in large quantities through standard fermentation infrastructure, ensuring a consistent and scalable supply of the active agent. Unlike complex chemical catalysts that may have long lead times or single-source dependencies, the yeast strain can be maintained and propagated in-house. This self-sufficiency reduces the risk of production stoppages due to upstream supplier failures, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: The process operates under mild conditions that are easily transferable from laboratory flasks to industrial fermenters, facilitating rapid commercial scale-up of complex chiral intermediates. The aqueous nature of the reaction minimizes the generation of hazardous organic waste, simplifying compliance with increasingly strict environmental regulations. This green chemistry profile not only reduces waste treatment costs but also enhances the corporate sustainability image, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microbial synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing framework. Understanding these nuances is essential for making informed decisions about process adoption and vendor qualification.
Q: What are the primary advantages of the microbial method over chemical resolution for Eslicarbazepine?
A: Unlike traditional chemical resolution which inherently wastes 50% of the starting material (the unwanted R-enantiomer), the microbial method utilizing Saccharomyces cerevisiae achieves high stereoselectivity (up to 100% ee) directly from the prochiral ketone. This eliminates the need for costly recycling steps of the unwanted isomer and significantly reduces raw material consumption.
Q: How does this biocatalytic process impact environmental compliance and safety?
A: The process operates under mild aqueous conditions (pH 5.0-8.0, 25-45°C) using water as the primary solvent, drastically reducing the reliance on volatile organic compounds (VOCs) and toxic heavy metal catalysts often found in asymmetric hydrogenation. This simplifies waste treatment and aligns with stringent global environmental regulations for API manufacturing.
Q: Is this microbial route suitable for large-scale industrial production?
A: Yes, the patent demonstrates that the biocatalyst (yeast cells) can be produced via standard fermentation techniques which are easily scalable from laboratory to industrial tank sizes. The robustness of the whole-cell catalyst allows for high substrate loading (up to 0.5 mol/L) and simplified downstream processing, making it highly viable for commercial supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eslicarbazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalytic technologies in the synthesis of high-value antiepileptic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale processes like the one described in CN102465159A can be successfully translated into robust industrial operations. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Eslicarbazepine meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to explore how this green synthesis route can optimize your supply chain and reduce overall production costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this critical pharmaceutical ingredient.
