Advanced Manufacturing of Atorvastatin Calcium: A High-Yield Convergent Strategy for Global Supply Chains
Introduction: Revolutionizing Statin Manufacturing with Patent CN101613312A
The pharmaceutical landscape for lipid-lowering agents has long been dominated by Atorvastatin Calcium, a critical Active Pharmaceutical Ingredient (API) requiring rigorous synthesis standards to ensure efficacy and safety. Patent CN101613312A introduces a transformative preparation method that addresses the chronic inefficiencies plaguing traditional manufacturing processes, specifically targeting the issues of prolonged reaction times and suboptimal yields that have historically constrained supply chains. By shifting from complex linear syntheses to a more robust convergent strategy, this technology leverages a readily available chiral intermediate, (3R, 5R)-3,5-bis(triethylsiloxy)-7-amino-N,N-diphenylheptanamide, to streamline the production workflow. The innovation lies not just in the chemical transformations but in the holistic optimization of the process parameters, resulting in an overall yield increase to 58.3% compared to the industry standard of roughly 45%. For R&D directors and procurement specialists alike, this patent represents a pivotal opportunity to enhance the economic viability and operational reliability of statin production, ensuring a steady supply of high-purity material for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Atorvastatin Calcium has relied heavily on linear strategies that are inherently inefficient and prone to yield erosion at every stage. Traditional routes often involve the assembly of the pyrrole ring followed by the attachment of the chiral side chain, a sequence that necessitates harsh reaction conditions and multiple purification steps which degrade the optical purity of the final product. Existing methods frequently suffer from difficult crystallization profiles, requiring extensive downstream processing to remove impurities and achieve the stringent purity specifications demanded by regulatory bodies. Furthermore, the reliance on expensive reagents and solvents that cannot be easily recycled drives up the cost of goods sold (COGS), while long reaction times create bottlenecks in manufacturing schedules, limiting the ability to respond rapidly to market demand fluctuations. These structural inefficiencies in legacy processes result in significant material loss and increased waste generation, posing both economic and environmental challenges for large-scale producers.
The Novel Approach
In stark contrast, the methodology disclosed in CN101613312A employs a convergent synthesis strategy that fundamentally restructures the assembly of the molecule to maximize efficiency and minimize waste. The core of this innovation is the utilization of a pre-formed, silyl-protected chiral amine intermediate which couples directly with the fluorophenyl pyrrole precursor, bypassing several tedious steps associated with linear construction. A standout feature of this novel approach is the use of glacial acetic acid as a dual-function medium, acting simultaneously as the solvent and the acid catalyst for the critical condensation reaction. This simplification eliminates the need for exotic catalysts and reduces the complexity of the reaction mixture, facilitating easier product isolation through simple filtration. The process demonstrates remarkable robustness, achieving a product purity of over 99% with significantly shortened reaction cycles, thereby offering a scalable solution that aligns perfectly with the needs of modern, high-throughput pharmaceutical manufacturing facilities.
Mechanistic Insights into Acid-Catalyzed Condensation and Silyl Protection
The mechanistic elegance of this synthesis lies in the strategic protection of the chiral diol moiety and the subsequent acid-mediated cyclization. In the initial phase, the hydroxyl groups of the heptanamide precursor are protected as triethylsilyl ethers using chlorotriethylsilane and triethylamine, a transformation that is crucial for preventing side reactions and maintaining the stereochemical integrity of the (3R, 5R) centers during subsequent harsh conditions. Following this, catalytic hydrogenation over nickel reduces the nitrile group to the primary amine without affecting the silyl protecting groups, yielding a stable and versatile building block. The pivotal step occurs when this amine reacts with the 4-fluorophenyl pyrrole derivative in glacial acetic acid; here, the acetic acid protonates the carbonyl oxygen of the ketone moiety, increasing its electrophilicity and facilitating nucleophilic attack by the amine nitrogen. This leads to the formation of an iminium intermediate which subsequently undergoes dehydration and cyclization to form the stable pyrrole-attached scaffold, a process driven to completion by the removal of water and the thermal energy provided by reflux.
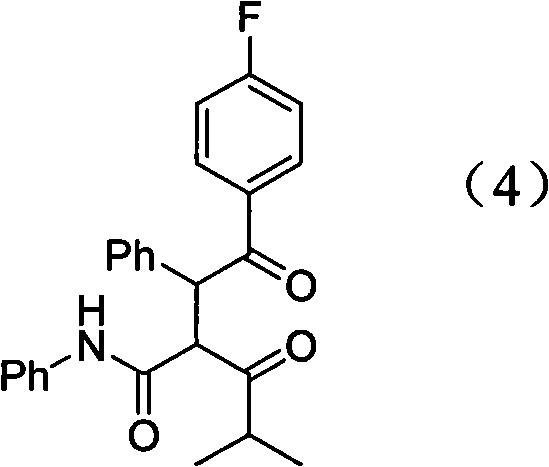
Furthermore, the control of impurities is intrinsically built into the reaction design through the choice of reagents and conditions that favor the thermodynamic product. The use of glacial acetic acid not only catalyzes the reaction but also helps in solubilizing polar byproducts while allowing the desired non-polar intermediate to precipitate upon cooling, effectively performing a crude purification in situ. The final hydrolysis step using hydrochloric acid is carefully controlled to remove the silyl groups without epimerizing the chiral centers, ensuring that the final calcium salt retains the high enantiomeric excess required for biological activity. This meticulous attention to mechanistic detail ensures that the impurity profile remains clean, reducing the burden on downstream purification units and guaranteeing a final API that meets the rigorous quality standards of international pharmacopeias.
How to Synthesize Atorvastatin Calcium Efficiently
The synthesis of Atorvastatin Calcium via this patented route involves a logical sequence of protection, coupling, and deprotection steps that are amenable to standard chemical engineering equipment. The process begins with the silylation of the chiral side chain followed by hydrogenation, proceeds to the key condensation in acetic acid, and concludes with hydrolysis and salification. Each step has been optimized to balance reaction kinetics with ease of workup, ensuring that the transition from laboratory scale to commercial production is seamless. For detailed operational parameters, stoichiometry, and specific temperature controls required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Protect the hydroxyl groups of the heptanamide precursor using chlorotriethylsilane and triethylamine, followed by nickel-catalyzed hydrogenation to generate the key amine intermediate.
- Perform a condensation reaction between the amine intermediate and the fluorophenyl pyrrole precursor in glacial acetic acid under reflux to form the protected atorvastatin skeleton.
- Execute hydrolysis using hydrochloric acid to remove silyl groups, neutralize with sodium hydroxide, and finalize with calcium chloride salification to obtain pure Atorvastatin Calcium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend far beyond simple chemical yield improvements, directly impacting the bottom line and operational resilience. The shift to a convergent strategy significantly reduces the number of unit operations required, which translates to lower capital expenditure on equipment and reduced labor costs per kilogram of produced API. By utilizing glacial acetic acid, a commodity chemical with a stable global supply chain, the process mitigates the risk of raw material shortages often associated with specialized reagents, ensuring consistent production scheduling. Additionally, the simplified isolation procedures, such as direct filtration of solids, drastically reduce solvent consumption and waste treatment costs, aligning the manufacturing process with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the ability to recycle the acetic acid solvent create a substantial reduction in variable production costs. The higher overall yield means that less starting material is required to produce the same amount of final API, effectively lowering the cost of goods sold and improving margin potential for generic manufacturers. Furthermore, the reduced reaction times allow for higher asset utilization rates, enabling facilities to produce more batches per year without additional capital investment.
- Enhanced Supply Chain Reliability: The reliance on readily available intermediates and common solvents minimizes dependency on single-source suppliers for exotic chemicals, thereby de-risking the supply chain against geopolitical or logistical disruptions. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the incidence of failed batches that can cause costly delays in supply. This reliability is critical for maintaining continuous supply to downstream formulation partners and meeting contractual obligations in the competitive generic drug market.
- Scalability and Environmental Compliance: The process is inherently scalable, with workup procedures that do not require complex chromatography or cryogenic conditions, making it ideal for multi-ton production campaigns. The reduction in solvent diversity and the use of recyclable acetic acid significantly lower the environmental footprint of the manufacturing process, facilitating easier permitting and compliance with green chemistry initiatives. This environmental advantage is increasingly becoming a key differentiator in supplier selection processes for major pharmaceutical companies committed to sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method, providing clarity on its practical application and benefits. These insights are derived directly from the experimental data and process descriptions within the patent documentation, offering a realistic view of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: How does the new synthesis method improve overall yield compared to traditional linear routes?
A: The patented convergent strategy improves overall yield from approximately 45% in conventional methods to 58.3% by utilizing a stable, easily accessible amine intermediate and optimizing the condensation step with glacial acetic acid.
Q: What are the cost advantages of using glacial acetic acid in the condensation step?
A: Glacial acetic acid serves a dual role as both the solvent and the catalyst, which eliminates the need for expensive additional catalysts and allows for solvent recovery and recycling, significantly lowering raw material costs.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability, featuring simplified workup procedures such as direct filtration of solids and reduced reaction times, which enhances throughput and operational efficiency in industrial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atorvastatin Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive landscape of generic pharmaceuticals, and we possess the technical expertise to bring this patented methodology to life. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is executed with precision and safety. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Atorvastatin Calcium meets the highest international standards, providing our partners with the confidence they need to navigate complex regulatory filings.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthesis technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the statin market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
