Scalable Biocatalytic Production of Chiral Mandelic Acid Intermediates for Global Pharma
Scalable Biocatalytic Production of Chiral Mandelic Acid Intermediates for Global Pharma
The pharmaceutical industry continuously demands more efficient and sustainable routes for producing chiral intermediates, particularly for semi-synthetic antibiotics and anti-tumor agents. A pivotal advancement in this domain is documented in patent CN101701222B, which discloses a highly efficient method for preparing single enantiomers of mandelic acid using a recombinant nitrilase derived from Alcaligenes sp. ECU0401. This technology represents a significant leap forward from traditional chemical synthesis, offering a green, cofactor-free pathway that achieves exceptional optical purity. The core innovation lies in the specific genetic sequence encoding the nitrilase, which enables the direct, enantioselective hydrolysis of mandelonitrile into (R)-(-)-mandelic acid. This approach not only simplifies the downstream processing but also aligns perfectly with modern green chemistry principles by eliminating heavy metal catalysts and reducing solvent usage. For R&D directors and procurement specialists, understanding the mechanistic depth and scalability of this patent is crucial for securing a reliable supply of high-value chiral intermediates.
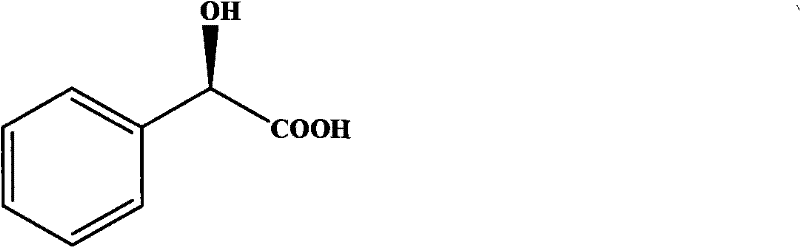
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure mandelic acid has relied heavily on classical chemical resolution techniques or less efficient biological transformations. Traditional chemical resolution typically involves the formation of diastereomeric salts using expensive chiral amines such as α-methylbenzylamine or ephedrine. This process is inherently inefficient because the maximum theoretical yield is limited to 50% unless a dynamic racemization step is introduced, which often requires harsh conditions. Furthermore, physical separation methods like chromatography or crystallization are energy-intensive and difficult to scale for bulk manufacturing. Previous biological attempts using wild-type strains or dehydrogenases often suffered from low catalytic activity, poor stability, and the critical requirement for expensive cofactors like NADH. These limitations result in high production costs, complex waste streams, and inconsistent supply reliability, posing significant challenges for large-scale pharmaceutical manufacturing where cost control and purity are paramount.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a genetically engineered recombinant nitrilase that catalyzes the one-step hydrolysis of nitriles to carboxylic acids without any cofactor participation. This method leverages the unique ability of the Alcaligenes sp. ECU0401 enzyme to distinguish between enantiomers, selectively hydrolyzing the (R)-nitrile while leaving the (S)-enantiomer intact, which can subsequently racemize under weak alkaline conditions to theoretically achieve 100% yield. The process operates under mild conditions, typically between 20°C and 60°C and pH 4.0 to 10.0, significantly reducing energy consumption compared to thermal chemical processes. The versatility of this biocatalyst is further demonstrated by its ability to accept various substituted substrates, as illustrated in the general reaction scheme below, making it a robust platform for diverse intermediate synthesis.
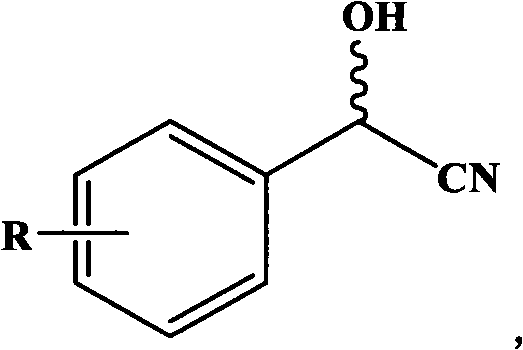
Mechanistic Insights into Nitrilase-Catalyzed Asymmetric Hydrolysis
The core of this technology is the specific DNA base sequence (SEQ NO.1) and its corresponding amino acid sequence (SEQ NO.2), which encode a nitrilase with a molecular weight of approximately 38.5 kDa. This enzyme functions through a precise catalytic mechanism where the active site accommodates the nitrile group of the substrate, facilitating nucleophilic attack by water to form an amide intermediate, which is subsequently hydrolyzed to the carboxylic acid and ammonia. The stereo-selectivity is governed by the chiral environment of the enzyme's active pocket, which sterically hinders the binding of the (S)-enantiomer while favoring the (R)-configuration. This high degree of specificity ensures that the resulting product possesses an enantiomeric excess (e.e.) greater than 99.5% after simple recrystallization. Unlike dehydrogenase-based systems, this nitrilase does not require external redox equivalents, thereby simplifying the reaction system and eliminating the need for complex cofactor regeneration systems that often limit reaction scale and increase operational complexity in industrial settings.
Impurity control in this biocatalytic process is inherently superior due to the high chemoselectivity of the enzyme. In chemical hydrolysis, side reactions such as over-hydrolysis or degradation of sensitive functional groups on the aromatic ring are common, leading to complex impurity profiles that require extensive purification. However, the recombinant nitrilase specifically targets the cyano group, leaving other substituents like chloro or hydroxy groups untouched, as evidenced by the successful conversion of o-chloromandelonitrile to (R)-(-)-o-chloromandelic acid with 85% yield and >99.7% e.e. The use of resting cells or immobilized cells further enhances purity by compartmentalizing the enzyme, preventing the release of intracellular proteases or other degradative enzymes into the reaction medium. This results in a cleaner crude product that requires fewer downstream purification steps, directly translating to higher overall recovery rates and reduced solvent consumption during the isolation phase.
How to Synthesize (R)-(-)-Mandelic Acid Efficiently
The implementation of this biocatalytic route involves a straightforward fermentation and bioconversion protocol that is highly amenable to industrial scale-up. The process begins with the cultivation of the recombinant host, typically E. coli transformed with the specific plasmid, in a defined medium optimized for high cell density and enzyme expression. Following harvest, the cells can be used directly as resting cells or immobilized in calcium alginate for enhanced stability and reusability. The bioconversion step is conducted in a buffered aqueous system where the substrate concentration can be pushed as high as 200 mmol/L without significant inhibition, demonstrating the robustness of the catalyst. Detailed standardized synthetic steps, including specific media compositions, induction protocols, and work-up procedures involving pH-controlled extraction, are outlined in the structured guide below to ensure reproducible high-quality output.
- Cultivate the recombinant Alcaligenes sp. ECU0401 transformant in a optimized medium containing glycerol and yeast extract at 30°C to express high levels of nitrilase activity.
- Suspend the harvested resting cells or immobilized cells in a buffered aqueous solution and introduce mandelonitrile substrate at concentrations up to 200 mmol/L.
- Maintain the reaction at pH 4.0-10.0 and 20-60°C, followed by acidification and organic solvent extraction to isolate the high-purity (R)-(-)-mandelic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic technology offers profound strategic advantages regarding cost structure and supply continuity. The elimination of expensive chiral resolving agents and cofactors fundamentally alters the cost equation, removing some of the most volatile and high-cost input materials from the bill of materials. Additionally, the ability to operate at high substrate concentrations means that reactor volumes can be minimized for a given output, improving capital efficiency and reducing the footprint required for production. The robustness of the immobilized cell system, which allows for repeated batch operations over 15 cycles without significant activity loss, drastically reduces the frequency of catalyst preparation and downtime. This operational stability ensures a predictable production schedule, mitigating the risks associated with batch-to-batch variability that often plague less mature biocatalytic processes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction system and the removal of costly auxiliaries. By utilizing a cofactor-free nitrilase, the process avoids the substantial expenses associated with purchasing and regenerating nicotinamide cofactors required by dehydrogenase pathways. Furthermore, the high enantioselectivity minimizes the loss of raw materials into unwanted isomers, maximizing the yield per kilogram of starting mandelonitrile. The downstream processing is also streamlined; since the reaction produces ammonia as a byproduct rather than complex salt waste, the neutralization and extraction steps are less chemically intensive. These factors combine to create a manufacturing process with a significantly lower variable cost profile, allowing for more competitive pricing in the global market for chiral intermediates.
- Enhanced Supply Chain Reliability: Supply security is bolstered by the use of a recombinant strain that can be propagated indefinitely, ensuring a consistent source of the biocatalyst independent of natural resource fluctuations. The substrate, mandelonitrile, is a commodity chemical available from multiple global suppliers, reducing the risk of single-source bottlenecks. Moreover, the tolerance of the enzyme to a wide pH and temperature range provides operational flexibility, allowing production to continue even if minor deviations in utility conditions occur. The capability to use immobilized cells extends the catalyst lifecycle, reducing the logistical burden of frequent enzyme shipments or fermentations. This resilience makes the supply chain less susceptible to disruptions, ensuring that downstream pharmaceutical manufacturers receive their critical intermediates on time and with consistent quality specifications.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this biocatalytic route aligns perfectly with increasingly stringent global standards for green manufacturing. The process operates in aqueous media, significantly reducing the volume of organic solvents required compared to traditional chemical resolution which often relies on large volumes of alcohols or hydrocarbons for crystallization. The absence of heavy metal catalysts eliminates the need for expensive and complex metal scavenging steps to meet residual metal limits in API intermediates. Waste generation is minimized as the primary byproduct is ammonia, which can be easily managed or utilized, and the high atom economy ensures that most of the carbon from the substrate ends up in the final product. This eco-friendly profile not only reduces waste disposal costs but also enhances the sustainability credentials of the final pharmaceutical product, a key consideration for modern ESG-focused supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrilase technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit for your specific manufacturing needs. Understanding these details is essential for technical teams assessing the feasibility of integrating this biocatalytic step into existing production lines or for procurement teams validating the quality assurances of potential suppliers.
Q: What are the primary advantages of using nitrilase over traditional chemical resolution for mandelic acid?
A: Unlike traditional chemical resolution which requires expensive chiral amines and generates stoichiometric waste, the nitrilase method described in CN101701222B utilizes a cofactor-free enzymatic hydrolysis. This results in significantly higher atom economy, eliminates the need for costly resolving agents, and achieves optical purity exceeding 99.5% e.e. directly through asymmetric synthesis.
Q: How does the recombinant Alcaligenes nitrilase improve process stability?
A: The recombinant strain ECU0401 demonstrates exceptional tolerance to high substrate concentrations up to 200 mmol/L and maintains activity across a broad pH range of 4.0 to 10.0. Furthermore, the enzyme can be utilized as immobilized cells, allowing for repeated batch operations over 15 cycles without significant loss of catalytic efficiency, ensuring consistent production quality.
Q: Can this biocatalytic method be applied to mandelic acid derivatives?
A: Yes, the patented nitrilase exhibits broad substrate specificity beyond simple mandelonitrile. It effectively catalyzes the hydrolysis of various substituted mandelonitriles, including o-chloro, m-chloro, p-chloro, and p-hydroxy derivatives, making it a versatile platform technology for synthesizing a wide range of chiral pharmaceutical building blocks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-(-)-Mandelic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of (R)-(-)-mandelic acid meets the exacting standards required for pharmaceutical synthesis. Our commitment to quality is backed by a deep understanding of the underlying biocatalytic mechanisms, allowing us to troubleshoot and optimize processes for maximum yield and optical purity.
We invite you to engage with our technical procurement team to discuss how this advanced nitrilase technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with a solid foundation of technical certainty and commercial viability.
