Advanced Synthesis of 2-(5-Fluoro-2,4-Dinitrophenoxy) Acetic Acid for Commercial Herbicide Production
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical agrochemical intermediates. Patent CN110240545B, published in April 2022, introduces a groundbreaking preparation method for 2-(5-fluoro-2,4-dinitrophenoxy) acetic acid, a pivotal precursor in the manufacturing of the widely used herbicide flumioxazin. This technical disclosure represents a significant paradigm shift from traditional, cost-prohibitive synthetic routes to a streamlined, environmentally benign process utilizing ethylene glycol. For R&D directors and procurement strategists, this patent offers a viable pathway to reduce raw material dependency on volatile fluorophenol markets while simultaneously enhancing overall process safety and yield. The methodology described herein not only addresses the chronic issue of low selectivity in nitration reactions but also leverages abundant commodity chemicals to stabilize supply chains against geopolitical fluctuations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(5-fluoro-2,4-dinitrophenoxy) acetic acid has relied heavily on the 3-fluorophenol method, a route fraught with significant economic and technical inefficiencies. As illustrated in the reaction scheme below, this conventional approach necessitates the initial etherification of 3-fluorophenol with chloroacetic acid, followed by a hazardous nitration step.
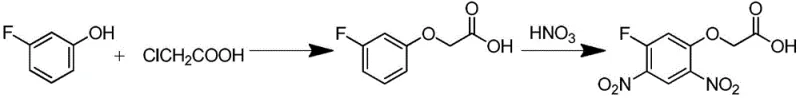
The reliance on 3-fluorophenol presents a major bottleneck; this starting material is notoriously expensive and difficult to source in bulk quantities, creating substantial cost pressure for manufacturers. Furthermore, the subsequent nitration step is chemically treacherous, as the ether bonds are prone to cleavage under strong acidic conditions, leading to poor selectivity and a complex impurity profile that requires extensive purification. Alternative routes involving 2,4-difluoronitrobenzene or dicyclohexylammonium glycolate suffer from similar drawbacks, including competitive side reactions with water and the high cost of specialized glycolate salts, ultimately capping the total yield between 28% and 81% and generating excessive chemical waste.
The Novel Approach
In stark contrast, the novel methodology disclosed in CN110240545B utilizes 1,5-difluoro-2,4-dinitrobenzene and ethylene glycol as the primary feedstocks, fundamentally restructuring the synthetic logic to maximize atom economy. Instead of attempting to nitrate a pre-formed ether, this route performs a nucleophilic aromatic substitution first, reacting the difluoro-dinitrobenzene directly with ethylene glycol under mild alkaline conditions to form the ethanol intermediate.
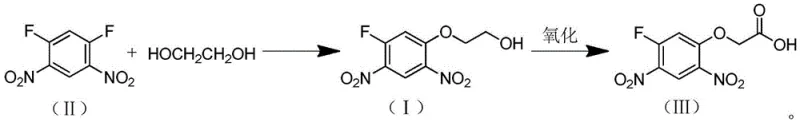
This strategic inversion of the reaction sequence eliminates the need for harsh nitration conditions entirely, thereby preserving the integrity of the ether linkage and ensuring high regioselectivity. The subsequent oxidation of the ethanol intermediate to the acetic acid can be achieved using green oxidants such as air or oxygen, often catalyzed by inexpensive metal salts. This approach not only simplifies the operational workflow by removing high-temperature requirements but also drastically reduces the generation of hazardous by-products, positioning it as a superior choice for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Nucleophilic Substitution and Catalytic Oxidation
The core chemical transformation in this novel process relies on a highly efficient nucleophilic aromatic substitution (SNAr) mechanism facilitated by the strong electron-withdrawing nature of the two nitro groups on the benzene ring. In the first stage, the hydroxyl group of ethylene glycol, activated by bases such as triethylamine, sodium hydroxide, or potassium carbonate, attacks the carbon atom bearing the fluorine at the 1-position. The presence of the ortho- and para-nitro groups significantly stabilizes the Meisenheimer complex intermediate, allowing the displacement of the fluoride ion to proceed rapidly even at room temperature. This mild condition is critical for preventing the degradation of the sensitive dinitro structure, a common failure point in older high-temperature protocols.
Following the formation of 2-(5-fluoro-2,4-dinitrophenoxy) ethanol, the process employs a sophisticated oxidation strategy to convert the primary alcohol to the carboxylic acid. The patent highlights the efficacy of a biomimetic catalytic system comprising TEMPO (tetramethyl piperidinyloxy), ferric nitrate nonahydrate, and an alkali metal halide. In this cycle, the nitroxyl radical mediates the hydrogen abstraction from the alcohol, while the iron species facilitates the regeneration of the active catalyst using molecular oxygen from the air. This mechanism ensures that the oxidation proceeds with high specificity, avoiding over-oxidation or degradation of the aromatic ring, and results in product purities exceeding 90% with yields reaching up to 97% in optimized examples.
How to Synthesize 2-(5-Fluoro-2,4-Dinitrophenoxy) Acetic Acid Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction atmosphere to replicate the high yields reported in the patent literature. The process begins with the careful mixing of 1,5-difluoro-2,4-dinitrobenzene and ethylene glycol in a mass ratio ranging from 1:1 to 1:5, ensuring an excess of glycol to drive the substitution to completion. The reaction is typically conducted at ambient temperatures for 2 to 3 hours in the presence of a base, followed by a straightforward aqueous workup to isolate the ethanol intermediate. For the oxidation step, operators can choose between simple air oxidation for cost sensitivity or the TEMPO/Fe(NO3)3 catalytic system for maximum speed and yield, with the final product crystallizing out upon cooling and concentration.
- React 1,5-difluoro-2,4-dinitrobenzene with ethylene glycol in the presence of an organic or inorganic base at room temperature to form the ethanol intermediate.
- Isolate the 2-(5-fluoro-2,4-dinitrophenoxy) ethanol intermediate via aqueous extraction and solvent removal.
- Oxidize the ethanol intermediate using air, oxygen, or a TEMPO-based catalytic system to obtain the final acetic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ethylene glycol-based route offers transformative benefits that extend far beyond simple yield improvements. By shifting away from the 3-fluorophenol dependency, manufacturers can decouple their production costs from the volatile pricing of specialized fluorinated phenols, which are often subject to tight supply constraints and regulatory scrutiny. The use of ethylene glycol, a ubiquitous and low-cost commodity chemical, provides a stable and predictable cost base, enabling more accurate long-term budgeting and pricing strategies for downstream herbicide products.
- Cost Reduction in Manufacturing: The elimination of expensive raw materials like 3-fluorophenol and dicyclohexylammonium glycolate results in a drastic reduction in direct material costs. Furthermore, the ability to perform the substitution reaction at room temperature significantly lowers energy consumption compared to traditional high-temperature nitration or reflux processes. The simplified post-treatment, which involves basic extraction and crystallization rather than complex chromatographic purification, further reduces operational expenditures related to solvents and labor.
- Enhanced Supply Chain Reliability: Sourcing 1,5-difluoro-2,4-dinitrobenzene and ethylene glycol is significantly more reliable than procuring niche fluorophenols, as these materials are produced at massive scales for various industrial applications. This abundance mitigates the risk of production stoppages due to raw material shortages. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in utility quality, ensuring consistent output even in diverse manufacturing environments.
- Scalability and Environmental Compliance: The process is inherently green, utilizing air or oxygen as the terminal oxidant in the preferred embodiments, which minimizes the generation of heavy metal waste associated with stoichiometric oxidants like chromates or permanganates. The reduction in "three wastes" (wastewater, waste gas, and solid waste) simplifies environmental compliance and lowers the cost of waste disposal. This environmental profile makes the technology highly scalable, allowing for seamless expansion from pilot batches to multi-ton commercial production without requiring prohibitive investments in pollution control infrastructure.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and optimization of this patented synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in CN110240545B, offering clarity on yield expectations, catalyst selection, and safety parameters for potential adopters of this technology.
Q: Why is the ethylene glycol route superior to the 3-fluorophenol method?
A: The ethylene glycol route avoids the expensive and hard-to-source 3-fluorophenol. It eliminates the harsh nitration step which often causes ether bond cleavage and low selectivity, resulting in significantly higher yields (up to 97%) and purity.
Q: What oxidants are suitable for the second step of this synthesis?
A: The patent discloses several green oxidant options including air, oxygen, hydrogen peroxide, and sodium hypochlorite. Preferably, a catalytic system using TEMPO and ferric nitrate with air or oxygen provides the highest efficiency and environmental profile.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at mild conditions (room temperature to 50°C), uses inexpensive raw materials like ethylene glycol, and generates less three-waste, making it highly scalable and compliant with modern environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(5-Fluoro-2,4-Dinitrophenoxy) Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a new synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-(5-fluoro-2,4-dinitrophenoxy) acetic acid meets the exacting standards required for flumioxazin synthesis.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating this cost-effective route into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this ethylene glycol-based method. Please contact our technical procurement team today to discuss specific COA data, route feasibility assessments, and how we can support your long-term goals for sustainable and profitable agrochemical manufacturing.
