Scalable Electrochemical Production of High-Purity Indolo[2,3-a]carbazole for Advanced OLED Applications
Scalable Electrochemical Production of High-Purity Indolo[2,3-a]carbazole for Advanced OLED Applications
The rapidly evolving landscape of Organic Light-Emitting Diode (OLED) technology demands materials that combine superior electron transport properties with sustainable manufacturing processes. Indolo[2,3-a]carbazole, a fused heterocyclic compound with a conjugated system exceeding that of standard carbazole, has emerged as a critical building block for next-generation electroluminescent materials. Recently, patent CN114318375A disclosed a groundbreaking electrochemical catalysis method for synthesizing this valuable scaffold. Unlike traditional routes that rely on harsh acidic conditions or expensive rare-earth metals, this innovation utilizes a mild, one-pot electrochemical strategy starting from readily available cyclohexanone and o-phenylenediamine. For R&D directors and procurement specialists in the electronic chemicals sector, this represents a significant paradigm shift towards greener, more cost-effective production of high-performance OLED intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indolo[2,3-a]carbazole has been plagued by significant operational and economic hurdles that hinder large-scale commercialization. One prominent literature route involves the condensation of diindolylmethane with trimethyl orthoformate under strong protonic acid catalysis at elevated temperatures (100°C). While effective, the starting material diindolylmethane is notoriously difficult to source and commands a high market price, severely limiting the economic viability of this pathway for bulk manufacturing. Another reported method employs a Fisher indole synthesis strategy using phenylhydrazine and 1,2-cyclohexanedione.
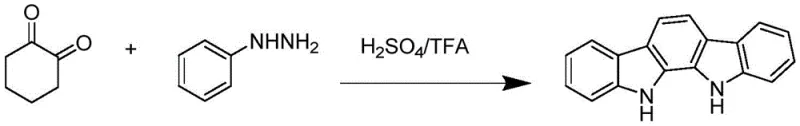
This route necessitates the use of corrosive strong acids such as sulfuric acid and trifluoroacetic acid (TFA), generating substantial volumes of acidic waste liquid that complicate three-waste treatment and increase environmental compliance costs. Furthermore, a third approach utilizing SmI2-catalyzed self-coupling of 3-indole formaldehyde suffers from the use of expensive, air-sensitive samarium reagents and multi-step procedures that result in low comprehensive yields (around 60%). These legacy methods collectively impose heavy burdens on supply chain stability and production costs.
The Novel Approach
In stark contrast to these cumbersome traditional pathways, the electrochemical method described in CN114318375A offers a streamlined, atom-economical solution. The core innovation lies in the direct oxidative coupling of cyclohexanone and o-phenylenediamine under mild electrochemical conditions. This transition-metal-free protocol eliminates the need for stoichiometric oxidants or hazardous strong acids, operating efficiently at room temperature. By leveraging anodic oxidation to drive the cyclization and aromatization steps simultaneously, the process achieves high yields (up to 89.19% in optimized examples) with exceptional purity (99.0% - 99.2%).
![Core electrochemical reaction scheme for Indolo[2,3-a]carbazole synthesis](/insights/img/indolo-carbazole-electrochemical-synthesis-oled-supplier-20260306100347-07.webp)
The simplicity of using commodity chemicals like cyclohexanone as the carbon source drastically reduces raw material expenditure. Moreover, the one-pot nature of the reaction minimizes unit operations, reducing labor intensity and equipment footprint. For supply chain managers, this translates to a robust manufacturing process that is less susceptible to the volatility of specialized reagent markets and easier to scale from laboratory to commercial tonnage without compromising quality or safety standards.
Mechanistic Insights into Electrochemical Oxidative Cyclization
The success of this synthesis hinges on a sophisticated interplay between electrochemical oxidation and Brønsted acid catalysis. The mechanism initiates with the condensation of cyclohexanone and o-phenylenediamine, catalyzed by an additive such as p-toluenesulfonic acid monohydrate (TsOH·H2O), to form an imine intermediate which subsequently tautomerizes to an enamine. Under the applied potential at the anode, this electron-rich enamine species undergoes single-electron oxidation to generate a reactive cation radical. This electro-generated intermediate is pivotal; it possesses sufficient electrophilicity to attack the electron-rich aromatic ring of the adjacent amine moiety, triggering the critical C-N bond formation required for ring closure.
A unique aspect of this patent is the strategic use of specific electrolytes, such as tetrabutylammonium dodecylsulfonate, which serve a dual function. Beyond acting as a conductive medium for electron transport, the bulky dodecylsulfonate anions create a steric environment around the cationic intermediate. This steric hindrance effectively suppresses unwanted side reactions, such as nucleophilic attacks by free amine molecules that could lead to polymerization or dimerization. Instead, it directs the reactivity towards intramolecular electrophilic addition, ensuring high selectivity for the desired indolo[2,3-a]carbazole scaffold. Following cyclization, a second anodic oxidation step facilitates deprotonation and aromatization, delivering the final stable product while protons are reduced at the cathode to evolve hydrogen gas, completing the redox cycle cleanly.
How to Synthesize Indolo[2,3-a]carbazole Efficiently
Implementing this electrochemical protocol requires precise control over reaction parameters to maximize yield and purity. The process is designed to be operationally simple, avoiding the complex inert atmosphere handling often required for organometallic chemistry. The following guide outlines the standardized procedure derived from the patent examples, suitable for pilot and commercial scale-up. Detailed standard operating procedures for specific reactor configurations can be found in the technical documentation provided by our engineering team.
- Feed cyclohexanone, o-phenylenediamine, TsOH·H2O additive, and electrolyte (e.g., tetrabutylammonium bromide) into a reaction vessel with solvent A (MeCN or DMA).
- Insert electrodes (Pt/C or Pb/Pt), apply constant current (10-15 mA) at room temperature, and react for 7-12 hours under monitoring.
- Extract the mixture with Solvent B (Water/Ethyl Acetate), remove solvent, and recrystallize the organic layer to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical methodology offers tangible strategic advantages beyond mere technical feasibility. The shift away from precious metal catalysts and hazardous strong acids fundamentally alters the cost structure and risk profile of producing indolo[2,3-a]carbazole. By simplifying the synthetic route and utilizing commodity feedstocks, manufacturers can achieve significant cost reductions in electronic chemical manufacturing while enhancing supply security.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts like SmI2 removes the need for costly metal scavenging steps and specialized waste disposal protocols associated with heavy metals. Furthermore, the use of inexpensive starting materials such as cyclohexanone and o-phenylenediamine, compared to specialized precursors like diindolylmethane, drastically lowers the Bill of Materials (BOM). The mild reaction conditions (room temperature) also reduce energy consumption related to heating and cooling, contributing to substantial overall operational expenditure (OPEX) savings without compromising output quality.
- Enhanced Supply Chain Reliability: Relying on globally available commodity chemicals mitigates the risk of supply disruptions often associated with niche reagents. The robustness of the electrochemical cell setup allows for continuous processing capabilities, which can significantly reduce lead times for high-purity OLED material intermediates. Additionally, the simplified workup procedure involving standard solvent extraction and recrystallization ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term contracts with downstream display manufacturers who require stringent quality assurance.
- Scalability and Environmental Compliance: The transition-metal-free nature of this process aligns perfectly with increasingly strict global environmental regulations regarding heavy metal discharge. By avoiding the generation of large volumes of acidic waste typical of the Fisher indole route, facilities can lower their environmental compliance costs and simplify permitting processes. The inherent safety of operating at ambient pressure and temperature further facilitates the commercial scale-up of complex heterocyclic compounds, allowing for safer expansion of production capacity to meet growing market demand for OLED components.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of indolo[2,3-a]carbazole. These answers are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a reliable foundation for process evaluation.
Q: What is the purity level achievable with this electrochemical method?
A: According to patent CN114318375A, the method achieves a product content of up to 99.2% after recrystallization, with impurity levels controlled below 1%.
Q: Does this process require expensive transition metal catalysts?
A: No, the process is transition-metal free. It utilizes electrochemical oxidation instead of traditional metal catalysts like SmI2, significantly reducing raw material costs and purification complexity.
Q: What are the optimal reaction conditions for scaling this synthesis?
A: The reaction operates at room temperature with a constant current of 10-15 mA. It uses commercially available solvents like DMF or DMA and simple electrode setups (Pt/C or Pb/Pt), facilitating easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolo[2,3-a]carbazole Supplier
As the demand for high-performance organic semiconductors continues to surge, securing a stable supply of key intermediates like indolo[2,3-a]carbazole is paramount for maintaining competitive advantage in the display industry. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging advanced electrochemical synthesis platforms to deliver superior value. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for OLED applications.
We invite you to collaborate with us to optimize your supply chain for next-generation display materials. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our electrochemical route can enhance your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to drive innovation in the electronic materials sector.
