Advanced Enzymatic Resolution for High-Purity Ruxolitinib Intermediates and Commercial Scale-Up
Advanced Enzymatic Resolution for High-Purity Ruxolitinib Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing chiral centers in complex active pharmaceutical ingredients (APIs). Patent CN114214375A introduces a groundbreaking preparation method for (R)-3-(4-halo-1H-pyrazol-1-yl)-3-cyclopentyl propionate and its subsequent conversion to the corresponding nitrile, which serves as a critical fragment in the synthesis of Ruxolitinib. This JAK1/2 inhibitor represents a vital therapy for myelofibrosis, making the efficient production of its intermediates a high-priority objective for global supply chains. The disclosed technology leverages selective lipase-catalyzed hydrolysis in a phosphate buffer system to achieve exceptional stereochemical control, bypassing the limitations of traditional metal-catalyzed hydrogenation or chiral column separation. By operating under mild reaction conditions ranging from 25°C to 40°C, this biocatalytic approach not only ensures high enantiomeric excess values but also aligns with green chemistry principles essential for modern regulatory compliance.
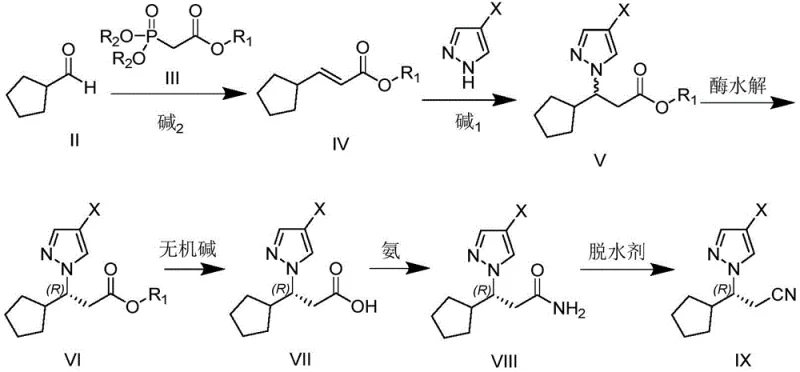
This novel pathway addresses the longstanding challenges associated with constructing the chiral center adjacent to the cyclopentyl ring, a structural motif that has historically required expensive chiral ligands or cumbersome resolution techniques. The integration of enzymatic resolution allows for the direct isolation of the desired (R)-configuration ester with high purity, which can then be seamlessly converted into the nitrile functionality through a streamlined sequence of hydrolysis, amidation, and dehydration. For procurement and technical teams evaluating potential partners, this patent signifies a shift towards more sustainable and cost-effective manufacturing paradigms. The ability to utilize readily available starting materials such as cyclopentanecarbaldehyde and various phosphonoacetates further enhances the commercial viability of this route, positioning it as a superior alternative for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ruxolitinib intermediates has been plagued by significant technical and economic hurdles that impede large-scale production efficiency. Prior art methods, such as those disclosed in earlier patents, often rely on asymmetric hydrogenation using precious metal catalysts like ruthenium or rhodium complexes equipped with chiral phosphine ligands. While chemically feasible, these routes suffer from low atom utilization rates and require multiple crystallization steps to achieve qualified chiral purity, leading to substantial raw material loss. Furthermore, alternative approaches utilizing chiral preparative columns for separation are economically prohibitive due to the high cost of stationary phases and the low throughput inherent to chromatographic processes. Other reported strategies involving chemical resolution with chiral amines frequently exhibit poor splitting yields, often reported in the range of 30-35%, which drastically inflates the cost of goods and generates excessive waste streams that complicate environmental compliance.
The Novel Approach
In stark contrast, the methodology presented in CN114214375A utilizes a highly selective biocatalytic strategy that fundamentally redefines the efficiency of chiral construction for this specific molecular scaffold. By employing selective lipases, such as Candida antarctica Lipase B or enzymes derived from Humicola, the process achieves kinetic resolution directly in an aqueous phosphate buffer system. This eliminates the need for expensive transition metals and harsh reaction conditions, thereby simplifying the downstream purification workflow significantly. The enzymatic hydrolysis selectively targets the (S)-enantiomer of the racemic ester, leaving the desired (R)-enantiomer intact with exceptional stereoselectivity. This approach not only streamlines the synthetic sequence but also dramatically improves the overall yield and optical purity, making it uniquely suitable for industrial scale-up production where consistency and cost control are paramount concerns for supply chain stability.
Mechanistic Insights into Enzymatic Kinetic Resolution
The core innovation of this technology lies in the precise mechanistic action of the selective lipase within a controlled phosphate buffer environment. The reaction operates on the principle of enantioselective ester hydrolysis, where the enzyme's active site exhibits a profound preference for the (S)-configuration of the 3-(4-halo-1H-pyrazol-1-yl)-3-cyclopentyl propionate substrate. Upon exposure to the biocatalyst, the (S)-enantiomer undergoes rapid hydrolysis to form the corresponding carboxylic acid, while the (R)-enantiomer remains largely unreacted as the ester. This differential reaction rate allows for the effective separation of the two enantiomers simply by adjusting the pH and performing an extraction, as the acid and ester possess distinct solubility profiles. The use of a phosphate buffer with a pH maintained between 7.0 and 8.0 is critical for maintaining enzyme stability and activity, ensuring that the biocatalyst functions optimally throughout the reaction duration without denaturation.
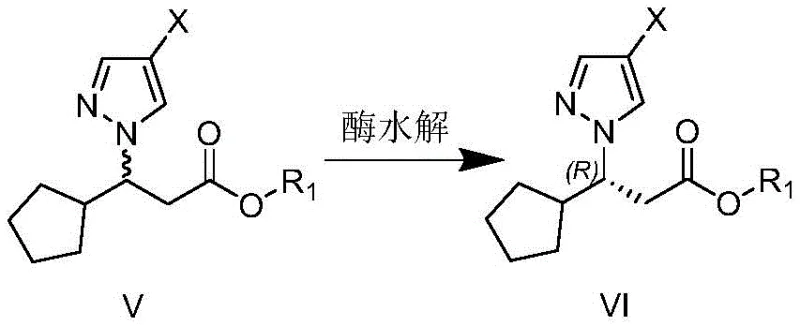
Furthermore, the impurity profile of the resulting product is significantly cleaner compared to metal-catalyzed routes, as the enzymatic process avoids the formation of heavy metal residues that require rigorous and costly removal steps. The specificity of the lipase minimizes side reactions such as over-hydrolysis or racemization, which are common pitfalls in chemical resolution methods. By fine-tuning parameters such as substrate concentration, typically optimized between 100 g/L and 200 g/L, and temperature, ideally kept between 30°C and 40°C, manufacturers can maximize the conversion rate while preserving the 100% ee value of the isolated (R)-ester. This level of control over the reaction microenvironment demonstrates a sophisticated understanding of biocatalysis that translates directly into higher batch consistency and reduced risk of off-specification material, a key metric for R&D directors evaluating process robustness.
How to Synthesize (R)-3-(4-halo-1H-pyrazol-1-yl)-3-cyclopentyl propionate Efficiently
The synthesis begins with the preparation of the racemic precursor via a Horner-Wadsworth-Emmons reaction followed by a Michael addition, setting the stage for the critical enzymatic resolution step. Detailed operational parameters regarding reagent ratios, solvent systems, and workup procedures are essential for replicating the high yields and purity described in the patent documentation. The subsequent transformation of the resolved ester into the final nitrile involves a carefully orchestrated sequence of base-mediated hydrolysis, activation with carbonyldiimidazole, ammonolysis, and final dehydration. Understanding the interplay between these chemical steps and the preceding biocatalytic step is vital for process engineers aiming to implement this technology. For a comprehensive breakdown of the specific experimental conditions and stoichiometry required to execute this synthesis successfully, please refer to the standardized guide below.
- Perform Horner-Wadsworth-Emmons reaction between cyclopentanecarbaldehyde and phosphonoacetate to form 3-cyclopentyl acrylate.
- Conduct Michael addition with 4-halopyrazole to generate the racemic ester precursor.
- Execute enzymatic kinetic resolution using selective lipase in phosphate buffer to isolate the (R)-enantiomer ester.
- Convert the resolved ester to the final nitrile via hydrolysis, amidation, and dehydration steps.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this enzymatic resolution technology offers transformative benefits that directly address the pain points of cost, reliability, and scalability in pharmaceutical manufacturing. Traditional methods relying on chiral chromatography or precious metal catalysts introduce significant volatility into the supply chain due to the fluctuating costs of rare earth metals and the limited capacity of purification columns. By shifting to a biocatalytic platform, manufacturers can leverage renewable enzyme sources and aqueous reaction media, which drastically simplifies waste management and reduces the environmental footprint of the production facility. This transition not only mitigates regulatory risks associated with heavy metal limits in APIs but also ensures a more predictable cost structure by eliminating dependencies on volatile commodity markets for specialized catalysts.
- Cost Reduction in Manufacturing: The elimination of expensive chiral ligands and precious metal catalysts results in a substantial decrease in raw material expenditures. Additionally, the high selectivity of the enzymatic process reduces the need for extensive purification steps, lowering solvent consumption and energy usage associated with distillation and crystallization. The ability to recycle the unreacted (S)-acid back into the racemic mixture further enhances atom economy, ensuring that raw material inputs are utilized to their maximum potential. This holistic reduction in processing complexity translates directly into a lower cost of goods sold, providing a competitive edge in pricing negotiations for long-term supply contracts.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as cyclopentanecarbaldehyde and common phosphonates ensures that the supply chain is resilient against shortages of exotic reagents. Enzymes used in this process are produced via fermentation on an industrial scale, guaranteeing a consistent and secure supply of the biocatalyst. The robustness of the reaction conditions, which tolerate a range of temperatures and pH levels, means that production can continue with minimal disruption even if minor variations in utility supplies occur. This stability is crucial for maintaining continuous manufacturing operations and meeting the stringent delivery schedules demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The aqueous nature of the enzymatic resolution step facilitates easy scale-up from laboratory to commercial production volumes without the need for specialized high-pressure equipment. The process generates significantly less hazardous waste compared to chemical resolution methods, simplifying effluent treatment and reducing disposal costs. Compliance with increasingly strict environmental regulations is inherently easier with this green chemistry approach, as it avoids the generation of toxic heavy metal sludge. This alignment with sustainability goals not only future-proofs the manufacturing asset but also enhances the brand reputation of the supplier as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on the practical aspects of adopting this technology. Understanding these nuances is essential for stakeholders making strategic decisions about process development and vendor selection. The answers reflect the proven capabilities of the method in terms of purity, yield, and operational flexibility.
Q: Why is enzymatic resolution preferred over chiral chromatography for this intermediate?
A: Enzymatic resolution offers significantly lower operational costs and higher scalability compared to chiral preparation columns, which suffer from low efficiency and prohibitive costs for industrial production.
Q: What represents the critical quality attribute in this synthesis route?
A: The enantiomeric excess (ee value) is the critical quality attribute, with the patented enzymatic method achieving up to 100% ee, ensuring high optical purity required for API synthesis.
Q: Which halogen substituents are compatible with this enzymatic process?
A: The process is robust across various halogen substituents, specifically supporting chlorine, bromine, and iodine at the 4-position of the pyrazole ring without compromising resolution efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruxolitinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the enzymatic resolution described in CN114214375A can be seamlessly translated into robust manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest standards of quality and safety. Our commitment to technical excellence allows us to navigate the complexities of chiral synthesis with precision, delivering products that facilitate the efficient development of life-saving medications.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages inherent in this enzymatic approach. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to serve as a dependable source for high-value pharmaceutical intermediates. Together, we can drive innovation and efficiency in the supply of critical drug substances.
