Advanced Synthesis of Paclitaxel Side Chain: A Scalable Route for Global API Manufacturing
Advanced Synthesis of Paclitaxel Side Chain: A Scalable Route for Global API Manufacturing
The global demand for paclitaxel, a first-grade drug for treating malignant tumors such as advanced ovarian cancer and breast cancer, continues to surge, driving the need for efficient and scalable synthesis of its critical side chain. Patent CN111763179A, published in October 2020, introduces a robust synthesis method that addresses the longstanding challenges of low yield and high cost associated with natural extraction and older semi-synthetic routes. This technology leverages a strategic oxazolidine protection group to ensure high stereoselectivity and purity, starting from the readily available (2R,3S)-3-phenylisoserine hydrochloride. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediates supplier, this patent represents a significant leap forward in process chemistry, offering a pathway that is not only chemically elegant but also industrially viable for commercial scale-up of complex chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of paclitaxel side chains has been plagued by significant technical and economic bottlenecks that hinder mass production. Traditional methods often rely on the resolution of racemic mixtures, which inherently limits the maximum theoretical yield to 50% and generates substantial waste, leading to high product prices that burden the healthcare system. Furthermore, alternative synthetic routes involving the ring-opening of epoxy compounds by ammonia or azide compounds frequently suffer from poor regioselectivity. This lack of selectivity results in the formation of large amounts of impurities and isomers, creating a nightmare for downstream purification teams who must employ costly and time-consuming chromatographic techniques to achieve pharmaceutical-grade purity. Additionally, the use of azide compounds introduces severe safety risks due to their explosive nature, making these processes unsuitable for large-scale manufacturing environments where operator safety and regulatory compliance are paramount.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a streamlined four-step sequence that prioritizes safety, efficiency, and ease of operation. The process begins with a straightforward esterification of the starting material, followed by benzoylation, setting the stage for the critical cyclization step. By employing a specific oxazolidine protection strategy, the synthesis effectively locks the stereochemistry of the molecule, preventing the formation of unwanted isomers that plague epoxy-based routes. This method eliminates the need for dangerous azide reagents and avoids the requirement for inert gas protection, allowing reactions to proceed under standard atmospheric conditions. The simplicity of the workup procedures, which rely on standard extraction and recrystallization rather than complex column chromatography, drastically reduces the production period and operational costs. This makes the technology highly attractive for cost reduction in API manufacturing while ensuring a consistent supply of high-purity paclitaxel side chain material.
Mechanistic Insights into PPTS-Catalyzed Cyclization and Protection
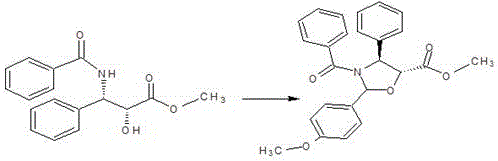
The cornerstone of this synthesis is the formation of the (4S, 5R)-5-methoxycarbonyl-2-(4-methoxyphenyl)-4-phenyl-3-benzoyl-1,3-oxazolidine intermediate, a step that dictates the overall stereochemical integrity of the final product. This cyclization protection reaction utilizes pyridinium p-toluenesulfonate (PPTS) as a mild yet effective acid catalyst in anhydrous toluene. The mechanism involves the condensation of the N-benzoyl-phenylisoserine methyl ester with 4-methoxybenzaldehyde dimethyl acetal. The PPTS catalyst facilitates the generation of an reactive oxocarbenium ion species from the acetal, which is then attacked by the beta-hydroxyl group of the serine derivative. This intramolecular nucleophilic attack forms the five-membered oxazolidine ring, effectively protecting both the amine and the hydroxyl functionalities simultaneously. The choice of 4-methoxybenzaldehyde dimethyl acetal is crucial, as the electron-donating methoxy group stabilizes the intermediate cation, promoting high conversion rates at moderate reflux temperatures of 60-68°C. This precise control over reaction conditions prevents epimerization at the chiral centers, ensuring that the (2R,3S) configuration of the starting material is faithfully transferred to the protected intermediate.
Following the cyclization, the control of impurities is managed through the robustness of the oxazolidine ring itself, which is stable under the subsequent reaction conditions but easily removable in the final step. The purification efficiency is enhanced by the physical properties of the oxazolidine intermediate, which allows for effective separation from unreacted starting materials and byproducts via simple aqueous workups and concentration. The final hydrolysis step employs a mild sodium carbonate solution in methanol, which selectively cleaves the oxazolidine ring and the methyl ester without affecting the sensitive beta-lactam-like structure or causing racemization. The pH is carefully adjusted to neutrality using dilute hydrochloric acid, precipitating the crude product which is then purified by recrystallization from a toluene and n-hexane mixture. This recrystallization step is critical for removing trace organic impurities and residual solvents, yielding a finished product with a content typically exceeding 90%, suitable for direct coupling with baccatin III derivatives in the final assembly of paclitaxel.
How to Synthesize Paclitaxel Side Chain Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The process is designed to be executed in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment. The initial esterification and benzoylation steps set the foundation, but the success of the entire campaign hinges on the precise execution of the cyclization and hydrolysis phases. Operators must maintain strict temperature controls during the reflux and ensure accurate pH adjustments during the workup to maximize yield and purity. The detailed standardized synthesis steps below provide the specific molar ratios, solvent volumes, and reaction times required to replicate the high yields reported in the patent examples, serving as an essential guide for process engineers aiming to implement this technology.
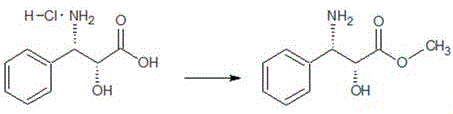
- Perform esterification of (2R,3S)-3-phenylisoserine hydrochloride using thionyl chloride and methanol to obtain the methyl ester.
- Conduct benzoylation of the methyl ester with benzoyl chloride in dichloromethane to form N-benzoyl-phenylisoserine methyl ester.
- Execute cyclization protection using 4-methoxybenzaldehyde dimethyl acetal and PPTS catalyst in toluene to form the oxazolidine intermediate.
- Complete the synthesis via hydrolysis with sodium carbonate followed by acidification and recrystallization to yield the final paclitaxel side chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible benefits that extend beyond mere chemical yield. The elimination of hazardous azide reagents removes a major regulatory and safety hurdle, simplifying the permitting process and reducing insurance costs associated with handling explosive materials. Furthermore, the reliance on common, commercially available raw materials such as phenylisoserine hydrochloride and benzoyl chloride ensures a stable supply chain that is less susceptible to the volatility often seen with exotic or custom-synthesized reagents. The ability to run reactions without inert gas protection significantly lowers the capital expenditure (CapEx) required for reactor infrastructure, as standard agitation and heating systems suffice, avoiding the need for complex nitrogen blanketing systems. These factors combine to create a manufacturing process that is not only safer but also economically superior, driving down the cost of goods sold (COGS) for the final API.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need for expensive transition metal catalysts and complex purification technologies like preparative HPLC. The use of simple recrystallization for final purification drastically reduces solvent consumption and waste disposal costs compared to chromatographic methods. Additionally, the high atom economy of the cyclization step minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into valuable product. This efficiency translates directly into lower production costs, allowing for more competitive pricing in the global market for paclitaxel intermediates.
- Enhanced Supply Chain Reliability: By utilizing starting materials that are conventional products and easy to purchase, the risk of supply disruption is markedly reduced. The synthesis does not depend on single-source suppliers for critical reagents, providing procurement teams with the flexibility to source from multiple vendors to mitigate risk. The robustness of the reaction conditions, which tolerate standard industrial environments without requiring ultra-dry or anaerobic conditions, further enhances reliability by reducing the likelihood of batch failures due to minor environmental fluctuations. This stability ensures consistent delivery schedules, a critical factor for maintaining continuous API production lines.
- Scalability and Environmental Compliance: The method is explicitly designed for industrial application, with reaction parameters that scale linearly from laboratory to commercial production. The absence of heavy metals and toxic azides simplifies wastewater treatment and aligns with increasingly stringent environmental regulations regarding pharmaceutical manufacturing. The solvent system, primarily consisting of methanol, toluene, and ethyl acetate, allows for efficient recovery and recycling, minimizing the environmental footprint of the process. This alignment with green chemistry principles not only ensures compliance but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this paclitaxel side chain synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing workflows. The clarity provided here aims to bridge the gap between academic patent language and practical industrial application.
Q: Why is the oxazolidine protection strategy superior to traditional epoxy ring-opening methods?
A: Traditional epoxy ring-opening methods often suffer from low regioselectivity, generating significant impurities and isomers that are difficult to purify. The oxazolidine protection strategy described in patent CN111763179A ensures high stereoselectivity and simplifies downstream purification, resulting in a final product with content exceeding 90% without complex chromatography.
Q: Does this synthesis method require inert gas protection or harsh conditions?
A: No, one of the primary advantages of this novel approach is that it does not require inert gas protection. The reactions can be conducted under standard atmospheric conditions at moderate temperatures (e.g., reflux at 60-68°C), which significantly lowers equipment requirements and operational complexity compared to methods requiring strict anhydrous or anaerobic environments.
Q: What are the safety benefits of this route compared to azide-based synthesis?
A: Azide-based synthesis routes involve hazardous azide compounds that pose huge potential safety hazards during production and storage. This patent utilizes safe, commercially available raw materials like phenylisoserine hydrochloride and avoids explosive or toxic reagents, making it much more suitable for large-scale industrial application and market popularization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against cancer. Our team of expert chemists has extensively analyzed the route described in CN111763179A and possesses the technical capability to optimize and scale this process to meet your specific volume requirements. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of paclitaxel side chain we deliver meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this advanced synthesis technology for your API production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current supply chain structure, demonstrating exactly how switching to this method can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a reliable, cost-effective, and high-quality supply of this vital oncology intermediate.
