Revolutionizing Paclitaxel Production: A One-Step Three-Component Reaction Strategy for High-Purity Intermediates
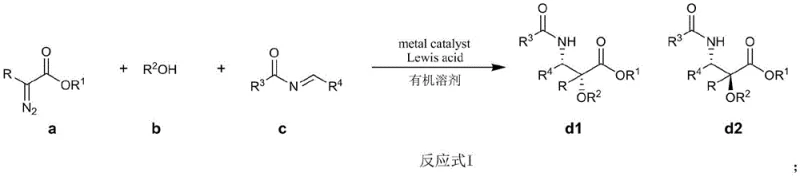
The pharmaceutical industry continuously seeks more efficient pathways for producing critical oncology therapeutics, and Paclitaxel (Taxol) remains a cornerstone in cancer treatment. Patent CN114573631A introduces a groundbreaking methodology for the one-step synthesis of the Paclitaxel C-13 side chain and its analogs using a novel three-component reaction. This technology addresses the long-standing challenges of complex multi-step syntheses by integrating a diazo compound, an alcohol, and an imine into a single catalytic cycle. For R&D directors and procurement specialists, this represents a significant shift towards greener, more atom-economical manufacturing processes that can drastically simplify the supply chain for high-value pharmaceutical intermediates. The ability to construct the chiral side chain in a single operation not only reduces waste but also enhances the overall feasibility of large-scale production.
Traditional methods for synthesizing the Paclitaxel side chain often involve cumbersome multi-step sequences requiring harsh reaction conditions, expensive chiral auxiliaries, and extensive purification protocols. These legacy processes frequently suffer from low overall yields and generate substantial chemical waste, creating bottlenecks in cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the reliance on multiple isolation steps increases the risk of impurity accumulation and extends lead times, which is detrimental to maintaining a robust supply chain for essential anti-cancer drugs. The complexity of protecting group manipulations in conventional routes further exacerbates these inefficiencies, making the transition from lab scale to commercial production both risky and capital intensive.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic routes for the Paclitaxel C-13 side chain typically rely on the condensation of protected phenylisoserine derivatives, which requires multiple protection and deprotection steps. These processes often utilize stoichiometric amounts of reagents and generate significant by-products, leading to poor atom economy. The need for cryogenic conditions in certain steps and the use of hazardous reagents pose safety risks and increase energy consumption. Additionally, achieving high stereoselectivity in traditional methods often demands expensive chiral catalysts or resolution steps, which further drives up the cost of goods sold (COGS). For supply chain managers, these factors translate into longer production cycles and higher vulnerability to raw material price fluctuations, making the consistent supply of high-purity pharmaceutical intermediates challenging to guarantee.
The Novel Approach
In stark contrast, the method disclosed in CN114573631A utilizes a direct three-component coupling reaction involving a diazo compound (a), an alcohol (b), and an imine (c). This approach leverages transition metal catalysis to facilitate O-H insertion and subsequent C-N bond formation in a concerted manner. The reaction proceeds under mild conditions, typically between -10°C and 30°C, eliminating the need for extreme thermal inputs. By employing a composite catalyst system comprising a metal catalyst (such as rhodium acetate) and a Lewis acid (such as silver hexafluorophosphate), the process achieves high efficiency and selectivity. This streamlined strategy significantly reduces the number of unit operations, thereby lowering operational costs and minimizing the environmental footprint associated with solvent use and waste disposal.
Mechanistic Insights into Rhodium-Catalyzed Multicomponent Coupling
The core of this innovation lies in the generation of a reactive metal-carbene intermediate from the diazo precursor. Upon decomposition of the diazo compound by the rhodium catalyst, a highly electrophilic metal-carbene species is formed. This species undergoes rapid O-H insertion with the alcohol component to generate an oxonium ylide or a similar reactive intermediate. Subsequently, this intermediate reacts with the imine component to form the final beta-amino ester structure characteristic of the Paclitaxel side chain. The presence of the Lewis acid is crucial for activating the imine and stabilizing the transition states, thereby controlling the stereochemical outcome of the reaction. Experimental data from the patent indicates that varying the Lewis acid can tune the diastereomeric ratio (dr), with certain silver salts yielding dr values favoring the desired isomer.
Impurity control is inherently built into this mechanism due to the high chemoselectivity of the rhodium catalyst. Unlike traditional methods where side reactions at multiple functional groups are common, this catalytic system specifically targets the diazo and imine functionalities. The use of molecular sieves as a water scavenger further prevents hydrolysis of sensitive intermediates, ensuring high product purity. The reaction tolerates a wide range of substituents on the diazo, alcohol, and imine components, allowing for the synthesis of diverse analogs without compromising yield. This robustness is essential for commercial scale-up of complex pharmaceutical intermediates, as it ensures consistent quality even when scaling from grams to kilograms. The ability to fine-tune the catalyst loading and reaction temperature provides additional levers for process optimization to meet stringent regulatory specifications.
How to Synthesize Paclitaxel Side Chain Efficiently
The synthesis protocol outlined in the patent offers a practical guide for implementing this technology in a GMP environment. The process begins with the preparation of a catalyst mixture containing the imine, rhodium catalyst, Lewis acid, and molecular sieves in an anhydrous organic solvent such as dichloromethane. A separate solution containing the diazo compound and alcohol is then added slowly to the catalyst mixture to control the exotherm and maintain steady-state concentration of the reactive carbene. Following the reaction, standard workup procedures involving filtration and solvent removal are sufficient to isolate the crude product, which can be purified via column chromatography. This straightforward procedure minimizes the need for specialized equipment, making it accessible for various manufacturing scales.
- Prepare Solution A by suspending the imine substrate, rhodium catalyst (e.g., rhodium acetate), Lewis acid (e.g., silver hexafluorophosphate), and molecular sieves in an organic solvent like dichloromethane under inert gas protection.
- Prepare Solution B by dissolving the diazo compound and the alcohol component (e.g., triethylsilanol) in a compatible organic solvent.
- Slowly add Solution B to Solution A at controlled temperatures between -10°C and 30°C, stir until completion, then purify the resulting side chain products via filtration and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this three-component reaction technology offers tangible strategic benefits beyond mere technical novelty. The consolidation of multiple synthetic steps into a single operation directly translates to reduced manufacturing lead times and lower inventory holding costs. By eliminating the need for intermediate isolations and the associated solvent exchanges, the process significantly cuts down on utility consumption and waste treatment expenses. The use of readily available starting materials, such as ethyl diazoacetate and benzaldehyde derivatives, ensures a stable and reliable supply base, mitigating the risk of raw material shortages that often plague complex synthetic routes. This stability is critical for maintaining continuous production schedules for life-saving medications.
- Cost Reduction in Manufacturing: The primary driver for cost savings is the drastic reduction in process steps. Traditional routes may require five or more steps to achieve the same structural complexity, each incurring labor, energy, and material costs. By condensing this into a one-pot reaction, the operational expenditure is significantly lowered. Furthermore, the high atom economy of the reaction means that a larger proportion of the raw material mass ends up in the final product, reducing the cost per kilogram of the active intermediate. The elimination of expensive chiral resolving agents or auxiliaries, replaced by catalytic stereoselection, further contributes to a leaner cost structure without sacrificing quality.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions allows for greater flexibility in manufacturing locations. Since the reaction does not require extreme temperatures or pressures, it can be performed in standard stainless steel reactors available in most multipurpose plants. This flexibility reduces dependency on specialized facilities and broadens the pool of potential contract manufacturing organizations (CMOs). Additionally, the robustness of the catalyst system against minor variations in feedstock quality ensures consistent output, reducing the frequency of batch failures and reworks. This reliability is paramount for reducing lead time for high-purity pharmaceutical intermediates and ensuring timely delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this reaction is straightforward due to the absence of hazardous reagents and the mild thermal profile. The reduced solvent usage per kilogram of product aligns with green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations. Lower waste generation simplifies the permitting process for new production lines and reduces the liability associated with hazardous waste disposal. The ability to recycle the organic solvents used in the reaction further enhances the sustainability profile of the process, making it an attractive option for companies aiming to reduce their carbon footprint while maintaining economic efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the key advantages of this three-component reaction over traditional methods?
A: This method significantly reduces synthetic steps by combining diazo, alcohol, and imine components in a single pot. It operates under mild conditions (room temperature) and utilizes commercially available catalysts, leading to improved atom economy and simplified purification compared to multi-step traditional routes.
Q: Can this method be applied to the total synthesis of Paclitaxel?
A: Yes, the patent describes applying this side chain synthesis as a key step in a two-step semi-synthesis of Paclitaxel. By replacing the simple diazo component with a 7-TES-baccatin diazo derivative, the method efficiently constructs the final Taxol molecule.
Q: What catalysts are recommended for optimal yield and stereoselectivity?
A: The patent highlights rhodium-based catalysts, specifically rhodium acetate, combined with silver-containing Lewis acids like silver hexafluorophosphate or silver trifluoromethanesulfonate. These combinations have demonstrated high yields and favorable diastereomeric ratios in experimental examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in CN114573631A for the global oncology market. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory methods into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and compliant with international standards. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Paclitaxel side chain meets the highest quality requirements for API synthesis.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis route for their Paclitaxel programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can optimize your supply chain and accelerate your time to market.
