Advanced Biocatalytic Route for High-Purity Chiral Amino Alcohol Intermediates
Advanced Biocatalytic Route for High-Purity Chiral Amino Alcohol Intermediates
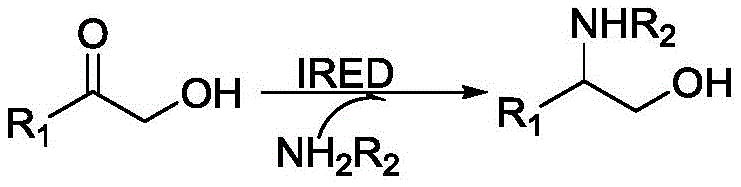
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to access high-value chiral building blocks. Patent CN112852771A introduces a groundbreaking biocatalytic method for the synthesis of optically pure chiral amino alcohol compounds, utilizing imine reductase (IRED) to catalyze the reductive amination of alpha-hydroxy ketones. This technology represents a significant leap forward from traditional chemical synthesis, offering a route that operates under mild physiological conditions while delivering exceptional stereoselectivity. By leveraging genetically engineered bacteria capable of expressing specific imine reductase variants, manufacturers can achieve precise control over the chirality of the final product, producing either R or S configurations with high enantiomeric excess. This innovation addresses critical pain points in API intermediate manufacturing, providing a reliable pharmaceutical intermediate supplier with a tool to enhance both product quality and process sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral amino alcohols has relied heavily on chemical methods that often involve harsh reaction conditions and toxic reagents. Common approaches include the reduction of amino acids or their derivatives using strong reducing agents like lithium aluminum hydride (LiAlH4), which poses significant safety hazards and requires stringent anhydrous conditions. Another classical route involves the Henry reaction followed by reduction, or the ring-opening of epoxides with amines; however, these methods frequently suffer from poor stereoselectivity, necessitating costly and yield-limiting chiral resolution steps to isolate the desired enantiomer. Furthermore, chemical catalysis often employs transition metals that can leave trace impurities in the final product, creating substantial downstream purification burdens to meet strict regulatory limits for pharmaceutical applications. These conventional processes are not only environmentally taxing due to the generation of hazardous waste but also economically inefficient due to low overall yields and complex work-up procedures.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a biocatalytic strategy that fundamentally transforms the synthesis landscape for these valuable intermediates. By employing imine reductases derived from a screened library of genetically engineered bacteria, the process achieves direct reductive amination of alpha-hydroxy ketones with small molecule amines in an aqueous environment. This method eliminates the need for hazardous chemical reducing agents and operates at mild temperatures (25-30°C) and neutral to slightly alkaline pH levels, significantly reducing energy consumption and safety risks. The enzymatic route inherently possesses high stereoselectivity, often yielding products with an enantiomeric excess (ee) greater than 99 percent, thereby removing the necessity for difficult chiral separations. This biological catalysis not only simplifies the synthetic workflow but also aligns with green chemistry principles, offering a cleaner and more atom-economical alternative for the production of complex chiral amino alcohols used in drug discovery and development.
Mechanistic Insights into Imine Reductase Catalyzed Reductive Amination
The core of this technological advancement lies in the specific catalytic mechanism of the imine reductase enzyme, which facilitates the asymmetric reduction of the transient imine intermediate formed between the alpha-hydroxy ketone and the amine. The enzyme binds the substrate in a highly specific orientation within its active site, ensuring that the hydride transfer from the cofactor NADPH occurs exclusively to one face of the imine double bond, thus dictating the stereochemical outcome of the reaction. To make this process industrially viable, the patent describes a coupled cofactor regeneration system using glucose dehydrogenase (GDH) and glucose, which continuously recycles the oxidized NADP+ back to NADPH, driving the reaction to completion without the need for stoichiometric amounts of expensive cofactors. This elegant cascade ensures high substrate conversion rates, often exceeding 90 percent, while maintaining the integrity of the sensitive hydroxyl group present in the substrate. The ability to tune the stereochemistry by selecting specific enzyme variants (e.g., IR-27 for S-configuration vs. IR-36 for R-configuration) provides unparalleled flexibility for synthesizing diverse chiral libraries.

Furthermore, the impurity profile of the biocatalytic process is markedly superior to chemical alternatives, primarily because the enzyme exhibits high chemoselectivity towards the imine functionality. Unlike chemical reducers that might indiscriminately reduce other functional groups such as esters or nitro groups potentially present in complex molecules, the imine reductase targets only the specific C=N bond formed in situ. This specificity minimizes the formation of side products and byproducts, resulting in a crude reaction mixture that is much easier to purify. The use of whole cells as biocatalysts further simplifies the process, as the cellular matrix can protect the enzyme and stabilize the reaction environment, while the subsequent work-up involving simple extraction and salification allows for the isolation of the product as a stable hydrochloride salt. This level of control over the reaction pathway ensures that the final high-purity pharmaceutical intermediate meets the rigorous quality standards required for clinical applications.
How to Synthesize Chiral Amino Alcohol Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these compounds, starting with the cultivation of the specific imine reductase-producing strains in a standard fermentation medium. Once the biomass is harvested, the whole cells are resuspended in a phosphate buffer and mixed with the alpha-hydroxy ketone substrate and the chosen amine donor, initiating the biotransformation. The detailed standardized synthesis steps见下方的指南 ensure reproducibility and high yield, making this method accessible for both laboratory scale optimization and large-scale manufacturing.
- Cultivate genetically engineered bacteria expressing specific imine reductase variants (e.g., IR-27, IR-36) in a fermentation medium containing carbon and nitrogen sources.
- Prepare the reaction system by resuspending the whole cells in a phosphate buffer (pH 8.0-10.0) and adding the alpha-hydroxy ketone substrate along with the amine donor.
- Initiate the reductive amination by adding a cofactor regeneration system (Glucose/GDH/NADP+) and maintain the reaction at 25-30°C until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology offers compelling economic and operational benefits that extend beyond mere technical feasibility. The shift from chemical to enzymatic synthesis fundamentally alters the cost structure of manufacturing chiral amino alcohols by eliminating the need for expensive chiral ligands, precious metal catalysts, and hazardous reagents that drive up raw material costs. Additionally, the simplified downstream processing required due to the high purity of the enzymatic reaction mixture translates into significant cost reduction in pharmaceutical intermediate manufacturing, as fewer purification columns and solvent exchanges are needed to meet specification. This efficiency gain allows suppliers to offer more competitive pricing while maintaining healthy margins, creating a win-win scenario for both the manufacturer and the buyer.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and chiral resolving agents drastically reduces the bill of materials, while the mild reaction conditions lower energy consumption associated with heating and cooling. By avoiding the use of toxic solvents and reagents, the costs related to hazardous waste disposal and environmental compliance are also substantially decreased, leading to a leaner and more cost-effective production model. The high conversion rates achieved by the engineered enzymes mean that less raw material is wasted, further optimizing the overall process economics and maximizing the yield of the valuable chiral product.
- Enhanced Supply Chain Reliability: Relying on fermentation-based production of the biocatalyst ensures a consistent and renewable supply of the key processing agent, reducing dependency on volatile markets for rare earth metals or specialized chemical reagents. The robustness of the whole-cell biocatalysts allows for flexible manufacturing schedules and easier scale-up from pilot to commercial production without the need for specialized high-pressure or cryogenic equipment. This stability in the supply chain mitigates the risk of production delays caused by raw material shortages, ensuring a steady flow of high-quality intermediates to meet the demanding timelines of drug development projects.
- Scalability and Environmental Compliance: The aqueous nature of the reaction and the use of biodegradable components align perfectly with increasingly stringent global environmental regulations, facilitating smoother regulatory approvals and reducing the carbon footprint of the manufacturing process. The process is inherently scalable, as fermentation technologies are well-established in the industry, allowing for seamless expansion from 100 kgs to 100 MT annual commercial production capacities. This scalability ensures that the supply can grow in tandem with the commercial success of the downstream drug, providing long-term security for partners investing in this technology.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical implementation and commercial viability of this enzymatic synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology can be integrated into existing workflows. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this biocatalytic method for their specific project needs.
Q: What are the advantages of using imine reductase over chemical reduction for chiral amino alcohols?
A: Imine reductase offers superior stereoselectivity (often >99% ee) under mild aqueous conditions, eliminating the need for hazardous reducing agents like LiAlH4 and complex chiral resolution steps required in traditional chemical synthesis.
Q: Can this biocatalytic process be scaled for industrial production?
A: Yes, the process utilizes whole-cell biocatalysts which are robust and easily produced via standard fermentation techniques, allowing for scalable manufacturing from kilograms to metric tons without significant process changes.
Q: What types of substrates are compatible with this enzymatic method?
A: The method demonstrates a broad substrate spectrum, successfully converting various alpha-hydroxy ketones including phenyl-substituted and alkyl-substituted derivatives into their corresponding chiral amino alcohols with high conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amino Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic synthesis route described in patent CN112852771A for producing high-value chiral amino alcohol intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale market supply. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to meet stringent purity specifications and deliver consistent quality for every batch of complex pharmaceutical intermediates we produce.
We invite you to collaborate with our technical procurement team to explore how this innovative enzymatic technology can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this biocatalytic route for your specific molecule. Please contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, and let us help you secure a reliable and sustainable source for your critical chiral building blocks.
