Advanced Enzymatic Synthesis of Chiral Amino Alcohols for Scalable Florfenicol Production
Advanced Enzymatic Synthesis of Chiral Amino Alcohols for Scalable Florfenicol Production
The pharmaceutical and agrochemical industries are constantly seeking more efficient and sustainable pathways for synthesizing complex chiral intermediates, particularly for broad-spectrum antibiotics like Florfenicol. Patent CN113322291A introduces a groundbreaking biocatalytic method for synthesizing chiral amino alcohol compounds, which serve as critical precursors in the production of veterinary drugs. This technology leverages a specific carbonyl reductase derived from Rhodotorula toruloides to achieve high stereoselectivity in a single enzymatic step. By shifting away from traditional chemical resolution methods, this innovation addresses key bottlenecks in purity and yield, offering a robust solution for manufacturers aiming to optimize their supply chains for high-value antibiotic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Florfenicol intermediates often rely on complex chemical sequences involving multiple protection and deprotection steps, harsh reagents, and inefficient resolution processes. As illustrated in prior art such as patent CN103936638A, the synthesis typically involves Friedel-Crafts acylation, halogenation, and oxidation steps followed by difficult chiral resolution to isolate the desired enantiomer. These chemical methods frequently suffer from low overall yields due to the inherent 50% loss associated with racemic resolution, alongside significant environmental burdens from heavy metal catalysts and organic solvent waste. The multi-step nature of these conventional pathways increases operational complexity and extends lead times, making it challenging to meet the rigorous demand for cost reduction in antibiotic manufacturing while maintaining high purity standards.
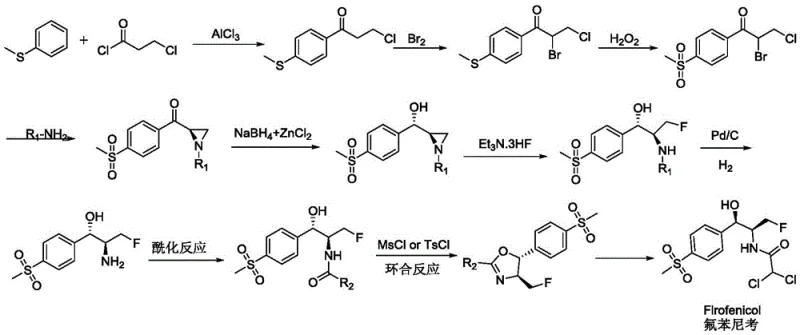
The Novel Approach
In stark contrast, the novel approach detailed in CN113322291A utilizes a highly specific biocatalytic reduction to directly convert a prochiral ketone into the desired chiral amino alcohol with exceptional stereocontrol. This method bypasses the need for chemical resolution entirely by employing a recombinant carbonyl reductase that selectively reduces the carbonyl group to a hydroxyl group with the correct (R,R) configuration. The process operates under mild aqueous conditions, significantly simplifying the workflow and reducing the reliance on hazardous organic solvents. By constructing two chiral centers simultaneously in a single enzymatic transformation, this technology drastically shortens the synthetic route, thereby enhancing production efficiency and facilitating the commercial scale-up of complex veterinary drug intermediates without the yield penalties of traditional chemistry.
Mechanistic Insights into Carbonyl Reductase-Catalyzed Reduction
The core of this technological advancement lies in the unique catalytic mechanism of the carbonyl reductase sourced from Rhodotorula toruloides. This enzyme demonstrates remarkable substrate specificity, recognizing the aziridine ring structure of the precursor and selectively reducing the adjacent ketone moiety. The reaction proceeds via a hydride transfer mechanism where the coenzyme NADH donates a hydride ion to the carbonyl carbon, while a proton is supplied from the solvent or an active site residue, resulting in the formation of the chiral alcohol. The enzyme's active site geometry ensures that the hydride attack occurs from a specific face of the planar carbonyl group, thereby enforcing the formation of the (R)-hydroxyl configuration while preserving the existing chirality of the aziridine ring, leading to the high diastereomeric excess observed in the final product.

Impurity control in this biocatalytic system is inherently superior due to the enzyme's high fidelity, which minimizes the formation of unwanted diastereomers or over-reduced byproducts. The patent specifies that the resulting chiral amino alcohol achieves a diastereomeric excess (de) value of greater than 99%, effectively eliminating the need for downstream chromatographic purification to remove stereoisomers. Furthermore, the integration of a coenzyme regeneration system, typically involving glucose dehydrogenase and glucose, ensures that the expensive cofactor NAD+ is continuously recycled throughout the reaction. This not only lowers the stoichiometric requirement for the cofactor to catalytic levels but also drives the equilibrium towards product formation, ensuring high conversion rates and minimizing the presence of unreacted starting material in the crude mixture.
How to Synthesize Chiral Amino Alcohol Efficiently
Implementing this enzymatic synthesis requires precise control over reaction parameters to maximize enzyme activity and stability. The process begins with the preparation of a whole-cell disruption enzyme solution containing the recombinant carbonyl reductase, which is then introduced into a buffered reaction system. The substrate, a racemic aziridine ketone, is dissolved in a co-solvent system compatible with the enzyme, such as DMSO or methanol, and added to the buffer. Critical to the success of the reaction is the maintenance of a pH around 8.0 using a Tris-HCl buffer and a temperature range of 25°C to 35°C, which provides the optimal environment for the biocatalyst to function. The addition of a coenzyme regeneration system allows the reaction to proceed to completion with minimal cofactor loading, making the process economically viable for large-scale operations.
- Construct a recombinant expression strain by optimizing the carbonyl reductase gene from Rhodotorula toruloides and transforming it into E. coli.
- Prepare the reaction system containing the carbonyl compound substrate, whole cell disruption enzyme solution, coenzyme NAD+, and a glucose dehydrogenase regeneration system in a Tris-HCl buffer.
- Conduct the reduction reaction at controlled pH and temperature, followed by product isolation via centrifugation, extraction, and purification to obtain the chiral amino alcohol.
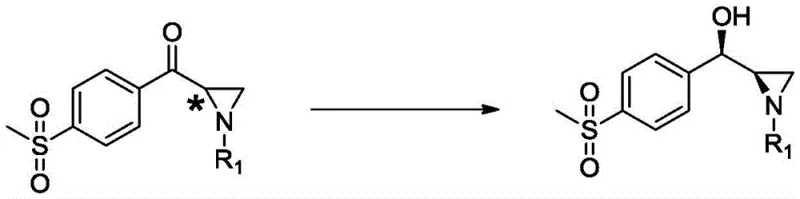
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic route offers transformative benefits in terms of cost structure and supply reliability. By replacing multi-step chemical syntheses with a single enzymatic step, manufacturers can significantly reduce the consumption of raw materials, solvents, and energy, leading to substantial cost savings in veterinary drug intermediate manufacturing. The elimination of chiral resolution steps removes the inherent 50% yield loss associated with discarding the unwanted enantiomer, effectively doubling the theoretical yield from the same amount of starting material. This efficiency gain translates directly into a lower cost of goods sold (COGS) and improved margin potential for downstream API producers, making the supply chain more resilient against fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The enzymatic process eliminates the need for expensive chiral resolving agents and precious metal catalysts often used in asymmetric hydrogenation, thereby lowering direct material costs. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity minimizes waste disposal costs associated with hazardous chemical byproducts. The ability to use whole-cell biocatalysts further reduces enzyme production costs compared to using purified proteins, creating a highly economical production model.
- Enhanced Supply Chain Reliability: Biocatalytic processes are generally more robust and easier to scale than complex chemical syntheses involving sensitive reagents. The use of fermentation-derived enzymes ensures a consistent and renewable supply of the catalyst, reducing dependency on volatile chemical markets. The simplified workflow also shortens the overall production cycle time, allowing for faster response to market demand and reducing the risk of supply disruptions caused by lengthy manufacturing lead times.
- Scalability and Environmental Compliance: This green chemistry approach aligns perfectly with increasingly stringent environmental regulations by reducing the E-factor (mass of waste per mass of product). The aqueous-based reaction system minimizes the use of volatile organic compounds (VOCs), simplifying waste treatment and permitting processes. The high conversion rates and purity reduce the burden on downstream purification units, facilitating smoother scale-up from pilot to commercial production volumes without compromising quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What is the stereoselectivity of the carbonyl reductase used in this process?
A: The carbonyl reductase derived from Rhodotorula toruloides exhibits exceptional stereoselectivity, achieving a diastereomeric excess (de) value of greater than 99% and an enantiomeric excess (ee) of 99.8% for the target chiral amino alcohol.
Q: How is the coenzyme regenerated during the enzymatic reduction?
A: The process utilizes an efficient coenzyme regeneration system involving glucose dehydrogenase and glucose as a co-substrate to continuously regenerate the oxidative coenzyme NAD+, significantly reducing the cost of cofactors.
Q: What are the advantages of this enzymatic method over traditional chemical resolution?
A: Unlike traditional chemical methods that require resolution and multiple steps, this enzymatic approach constructs two chiral centers in a single step under mild conditions, eliminating the need for harsh chemicals and improving overall yield and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amino Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-purity intermediates in the global veterinary pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the carbonyl reductase-mediated synthesis can be seamlessly transferred from the laboratory to industrial manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral amino alcohol meets the exacting standards required for API synthesis, providing our partners with a secure and compliant supply source.
We invite you to collaborate with us to leverage this advanced biocatalytic technology for your Florfenicol production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in enzymatic synthesis can drive efficiency and value for your organization.
