Revolutionizing Aryl Tri-n-butyl Tin Production: Scalable Palladium-Catalyzed Synthesis for Global Pharmaceutical Supply Chains
Patent CN108864173B introduces a groundbreaking methodology for converting substituted sodium aryl sulfinate to aryl tri-n-butyl tin compounds, representing a significant advancement in organotin chemistry with profound implications for pharmaceutical and agrochemical manufacturing sectors. This innovative process addresses longstanding limitations in traditional synthetic routes by utilizing readily available sodium aryl sulfinate as starting material instead of scarce aromatic halides, thereby establishing a more sustainable and economically viable pathway for producing these critical intermediates. The patent demonstrates exceptional functional group tolerance across diverse substituents including hydrogen, methyl, tert-butyl, halogens, cyano, trifluoromethyl, nitro, acetyl, and ethyl ester groups, enabling the synthesis of a wide range of aryl tri-n-butyl tin derivatives with remarkable consistency. This breakthrough methodology operates under milder reaction conditions with significantly reduced catalyst loading compared to conventional approaches, making it particularly suitable for large-scale commercial production while maintaining stringent quality standards required by global pharmaceutical manufacturers. The process eliminates the need for expensive organometallic reagents and harsh reaction environments that have historically constrained the widespread adoption of aryl tri-n-butyl tin compounds in industrial applications. This patent represents a strategic advancement that bridges the gap between academic research and industrial implementation for these valuable synthetic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for aryl tri-n-butyl tin compounds have been severely constrained by their reliance on aromatic halides as starting materials, which are often limited in availability and require complex multi-step preparations that significantly increase production costs and environmental impact. These conventional methods typically employ either stoichiometric organometallic reagents such as Grignard or organolithium compounds that necessitate strict anhydrous conditions and generate substantial waste streams, or precious metal-catalyzed reactions that demand expensive palladium catalysts in higher loadings than what is economically sustainable for large-scale manufacturing operations. The harsh reaction conditions required by these traditional routes—including cryogenic temperatures for organometallic preparations and extended reaction times under inert atmospheres—create significant operational challenges that compromise process safety and scalability while increasing energy consumption and production costs. Furthermore, the limited functional group tolerance of conventional methods restricts the diversity of aryl tri-n-butyl tin derivatives that can be practically synthesized, thereby limiting their application scope in pharmaceutical development where complex molecular architectures are increasingly required. These inherent limitations have historically prevented the widespread adoption of aryl tri-n-butyl tin compounds despite their demonstrated value in Stille coupling reactions and other critical synthetic transformations within the pharmaceutical industry.
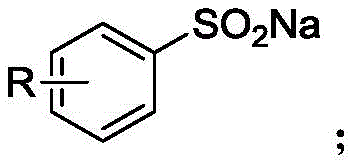
The Novel Approach
The patented methodology represents a paradigm shift by utilizing sodium aryl sulfinate as the primary starting material, which is significantly more accessible and cost-effective than aromatic halides while offering superior environmental compatibility through reduced hazardous waste generation. This innovative process employs a highly efficient palladium catalytic system featuring bis(tri-tert-butylphosphine) palladium at remarkably low loadings (3 mol%) that dramatically reduces catalyst costs while maintaining excellent reaction efficiency across diverse substrate classes. The reaction proceeds under relatively mild thermal conditions (80-140°C) with short reaction times (1-8 hours), eliminating the need for cryogenic equipment or specialized handling procedures required by traditional organometallic approaches. The exceptional functional group tolerance demonstrated by this method—accommodating hydrogen, methyl, tert-butyl, fluorine, chlorine, bromine, cyano, trifluoromethyl, nitro, acetyl, and ethyl ester substituents—enables the synthesis of previously inaccessible aryl tri-n-butyl tin derivatives with consistent high yields (38-97%) while maintaining exceptional purity profiles required for pharmaceutical applications. This novel approach establishes a robust foundation for scalable commercial production that addresses both economic and environmental concerns while expanding the synthetic utility of these valuable organotin compounds in pharmaceutical manufacturing.
Mechanistic Insights into Palladium-Catalyzed Conversion of Sodium Aryl Sulfinate to Aryl Tri-n-butyl Tin
The catalytic cycle begins with oxidative addition of the palladium(0) catalyst to the hexa-n-butylditin reagent, forming a palladium(II) species that subsequently coordinates with the sodium aryl sulfinate substrate through its sulfur atom. This coordination facilitates transmetalation where the aryl group transfers from sulfur to palladium, generating an aryl-palladium intermediate that undergoes reductive elimination with the tin moiety to form the final aryl tri-n-butyl tin product while regenerating the palladium(0) catalyst for subsequent cycles. The silver carbonate additive plays a crucial role in this mechanism by facilitating the oxidation state changes required for catalytic turnover while simultaneously acting as a base to neutralize any acidic byproducts formed during the reaction sequence. This elegant catalytic pathway operates with remarkable efficiency due to the steric and electronic properties of the tri-tert-butylphosphine ligand, which creates an optimal coordination environment around the palladium center that promotes both substrate activation and product release while minimizing unwanted side reactions or catalyst deactivation pathways.
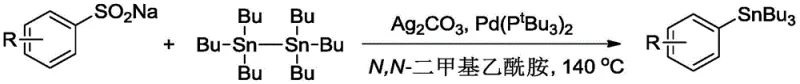
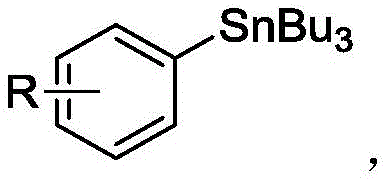
The exceptional purity profile achieved by this methodology stems from several key mechanistic features that minimize impurity formation throughout the reaction sequence. The mild reaction conditions prevent thermal decomposition pathways that commonly generate impurities in traditional high-temperature processes, while the selective nature of the palladium-catalyzed transmetalation step ensures minimal formation of homocoupling byproducts or other undesired side products. The use of N,N-dimethylacetamide as solvent provides an optimal polar aprotic environment that facilitates both reactant solubility and catalyst stability while preventing unwanted solvent participation in side reactions. The carefully optimized molar ratios (sodium aryl sulfinate: silver carbonate: hexa-n-butylditin = 1.5:1.5:(1-2)) create a balanced reaction environment that maximizes conversion while minimizing residual starting materials or intermediate species that could compromise final product purity. This mechanistic understanding enables precise control over impurity profiles through strategic manipulation of reaction parameters such as temperature ramping profiles, mixing efficiency, and concentration gradients during scale-up operations.
How to Synthesize Aryl Tri-n-butyle Tin Efficiently
This patented methodology represents a significant advancement in organotin chemistry that overcomes traditional limitations through innovative use of sodium aryl sulfinate as starting material instead of scarce aromatic halides. The process demonstrates exceptional functional group tolerance across diverse substituents while operating under milder conditions with reduced catalyst loading compared to conventional approaches. Detailed standardized synthesis steps are provided below to ensure consistent high-quality production outcomes across various manufacturing scales.
- Prepare the reaction mixture by combining sodium aryl sulfinate, silver carbonate, bis(tri-tert-butylphosphine) palladium catalyst, and hexa-n-butylditin in N,N-dimethylacetamide solvent with precise molar ratios of 1.5: 1.5:(1-2).
- Conduct the reaction at controlled temperatures between 80°C and 140°C for a duration of 1 to 8 hours, monitoring reaction progress through standard analytical techniques.
- After reaction completion, concentrate the mixture using atmospheric or vacuum distillation, followed by column chromatography purification with petroleum ether and/or ethyl acetate as mobile phase to obtain high-purity aryl tri-n-butyl tin product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in traditional organotin compound production that have historically constrained supply chain reliability and increased manufacturing costs for pharmaceutical intermediates. By replacing scarce aromatic halides with widely available sodium aryl sulfinate as starting material, this process establishes a more resilient supply chain foundation while simultaneously reducing raw material costs through utilization of more economical feedstocks with broader supplier networks.
- Cost Reduction in Manufacturing: The elimination of expensive organometallic reagents and reduction in precious metal catalyst loading significantly lowers raw material costs while simplifying process requirements that reduce operational expenses associated with specialized equipment and stringent environmental controls required by traditional methods. The use of readily available sodium aryl sulfinate feedstocks with multiple qualified suppliers creates competitive pricing dynamics that further enhance cost efficiency without compromising quality standards.
- Enhanced Supply Chain Reliability: The broad availability of sodium aryl sulfinate starting materials from multiple global suppliers creates a more resilient supply chain foundation compared to traditional routes dependent on limited aromatic halide sources. This diversification of raw material sources significantly reduces supply chain vulnerability while enabling more predictable lead times through established supplier relationships and inventory management practices that support just-in-time manufacturing requirements.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward purification process facilitate seamless scale-up from laboratory to commercial production volumes while generating significantly less hazardous waste compared to conventional methods that require cryogenic operations or produce toxic byproducts. This environmentally friendly profile aligns with increasingly stringent regulatory requirements while supporting corporate sustainability initiatives through reduced energy consumption and waste treatment costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding the implementation of this patented synthesis methodology for aryl tri-n-butyle tin production based on extensive process validation data and manufacturing experience.
Q: How does this novel synthesis method improve upon traditional approaches for aryl tri-n-butyl tin production?
A: This method replaces expensive and limited aromatic halides with widely available sodium aryl sulfinate as starting material, eliminating the need for harsh reaction conditions and expensive metal reagents required in conventional methods. The process demonstrates excellent functional group tolerance and operates under milder conditions with lower catalyst loading.
Q: What are the key advantages of using sodium aryl sulfinate instead of aromatic halides in this synthesis?
A: Sodium aryl sulfinate is more readily available, cost-effective, and environmentally friendly compared to aromatic halides. This substitution eliminates the need for stoichiometric organometallic reagents or expensive precious metal catalysts typically required in traditional approaches, resulting in significant process simplification and cost reduction.
Q: How does this process ensure high purity and scalability for commercial production?
A: The reaction demonstrates excellent functional group tolerance across diverse substituents (hydrogen, methyl, tert-butyl, halogens, cyano, trifluoromethyl, nitro, acetyl, ethyl ester), enabling production of various aryl tri-n-butyl tin derivatives with consistent high purity. The mild reaction conditions and straightforward purification process facilitate seamless scale-up from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Tri-n-butyle Tin Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex organotin intermediates while maintaining stringent purity specifications required by global pharmaceutical manufacturers. With rigorous QC labs equipped to validate every batch against exacting quality standards and a proven track record in delivering high-purity aryl tri-n-butyle tin compounds for critical pharmaceutical applications, we provide the technical expertise and manufacturing capability needed to support your most demanding projects from development through commercial production phases.
Request our Customized Cost-Saving Analysis today to understand how our patented synthesis methodology can optimize your supply chain economics while ensuring consistent quality delivery. Contact our technical procurement team to receive specific COA data and route feasibility assessments tailored to your unique manufacturing requirements.
