Advanced Palladium-Catalyzed Synthesis of Aryl Tri-n-butyl Tin for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of Aryl Tri-n-butyl Tin for Commercial Scale-up
The landscape of organic synthesis for high-value intermediates is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN108864173B, which discloses a robust process for converting substituted sodium aryl sulfinate into aryl tri-n-butyl tin compounds. This innovation represents a paradigm shift from traditional halide-based routes, offering a reliable pharmaceutical intermediate supplier with a distinct competitive edge. By leveraging a palladium-catalyzed system utilizing silver carbonate as an oxidant, this method achieves high yields under relatively mild conditions, addressing critical pain points in the manufacturing of Stille coupling partners. The technology is particularly relevant for the production of complex APIs and electronic materials where purity and functional group tolerance are paramount.
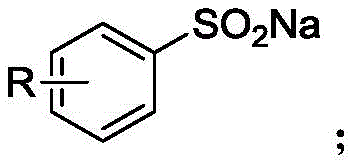
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl tri-n-butyl tin compounds has relied heavily on aromatic halides as the primary starting materials. Traditional pathways often involve the generation of highly reactive organometallic species, such as aryl Grignard reagents or aryllithium reagents, which subsequently react with tri-n-butyltin chloride. These conventional methods suffer from severe drawbacks, including the requirement for strictly anhydrous and oxygen-free conditions, which complicates operational safety and increases infrastructure costs. Furthermore, the reliance on aromatic iodides or bromides introduces significant cost volatility, as these halides are often more expensive and less atom-economical than their sulfinate counterparts. The use of stoichiometric amounts of metal reagents also generates substantial quantities of inorganic waste, posing environmental challenges that modern green chemistry initiatives strive to eliminate. Additionally, the harsh conditions required for Grignard formation limit the scope of substrates, often leading to incompatibility with sensitive functional groups present in advanced drug candidates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes sodium aryl sulfinate as the electrophilic partner, reacting it with hexa-n-butylditin in the presence of a palladium catalyst and silver carbonate. This methodology circumvents the need for pre-formed organometallic reagents, thereby drastically simplifying the operational procedure and enhancing safety profiles. The use of sodium aryl sulfinate is a major supplement to existing synthetic toolkits, as these salts are widely available, inexpensive, and stable under ambient conditions, facilitating easier storage and handling. The reaction demonstrates exceptional universality, accommodating a broad spectrum of substituents on the aromatic ring, including electron-withdrawing groups like nitro and cyano, as well as electron-donating groups like methyl and tert-butyl. This versatility ensures that the process can be adapted for the cost reduction in pharmaceutical intermediate manufacturing without necessitating extensive re-optimization for each new substrate. The transition from halides to sulfinates thus represents not just a chemical modification, but a strategic supply chain improvement.
Mechanistic Insights into Pd-Catalyzed Desulfitative Stannylation
The core of this technological advancement lies in the intricate catalytic cycle mediated by bis(tri-tert-butylphosphine)palladium. Unlike traditional cross-coupling mechanisms that rely on oxidative addition into a carbon-halogen bond, this process involves the activation of the carbon-sulfur bond in the sulfinate salt. The palladium catalyst, likely in a zero-valent state initially, interacts with the sulfinate substrate, potentially facilitated by the silver carbonate oxidant which assists in the removal of the sulfonyl group as sulfur dioxide. This desulfitative process generates an aryl-palladium species that is poised for transmetallation. The hexa-n-butylditin serves as the tin source, transferring a tri-n-butyltin group to the palladium center. The bulky tri-tert-butylphosphine ligands play a crucial role in stabilizing the active palladium species and preventing catalyst deactivation through aggregation. This ligand architecture ensures that the catalytic turnover number remains high even at low catalyst loadings, typically around 3 mol% relative to the tin reagent. The final step involves reductive elimination to release the desired aryl tri-n-butyl tin product and regenerate the active catalyst, completing the cycle efficiently.
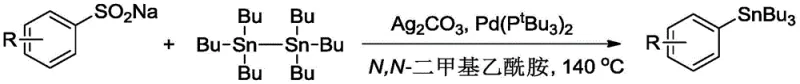
Impurity control is another critical aspect where this mechanism excels. In conventional Grignard routes, side reactions such as homocoupling of the aryl group or protonation of the organometallic intermediate can lead to difficult-to-remove impurities. The palladium-catalyzed desulfitative stannylation minimizes these pathways due to the specific nature of the transmetallation step. The use of silver carbonate as a stoichiometric oxidant helps drive the reaction to completion while scavenging potential sulfur-containing byproducts. Furthermore, the reaction conditions, typically ranging from 80°C to 140°C in N,N-dimethylacetamide, are optimized to balance reaction rate with selectivity. The resulting crude products are often of high purity, requiring only standard column chromatography for final isolation. This high level of chemoselectivity is vital for R&D directors who need to ensure that the impurity profile of the intermediate does not complicate downstream purification steps in the final API synthesis.
How to Synthesize Aryl Tri-n-butyl Tin Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction parameters to maximize yield and reproducibility. The patent outlines a straightforward protocol where sodium aryl sulfinate, silver carbonate, the palladium catalyst, and hexa-n-butylditin are combined in a solvent system. The choice of N,N-dimethylacetamide as the solvent is critical for solubilizing the inorganic salts and maintaining the stability of the catalytic species. Reaction times can vary from 1 to 8 hours depending on the electronic nature of the substrate, with electron-deficient substrates often reacting faster. For detailed operational procedures, safety data, and specific workup protocols, please refer to the standardized synthesis guide below.
- Mix sodium aryl sulfinate, silver carbonate, bis(tri-tert-butylphosphine)palladium, and hexa-n-butylditin in N,N-dimethylacetamide solvent.
- Heat the reaction mixture to a temperature range of 80-140°C and maintain stirring for 1 to 8 hours to ensure complete conversion.
- Concentrate the reaction mixture to remove solvent and purify the crude product via column chromatography using petroleum ether or ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sulfinate-based route offers tangible benefits that extend beyond mere chemical curiosity. The shift away from aromatic halides mitigates the risk associated with the price volatility of iodine and bromine commodities. Sodium aryl sulfinates are generally derived from abundant sulfur sources and are produced on a massive scale for other industries, ensuring a stable and continuous supply chain. This stability is crucial for long-term production planning and contract manufacturing agreements. Moreover, the elimination of pyrophoric reagents like Grignard reagents reduces the need for specialized containment equipment and lowers insurance and compliance costs related to hazardous material handling. The simplified workflow also translates to reduced labor hours and faster batch turnover times, enhancing overall plant throughput.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling. By replacing expensive aromatic iodides or bromides with cheaper sodium aryl sulfinates, the raw material cost per kilogram of product is significantly lowered. Additionally, the low catalyst loading of the palladium complex means that precious metal consumption is minimized, further driving down the cost of goods sold. The process avoids the generation of large volumes of magnesium or lithium salts, simplifying waste treatment and reducing disposal fees. These cumulative savings allow for a more competitive pricing structure in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of the key starting materials. Sodium aryl sulfinates are stable solids that do not require the stringent storage conditions necessary for liquid halides or organometallic reagents. This stability reduces the risk of spoilage during transit and storage, ensuring that inventory remains viable for longer periods. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in utility supply, such as cooling water temperature fluctuations, making it ideal for multi-site manufacturing strategies.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with green chemistry principles. The atom economy is improved by avoiding the formation of stoichiometric metal halide waste. The reaction produces sulfur dioxide as a byproduct, which can be effectively managed with standard scrubbing systems already present in most chemical facilities. The scalability of the process has been demonstrated across a wide range of substrates, indicating that translation from laboratory scale to commercial production (100 kgs to 100 MT) can be achieved with minimal technical risk. This ease of scale-up ensures that supply commitments can be met reliably as demand grows.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using sodium aryl sulfinate over aromatic halides?
A: Sodium aryl sulfinate serves as a more cost-effective and widely available raw material compared to aromatic iodides or bromides. It eliminates the need for harsh Grignard or organolithium reagents, improving operational safety and reducing production costs significantly.
Q: What is the functional group tolerance of this stannylation method?
A: The method exhibits excellent universality and tolerance towards various functional groups including methyl, tert-butyl, fluoro, chloro, bromo, cyano, trifluoromethyl, nitro, acetyl, and ethyl ester groups, making it suitable for complex molecule synthesis.
Q: What are the typical reaction conditions and catalyst loading?
A: The reaction typically proceeds in N,N-dimethylacetamide at temperatures between 80°C and 140°C. The catalyst loading is low, with a molar ratio of catalyst to hexa-n-butylditin around 3:100, ensuring economic feasibility for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Tri-n-butyl Tin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed desulfitative stannylation technology for the global pharmaceutical and electronic materials sectors. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade intermediates. We understand that consistency and quality are non-negotiable in the life sciences industry, and our commitment to excellence drives every batch we produce.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs.
