Advanced Synthesis of Molnupiravir Intermediates: A Breakthrough in Purity and Scalability
The global pharmaceutical landscape has been profoundly impacted by the emergence of potent antiviral agents, with Molnupiravir standing out as a critical small-molecule nucleoside analogue developed for the treatment of SARS-CoV-2 infections. As detailed in the recent patent literature, specifically CN115572317A, the demand for high-quality intermediates in the synthesis of this life-saving medication has necessitated a re-evaluation of traditional manufacturing protocols. This patent introduces a refined preparation method for a key Molnupiravir intermediate, addressing long-standing challenges related to chemoselectivity and impurity profiles that have plagued earlier synthetic routes. By shifting the paradigm from harsh basic conditions to a more nuanced system utilizing carbonate acid-binding agents and non-polar organic solvents, the invention offers a pathway to significantly enhanced product purity and yield. For industry stakeholders, this represents not merely a chemical optimization but a strategic opportunity to secure a more robust and reliable supply chain for essential antiviral therapeutics.
The significance of this technological advancement cannot be overstated when considering the clinical efficacy and safety requirements of modern antiviral drugs. Molnupiravir functions as a polymerase inhibitor, and its therapeutic success relies heavily on the structural integrity and purity of its precursor molecules. Any deviation in the synthesis of the intermediate can lead to the presence of genotoxic or pharmacologically active impurities that complicate regulatory approval and patient safety. The methodology described in CN115572317A provides a rigorous framework for controlling these variables, ensuring that the resulting intermediate possesses the requisite quality for subsequent oximation and deprotection steps. This level of control is paramount for manufacturers aiming to meet the stringent Good Manufacturing Practice (GMP) standards required by global health authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key 5'-ester intermediate involved the use of strong organic bases such as DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) in polar aprotic solvents like acetonitrile. While effective in driving the esterification reaction, this conventional approach suffers from significant drawbacks that hinder large-scale production. The primary issue lies in the lack of chemoselectivity; the cytidine substrate contains multiple nucleophilic sites, including the exocyclic amino group and various hydroxyl groups. Under the influence of strong bases like DBU, uncontrolled acylation occurs, leading to the formation of undesirable byproducts such as diacylated impurities and N-acylated impurities. Furthermore, DBU is notoriously difficult to remove completely from the reaction mixture, often requiring extensive and costly purification procedures such as silica gel column chromatography, which is impractical for multi-kilogram or ton-scale manufacturing.
Additionally, prior art processes, including those disclosed in Indian patent applications, have demonstrated suboptimal purity profiles, with target intermediate purity often hovering around 83% and significant levels of impurities persisting even after workup. These impurities not only reduce the overall yield of the final active pharmaceutical ingredient (API) but also impose a heavy burden on downstream processing teams who must dedicate resources to removing these contaminants. The reliance on polar solvents and difficult-to-remove bases creates a bottleneck in the production workflow, increasing both the time-to-market and the overall cost of goods sold (COGS). For procurement and supply chain managers, these inefficiencies translate into volatility in supply and unpredictable production timelines.
The Novel Approach
In stark contrast to the limitations of the prior art, the novel approach outlined in CN115572317A leverages a synergistic combination of mild inorganic bases and non-polar reaction media to achieve superior selectivity. By employing alkali metal carbonates or bicarbonates—such as potassium carbonate or sodium carbonate—as the acid-binding agent, the reaction environment is buffered in a way that favors O-acylation over N-acylation. This subtle shift in basicity is complemented by the use of non-polar organic solvents like toluene or tetrahydrofuran, which alter the solvation shell around the nucleophilic sites, further discouraging side reactions. The result is a dramatic suppression of the formation of Impurity A (diacylation) and Impurity B (N-acylation), yielding a crude product of exceptional purity that often requires minimal purification.
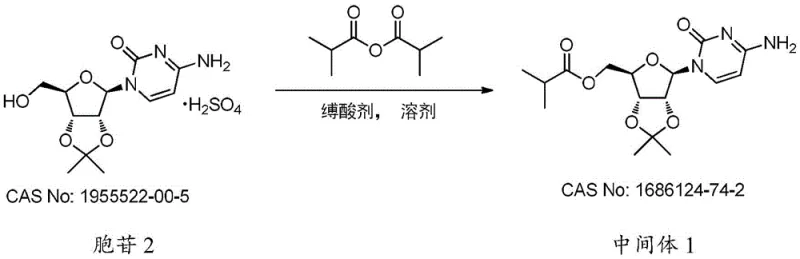
The implementation of this novel route transforms the manufacturing landscape for this critical intermediate. Instead of grappling with stubborn impurities and complex chromatographic separations, manufacturers can utilize simple aqueous workups and crystallization techniques to isolate the product. The process operates under mild temperatures, typically between 15°C and 30°C, which reduces energy consumption and enhances operational safety. This streamlined approach not only boosts the yield to over 90% in optimized examples but also ensures a consistent quality profile that is essential for regulatory compliance. For a reliable pharmaceutical intermediate supplier, adopting this methodology means delivering a product that facilitates smoother downstream synthesis for their clients, ultimately accelerating the availability of the final antiviral medication.
Mechanistic Insights into Chemoselective Esterification
To fully appreciate the technical merit of this invention, one must delve into the mechanistic nuances that govern the selectivity of the acylation reaction. The cytidine derivative possesses a primary hydroxyl group at the 5'-position, which is the desired site for acylation, alongside a secondary hydroxyl and an exocyclic amine. In traditional polar protic or aprotic environments with strong bases, the nucleophilicity of the amine is often enhanced, or the solvation effects fail to differentiate sufficiently between the oxygen and nitrogen nucleophiles. However, in the non-polar medium prescribed by the new patent, the solubility and reactivity of the ionic species are modulated. The carbonate base acts as a heterogeneous or semi-heterogeneous scavenger of the generated acid, maintaining a localized pH that is sufficient to activate the alcohol but insufficient to promote significant attack by the amine nitrogen.
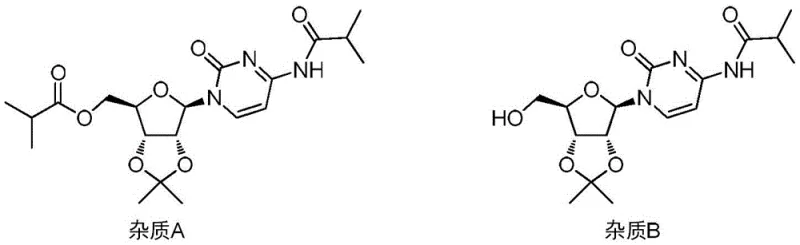
Furthermore, the steric and electronic environment created by the DMAP catalyst in conjunction with the carbonate base favors the formation of the reactive acyl-pyridinium intermediate, which then selectively transfers the acyl group to the less hindered and more accessible 5'-hydroxyl group. The suppression of Impurity A and Impurity B is not merely a result of thermodynamic control but is kinetically driven by the specific solvent-base matrix. By minimizing the presence of free, highly reactive anionic species that could indiscriminately attack the electrophile, the process ensures that the reaction trajectory remains focused on the desired mono-esterification. This mechanistic precision is what allows the process to achieve such low impurity levels without the need for excessive reagent equivalents or harsh conditions, representing a triumph of green chemistry principles applied to complex nucleoside synthesis.
How to Synthesize Molnupiravir Intermediate Efficiently
The practical execution of this synthesis is designed to be straightforward yet precise, ensuring reproducibility across different manufacturing scales. The process begins with the careful preparation of the reaction mixture, where the stoichiometry of the acid-binding agent is critical to neutralizing the byproduct acid without creating an overly basic environment. The controlled addition of isobutyric anhydride at low temperatures prevents exothermic spikes that could degrade the sensitive nucleoside structure. Following the reaction period, the workup procedure is remarkably simple, involving phase separation and washing, which avoids the generation of large volumes of hazardous waste associated with chromatographic purification. For detailed operational parameters and specific molar ratios, please refer to the standardized guide below.
- Prepare a mixed solution of Cytidine 2, DMAP catalyst, and a carbonate/bicarbonate acid-binding agent in a non-polar solvent like toluene.
- Cool the mixture to 5-10°C and slowly add isobutyric anhydride over a period of 1 to 3 hours while maintaining strict temperature control.
- Allow the reaction to proceed at 20-25°C for 8 to 16 hours, followed by aqueous workup and concentration to isolate the high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers compelling advantages that resonate deeply with procurement managers and supply chain directors. The primary value proposition lies in the drastic simplification of the purification process. By eliminating the need for silica gel column chromatography—a technique that is resource-intensive, slow, and difficult to scale—the new method significantly reduces the operational expenditure associated with manufacturing. The removal of expensive and hard-to-source reagents like DBU in favor of commodity chemicals like potassium carbonate further drives down the raw material costs. This shift not only lowers the direct cost of production but also mitigates the risk of supply disruptions associated with specialty reagents, ensuring a more stable and predictable manufacturing timeline.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven by the elimination of complex purification steps and the use of inexpensive, widely available reagents. By avoiding the need for large quantities of silica gel and organic solvents required for column chromatography, the facility can realize substantial savings in waste disposal and material costs. Additionally, the higher yield achieved through improved selectivity means that less starting material is wasted, directly improving the atom economy and reducing the cost per kilogram of the final intermediate. These efficiencies compound over large production runs, leading to a significantly more competitive pricing structure for the finished API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on generic, off-the-shelf chemicals rather than specialized catalysts that may have long lead times. Potassium carbonate, sodium carbonate, and toluene are produced globally in massive quantities, insulating the manufacturing process from the volatility of the fine chemical market. Furthermore, the robustness of the reaction conditions allows for greater flexibility in production scheduling, as the process is less sensitive to minor variations in temperature or reagent quality. This reliability ensures that downstream customers receive their shipments on time, maintaining the continuity of their own drug production schedules.
- Scalability and Environmental Compliance: The transition to a non-polar solvent system and inorganic bases aligns perfectly with modern environmental, health, and safety (EHS) standards. Non-polar solvents like toluene are easier to recover and recycle compared to polar aprotic solvents, reducing the environmental footprint of the operation. The absence of difficult-to-remove organic bases simplifies wastewater treatment, lowering the burden on effluent processing facilities. From a scalability standpoint, the exotherm control and simple workup make the process ideal for transfer from pilot plant to multi-ton commercial reactors, facilitating rapid capacity expansion to meet surging global demand for antiviral treatments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear picture of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: Why is the new carbonate-based method superior to traditional DBU methods?
A: Traditional methods using DBU often result in difficult-to-remove residues and higher levels of diacylation impurities. The new method utilizes mild carbonate bases in non-polar solvents, which significantly suppresses side reactions and simplifies downstream purification.
Q: What are the critical impurities controlled in this synthesis?
A: The process specifically targets the reduction of Impurity A (a diacylated byproduct) and Impurity B (an N-acylated byproduct), ensuring the final intermediate meets stringent quality specifications for antiviral drug production.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the elimination of complex column chromatography and the use of common, inexpensive reagents like potassium carbonate and toluene make this route highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Molnupiravir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving antiviral drugs depends on the availability of high-quality, consistently supplied intermediates. Our technical team has thoroughly analyzed the advancements presented in CN115572317A and is fully equipped to implement this superior synthesis route at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are designed to handle complex nucleoside chemistry with stringent purity specifications, supported by rigorous QC labs that verify every batch against the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your Molnupiravir supply chain. By partnering with our expert team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your production strategy and secure a competitive advantage in the rapidly evolving antiviral market.
