Scaling High-Purity BVPE Production: A Novel Low-Cost Industrial Route
Introduction to Patent CN113372187A
The chemical industry is constantly seeking more efficient pathways to produce high-value olefin monomers, and the recent disclosure in patent CN113372187A represents a significant leap forward in the industrial synthesis of BVPE (P,P'-divinyl-1,2-diphenylethane). This versatile monomer is a critical building block for advanced polyphenylene ether resins and high-performance chromatographic columns, yet its widespread adoption has historically been hindered by costly and hazardous production methods. The inventors have successfully developed a robust three-step synthetic route that starts from the inexpensive and readily available 1,2-diphenylethane, effectively bypassing the need for expensive precursors like p-chlorostyrene. By optimizing reaction conditions and introducing a novel dual-inhibitor system during the final dehydration step, this methodology achieves a comprehensive yield of up to 76.5%, which is a substantial improvement over the 44.4% yield reported in prior art literature. For R&D directors and procurement specialists alike, this patent offers a compelling blueprint for reducing manufacturing costs while ensuring the supply continuity of this essential specialty chemical.
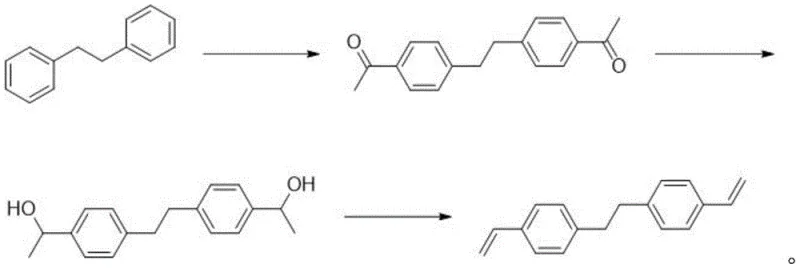
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of BVPE and similar divinyl compounds has been plagued by severe technical and economic bottlenecks that limit their application in large-scale polymer manufacturing. One common approach involves the use of p-chlorostyrene as a starting material, which is not only prohibitively expensive but also introduces halogenated impurities that are difficult to remove, thereby compromising the quality of the final polymer. Another widely cited method, described in the Russian Journal of Applied Chemistry, relies on a Friedel-Crafts acylation followed by high-pressure hydrogenation using Raney nickel. This specific step requires operating pressures as high as 7.7 MPa, demanding specialized and costly autoclave equipment that poses significant safety risks in a plant environment. Furthermore, the purification processes in these legacy routes are inefficient, often requiring massive volumes of solvent—up to 27 times the mass of the substrate—to achieve acceptable purity levels. These factors combine to create a high barrier to entry, resulting in elevated production costs and unreliable supply chains for downstream users who depend on consistent monomer quality.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the method disclosed in CN113372187A utilizes a streamlined strategy that prioritizes mild reaction conditions and operational simplicity. The new route replaces the dangerous high-pressure hydrogenation step with a gentle reduction using sodium borohydride, which can be performed at ambient pressure and moderate temperatures, significantly lowering the capital expenditure required for reactor infrastructure. Additionally, the process employs a highly effective purification protocol using toluene or dichloroethane, which drastically reduces solvent consumption compared to the ethanol-heavy washes of the past. Perhaps most critically, the inventors have solved the persistent issue of product polymerization during the final acid-catalyzed dehydration by employing a synergistic mixture of p-tert-butylcatechol (TBC) and phenothiazine. This innovation prevents the formation of dimers and oligomers, ensuring that the crude product yield remains exceptionally high at 97%, thus minimizing waste and maximizing the throughput of the reliable BVPE supplier networks adopting this technology.
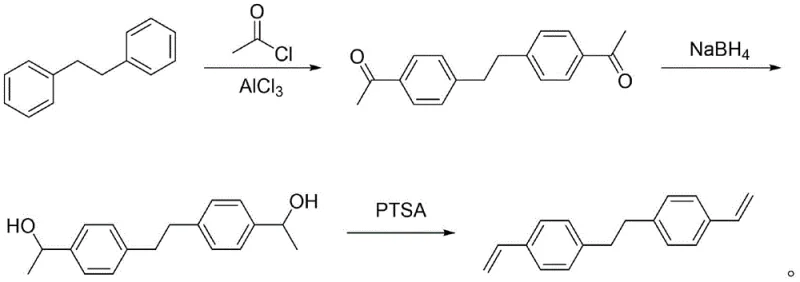
Mechanistic Insights into the Three-Step Synthetic Sequence
The core of this technological breakthrough lies in the precise control of reaction kinetics and thermodynamics across the three distinct transformation stages. The process initiates with a classic Friedel-Crafts acylation, where 1,2-diphenylethane reacts with acetyl chloride in the presence of aluminum trichloride. This electrophilic aromatic substitution is carefully managed at temperatures between 0°C and 40°C to ensure regioselectivity, predominantly yielding the desired 4,4'-diacetyl diaryl ethane isomer while minimizing ortho-substitution byproducts. Following acylation, the diketone intermediate undergoes reduction using sodium borohydride in a protic solvent system such as ethanol or methanol. Unlike catalytic hydrogenation which can be non-selective and require harsh conditions, this hydride reduction proceeds smoothly to form the corresponding bis(alpha-methyl benzyl alcohol) diol with near-quantitative conversion, setting the stage for the final elimination reaction.
The final and most mechanistically complex step is the acid-catalyzed dehydration of the diol to form the vinyl groups of BVPE. This step is inherently risky because the resulting styrenic double bonds are highly reactive and prone to cationic polymerization in the presence of the strong acid catalyst, p-toluenesulfonic acid (PTSA). The patent elucidates a critical mechanism where the addition of phenothiazine acts as a radical scavenger that complements the inhibitory action of TBC. While TBC traps growing polymer chains, phenothiazine stabilizes the reaction medium against oxidative initiation, effectively suppressing the formation of dimers which typically account for over 10% of the product mass in single-inhibitor systems. By maintaining the dimer content at approximately 2%, this dual-inhibitor strategy ensures that the high-purity BVPE obtained after simple silica gel column chromatography and recrystallization meets the stringent specifications required for electronic and optical applications, demonstrating a profound understanding of impurity control mechanisms.
How to Synthesize BVPE Efficiently
Implementing this synthesis route in a pilot or production facility requires strict adherence to the optimized parameters regarding stoichiometry, temperature control, and solvent ratios to maximize the economic benefits. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors without the need for exotic equipment, making it an ideal candidate for commercial scale-up of complex olefin monomers. Operators must pay particular attention to the dehydration step, ensuring that water is continuously removed via azeotropic distillation to drive the equilibrium toward the alkene product while maintaining the inhibitor concentration within the narrow optimal window defined in the patent examples.
- Perform Friedel-Crafts acylation of 1,2-diphenylethane with acetyl chloride and AlCl3 in dichloromethane to obtain 4,4'-diacetyl diaryl ethane.
- Reduce the diketone intermediate using sodium borohydride in ethanol or methanol to form the bis(alpha-methyl benzyl alcohol) diol.
- Dehydrate the diol using p-toluenesulfonic acid in toluene with a dual inhibitor system of TBC and phenothiazine to yield high-purity BVPE.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates directly into tangible operational efficiencies and risk mitigation strategies that strengthen the overall value proposition of the supply base. The shift away from high-pressure hydrogenation equipment not only reduces the initial capital investment but also lowers the ongoing maintenance and safety compliance costs associated with handling compressed hydrogen gas, leading to a more resilient production infrastructure. Furthermore, the use of 1,2-diphenylethane as a starting material leverages a commodity chemical with a stable global supply, insulating the manufacturing process from the volatility and scarcity issues often associated with specialized halogenated precursors like p-chlorostyrene.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and high-pressure reactors significantly lowers the fixed and variable costs of production. By achieving a comprehensive yield that is nearly double that of previous methods, the process minimizes raw material waste and energy consumption per kilogram of output, driving down the unit cost of the final monomer substantially.
- Enhanced Supply Chain Reliability: The mild reaction conditions allow for faster batch turnover times and reduce the likelihood of unplanned shutdowns due to equipment failure or safety incidents. The robustness of the dual-inhibitor system ensures consistent product quality across different batches, reducing the need for extensive rework or rejection of off-spec material, thereby guaranteeing a steady flow of goods to customers.
- Scalability and Environmental Compliance: The simplified workup procedures, which rely on standard solvent extraction and recrystallization rather than complex distillation or sublimation, facilitate easier scaling from pilot plants to multi-ton production lines. Additionally, the reduction in solvent usage and the avoidance of heavy metal catalysts align with increasingly strict environmental regulations, simplifying waste disposal and enhancing the sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity for stakeholders evaluating its potential for integration into their existing supply chains. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the practical advantages of the new methodology over traditional approaches.
Q: Why is the dual inhibitor system critical in BVPE synthesis?
A: Styrene derivatives are highly prone to polymerization under acidic dehydration conditions. The patent reveals that using only TBC results in significant dimer formation (11%). Adding phenothiazine alongside TBC suppresses this side reaction, reducing dimer content to ~2% and boosting yield from 65% to 85%.
Q: How does this route improve upon previous high-pressure methods?
A: Conventional routes often require Raney nickel hydrogenation at extreme pressures (7.7 MPa), necessitating specialized autoclaves. This novel method utilizes mild sodium borohydride reduction at ambient pressure, drastically lowering equipment costs and safety risks while maintaining high conversion rates.
Q: What is the expected purity of BVPE produced via this method?
A: The optimized process achieves a crude product yield of 97% with a GC content of 99.6% after simple column purification and recrystallization. This high purity is essential for downstream polymerization applications where impurities can act as chain terminators.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BVPE Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in CN113372187A and are uniquely positioned to leverage this technology for our global clientele. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the low dimer content and high GC purity that define the success of this specific BVPE route, giving our partners confidence in every shipment.
We invite you to collaborate with us to unlock the full economic potential of this advanced manufacturing process. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, allowing you to quantify the benefits of switching to this superior supply source. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your projects benefit from the highest quality intermediates available in the market.
