Scalable Industrial Synthesis of BVPE via Novel Friedel-Crafts Pathway
Introduction to Advanced BVPE Manufacturing Technology
The landscape of olefin monomer synthesis is undergoing a significant transformation driven by the need for cost-effective and scalable processes. Patent CN113372187A introduces a groundbreaking industrial synthesis method for BVPE (P,P'-divinyl-1,2-diphenylethane), a critical monomer used in high-performance polypropylene films, polyphenylene ether resins, and HPLC chromatographic columns. This technology addresses the longstanding economic and technical barriers associated with traditional production routes, which often rely on expensive precursors like p-chlorostyrene or require hazardous high-pressure hydrogenation. By leveraging a robust three-step sequence starting from the readily available 1,2-diphenylethane, this innovation delivers a comprehensive yield of up to 76.5% and product purity exceeding 99.6%. For global procurement leaders and R&D directors, this represents a pivotal shift towards more sustainable and economically viable supply chains for specialty polymer intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of BVPE has been plagued by severe technical inefficiencies and prohibitive costs that hinder large-scale adoption. The conventional route utilizing p-chlorostyrene as a starting material suffers from exorbitant raw material costs, rendering the final product economically uncompetitive for bulk polymer applications. Furthermore, alternative literature methods, such as those reported in the Russian Journal of Applied Chemistry, rely on a multi-step pathway involving Raney nickel catalyzed hydrogenation. This approach necessitates specialized high-pressure equipment capable of withstanding 7.7 MPa, introducing significant safety hazards and capital expenditure burdens. Additionally, these legacy processes often exhibit poor atom economy and low overall yields, typically hovering around 44.4%, due to uncontrolled side reactions and difficult purification steps that require excessive solvent volumes.
The Novel Approach
In stark contrast, the novel industrial synthesis method outlined in the patent data offers a streamlined, three-step pathway that eliminates the need for high-pressure infrastructure and expensive halogenated starting materials. By initiating the synthesis with 1,2-diphenylethane, the process leverages a cost-effective feedstock that is widely available in the fine chemical market. The route proceeds through a highly selective Friedel-Crafts acylation followed by a mild reduction and a carefully controlled dehydration. This strategic redesign not only simplifies the operational workflow but also drastically improves the mass balance of the reaction. The implementation of this route allows manufacturers to bypass the complex safety protocols associated with high-pressure hydrogenation, thereby reducing the barrier to entry for commercial scale-up.
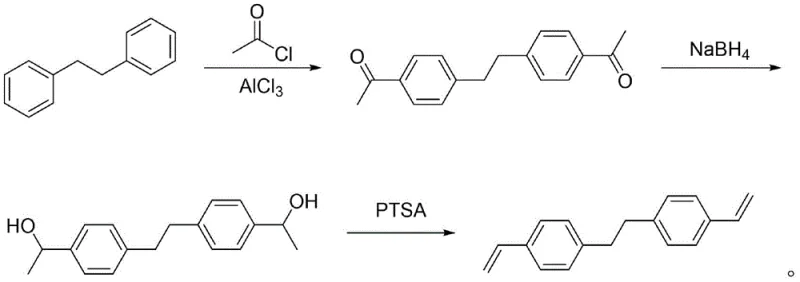
Mechanistic Insights into the Three-Step Synthetic Sequence
The core of this technological advancement lies in the precise control of reaction selectivity across three distinct chemical transformations. The first step involves a Friedel-Crafts acylation where 1,2-diphenylethane reacts with acetyl chloride in the presence of aluminum trichloride. This electrophilic aromatic substitution is highly regioselective, favoring the para-position to yield 4,4'-diacetyl diaryl ethane with minimal ortho-substitution byproducts. The subsequent reduction step utilizes sodium borohydride, a mild and safe reducing agent, to convert the ketone functionalities into secondary alcohols. Unlike catalytic hydrogenation, this hydride reduction operates at atmospheric pressure and ambient to reflux temperatures, ensuring that the sensitive ethane bridge remains intact while achieving near-quantitative conversion to the bis(alpha-methyl benzyl alcohol) intermediate.
The final and most critical stage is the acid-catalyzed dehydration of the diol intermediate to form the divinyl product. This step is inherently prone to cationic polymerization and dimerization due to the high reactivity of the styrenic double bonds. To counteract this, the patent discloses a sophisticated dual-inhibitor system comprising p-tert-butylcatechol (TBC) and phenothiazine. While TBC acts as a radical scavenger, phenothiazine provides additional stabilization against thermal polymerization during the reflux conditions required for water removal. This synergistic effect is crucial; comparative data indicates that using TBC alone results in significant dimer formation (up to 11%), whereas the dual system keeps dimer content as low as 2%. This mechanistic refinement ensures that the crude reaction mixture maintains a high GC content, simplifying downstream purification and maximizing the recovery of the target olefin monomer.
How to Synthesize BVPE Efficiently
Implementing this synthesis route requires strict adherence to the optimized molar ratios and temperature profiles defined in the patent to ensure maximum yield and purity. The process begins with the preparation of the diacetyl intermediate in a chlorinated solvent, followed by an aqueous workup and recrystallization to secure high purity before reduction. The reduction step is notably forgiving, allowing for the use of wet alcohols as solvents, which further reduces material costs. Finally, the dehydration must be conducted under azeotropic distillation conditions to drive the equilibrium forward while maintaining the inhibitor concentration within the optimal window.
- Perform Friedel-Crafts acylation of 1,2-diphenylethane using aluminum trichloride and acetyl chloride to obtain 4,4'-diacetyl diaryl ethane.
- Reduce the diacetyl intermediate using sodium borohydride in an alcohol solvent to form the bis(alpha-methyl benzyl alcohol) derivative.
- Execute acid-catalyzed dehydration using p-toluenesulfonic acid with a dual inhibitor system of TBC and phenothiazine to yield high-purity BVPE.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel BVPE synthesis route offers substantial strategic benefits beyond mere technical feasibility. The elimination of high-pressure hydrogenation equipment translates directly into reduced capital expenditure (CAPEX) and lower operational complexity, making the process accessible to a wider range of manufacturing facilities. Furthermore, the reliance on commodity chemicals like 1,2-diphenylethane and acetyl chloride insulates the supply chain from the volatility associated with specialized halogenated precursors. The robust nature of the reaction conditions also implies a lower risk of batch failures, ensuring consistent delivery schedules for downstream polymer manufacturers who rely on just-in-time inventory models.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the substitution of expensive raw materials with cost-effective alternatives and the removal of energy-intensive high-pressure steps. By avoiding the need for p-chlorostyrene and specialized hydrogenation reactors, the variable cost of goods sold is significantly decreased. Additionally, the high selectivity of the dual-inhibitor system reduces the load on purification units, leading to lower solvent consumption and waste disposal costs. These factors combine to create a leaner manufacturing profile that enhances margin potential without compromising product quality.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key starting material, 1,2-diphenylethane, is a standard industrial chemical with a mature global supply network. This contrasts sharply with the niche supply chains required for p-chlorostyrene or high-pressure catalysts. The simplified process flow, consisting of only three main reaction steps, also reduces the number of potential bottlenecks in production. Consequently, manufacturers can maintain higher inventory turnover rates and respond more agilely to fluctuations in market demand for BVPE-based polymers and electronic materials.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the replacement of high-pressure hydrogenation with atmospheric hydride reduction significantly lowers the safety footprint of the facility. The process generates less hazardous waste and avoids the use of heavy metal catalysts like Raney nickel, which require complex disposal protocols. The mild reaction conditions facilitate easier scale-up from pilot plants to multi-ton commercial production, allowing for rapid capacity expansion to meet growing demand in the electronics and automotive sectors while adhering to stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this BVPE synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations, purity profiles, and process safety. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new BVPE synthesis route compare to conventional methods in terms of equipment requirements?
A: Unlike conventional methods requiring high-pressure hydrogenation equipment (up to 7.7 MPa), this novel route operates under atmospheric pressure using mild reduction conditions, significantly lowering capital expenditure and safety risks.
Q: What mechanism prevents polymerization during the final dehydration step?
A: The process utilizes a synergistic dual-inhibitor system combining p-tert-butylcatechol (TBC) and phenothiazine. This combination effectively suppresses dimer formation and polymerization, achieving a crude GC content of up to 96% before purification.
Q: What is the achievable purity and yield for industrial scale BVPE production?
A: Through optimized recrystallization and column purification, the process achieves a final product purity of 99.6% (GC) with a comprehensive yield of approximately 76.5%, which is substantially higher than literature precedents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BVPE Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers like BVPE play in the advancement of next-generation polymers and electronic materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis route, including rigorous QC labs that enforce stringent purity specifications to guarantee a product free from detrimental dimers and oligomers. We are committed to delivering value through technical excellence and operational reliability.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for specialty chemical intermediates.
