Advanced Catalytic Synthesis of Hydroxyethyl Ethylenediamine for Commercial Scale-up
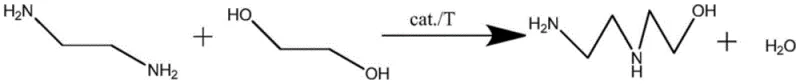
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and safe synthetic routes for critical intermediates like hydroxyethyl ethylenediamine. A significant technological breakthrough in this domain is detailed in patent CN111116386B, which discloses a novel method for synthesizing hydroxyethyl ethylenediamine through the intermolecular dehydration condensation of ethylenediamine and ethylene glycol. This innovation addresses long-standing safety and efficiency challenges by replacing hazardous reagents with safer, cost-effective alternatives while utilizing advanced Raney-type catalytic systems. For R&D directors and procurement strategists, this patent represents a pivotal shift towards greener chemistry that does not compromise on yield or purity. The process operates under controlled high-pressure hydrogen conditions, achieving exceptional conversion rates and simplifying downstream purification, thereby offering a compelling value proposition for large-scale manufacturing of veterinary drugs and pharmaceutical raw materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of hydroxyethyl ethylenediamine has relied on three primary routes, each fraught with significant operational and economic drawbacks that hinder optimal supply chain efficiency. The ethanolamine process, while capable of high selectivity, suffers from notoriously low conversion rates of approximately 30%, leading to massive recycling loops and energy inefficiencies that drive up operational costs. Alternatively, the chlorohydrin method generates corrosive hydrogen chloride gas as a byproduct, imposing severe environmental compliance burdens and requiring expensive corrosion-resistant equipment and waste treatment protocols. Perhaps most critically, the widely used ethylene oxide route presents unacceptable safety risks due to the explosive nature of ethylene oxide, alongside a tendency to produce complex mixtures of di-, tri-, and tetra-hydroxylated byproducts that are difficult and costly to separate, ultimately reducing the overall yield and purity of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes ethylene glycol as a hydroxyethylation reagent, fundamentally altering the risk and cost profile of the synthesis. By employing a specialized Raney skeleton catalyst under hydrogen pressure, this method facilitates a clean intermolecular dehydration condensation that produces only hydroxyethyl ethylenediamine and water as products. This atom-economic reaction pathway eliminates the need for hazardous ethylene oxide and corrosive chlorinating agents, drastically simplifying the safety infrastructure required for production. Furthermore, the high selectivity of the new catalytic system minimizes the formation of poly-substituted impurities, allowing for a straightforward purification process via reduced pressure rectification. This transition not only enhances operator safety but also streamlines the manufacturing workflow, making it an ideal candidate for reliable agrochemical intermediate supplier networks seeking to modernize their production capabilities.
Mechanistic Insights into Raney Catalyst-Mediated Dehydration Condensation
The core of this technological advancement lies in the sophisticated design of the heterogeneous catalyst system, which typically comprises a Raney skeleton of nickel or copper doped with auxiliary promoter elements such as manganese, chromium, zinc, or iron. The mechanistic pathway involves the activation of the hydroxyl group of ethylene glycol and the amine group of ethylenediamine on the catalyst surface, facilitating the elimination of a water molecule to form the C-N bond. The presence of hydrogen gas at pressures ranging from 0.5 MPa to 10.0 MPa plays a crucial role in maintaining the reduced state of the metal catalyst and suppressing side reactions that could lead to catalyst deactivation or the formation of unwanted oligomers. Optimization of the molar ratio between ethylene glycol and ethylenediamine, preferably between 1.05 and 1.15, ensures that the reaction kinetics favor the mono-hydroxyethylated product over di- or tri-substituted derivatives, thereby maximizing the yield of the target molecule.
Impurity control is rigorously managed through both kinetic regulation during the reaction and thermodynamic separation during the workup phase. The reaction temperature is carefully maintained between 100°C and 250°C, with an optimal window around 150°C, to balance reaction rate against thermal degradation or excessive byproduct formation. Following the reaction, the solid catalyst is efficiently removed via filtration, allowing for potential regeneration and reuse, which further contributes to process sustainability. The crude reaction liquid is then subjected to reduced pressure rectification at 2100 to 2200 Pa, where the target hydroxyethyl ethylenediamine is collected within a narrow boiling point range of 136-140°C. This precise distillation cut effectively separates the product from unreacted starting materials and any trace high-boiling impurities, ensuring the final material meets stringent purity specifications required for high-purity OLED material or pharmaceutical applications.
How to Synthesize Hydroxyethyl Ethylenediamine Efficiently
To implement this synthesis route effectively, manufacturers must adhere to strict operational parameters regarding catalyst preparation, reactor pressurization, and thermal management. The process begins with the precise loading of ethylenediamine and ethylene glycol into a high-pressure autoclave followed by the addition of the pre-activated Raney catalyst slurry. Detailed standard operating procedures regarding the sequential addition of reagents, the ramping of hydrogen pressure, and the control of exothermic heat during the dehydration phase are critical for ensuring batch-to-batch consistency and safety. The following guide outlines the standardized synthesis steps derived from the patent data to assist technical teams in replicating this high-yield process.
- Load ethylenediamine and ethylene glycol (molar ratio 1.00-1.30) along with a Raney nickel or copper catalyst into a high-pressure reactor.
- Purge the system and introduce hydrogen gas to achieve a pressure between 0.5 MPa and 10.0 MPa.
- Heat the mixture to 100-250°C with stirring to facilitate intermolecular dehydration condensation.
- Filter the catalyst after reaction completion and perform reduced pressure rectification (2100-2200Pa) collecting the fraction at 136-140°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ethylene glycol-based synthesis route offers transformative advantages in terms of cost structure and logistical reliability. By shifting away from volatile and expensive ethylene oxide to stable and commodity-grade ethylene glycol, manufacturers can significantly reduce raw material procurement costs and mitigate the risks associated with hazardous material transport and storage. The elimination of corrosive byproducts like hydrogen chloride removes the need for specialized scrubbing systems and expensive alloy reactors, leading to substantial capital expenditure savings and lower maintenance overheads. Furthermore, the simplified post-treatment process, which relies on basic filtration and distillation rather than complex extraction or chromatography, reduces utility consumption and shortens the overall production cycle time, enhancing the agility of the supply chain.
- Cost Reduction in Manufacturing: The substitution of high-cost, high-risk ethylene oxide with inexpensive ethylene glycol serves as a primary driver for lowering the variable cost of goods sold. Additionally, the high selectivity of the catalyst minimizes waste generation and maximizes the yield of the desired product per unit of raw material input, directly improving the mass balance efficiency of the plant. The ability to operate without organic solvents further reduces solvent purchase and recovery costs, contributing to a leaner and more economically resilient manufacturing model that supports cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Ethylene glycol and ethylenediamine are widely available commodity chemicals with stable global supply chains, reducing the vulnerability of production schedules to raw material shortages. The robustness of the Raney catalyst system allows for extended campaign lengths and potential catalyst recycling, minimizing downtime associated with frequent catalyst change-outs. This stability ensures consistent output volumes, enabling suppliers to meet demanding delivery schedules and reducing lead time for high-purity pharmaceutical intermediates even during periods of market fluctuation.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in high-pressure reactor systems suitable for commercial scale-up of complex fine chemicals. From an environmental perspective, the generation of water as the sole byproduct aligns perfectly with green chemistry principles, drastically reducing the burden on wastewater treatment facilities. The absence of halogenated waste streams and toxic gases simplifies regulatory compliance and permits easier expansion of production capacity in regions with strict environmental enforcement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is essential for technical teams assessing the integration of this method into existing production lines.
Q: Why is the ethylene glycol route preferred over the ethylene oxide route?
A: The ethylene glycol route eliminates the high safety risks associated with explosive ethylene oxide and avoids the formation of difficult-to-separate poly-hydroxylated byproducts, resulting in higher selectivity and simpler post-treatment.
Q: What type of catalyst is required for this synthesis?
A: The process utilizes a Raney skeleton catalyst composed of nickel or copper, promoted by auxiliary elements such as aluminum, manganese, chromium, zinc, or iron to enhance activity and selectivity.
Q: What purity levels can be achieved with this method?
A: Through optimized reaction conditions and reduced pressure distillation, the method consistently achieves gas phase purities exceeding 99% with molar yields often surpassing 95%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxyethyl Ethylenediamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering hydroxyethyl ethylenediamine that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the catalytic dehydration technology described in CN111116386B, we can offer a product profile that balances high quality with economic efficiency, catering to the exacting standards of the pharmaceutical and agrochemical sectors.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific supply chain requirements. Whether you require a Customized Cost-Saving Analysis to evaluate the economic impact of switching to this greener process or need to review specific COA data and route feasibility assessments for your next project, we are ready to provide the support you need. Contact us today to secure a reliable supply of high-quality intermediates and partner with a manufacturer dedicated to technological excellence and sustainable growth.
