Advanced Stereocontrolled Synthesis of Dicyclohexyl-18-Crown-6 for Commercial Scale-Up of Complex Macrocyclic Polyethers
Advanced Stereocontrolled Synthesis of Dicyclohexyl-18-Crown-6 for Commercial Scale-Up of Complex Macrocyclic Polyethers
The chemical industry continuously seeks robust methodologies for synthesizing macrocyclic polyethers with precise stereochemical control, particularly for applications in nuclear fuel reprocessing and advanced separation technologies. Patent CN102040584A introduces a transformative approach to the production of Dicyclohexyl-18-Crown-6 (DCH18C6), utilizing a specialized nano-ruthenium metal powder catalyst to achieve unprecedented levels of cis-isomer selectivity. This innovation addresses the longstanding challenge where traditional hydrogenation methods yield complex mixtures of stereoisomers, necessitating costly and yield-reducing purification steps. By leveraging high-pressure catalytic hydrogenation conditions between 100-200°C and 2-10 MPa, this technology enables the direct production of crude material with cis-isomer content exceeding 80%. For R&D directors and procurement specialists, this represents a pivotal shift towards more efficient, high-purity fine chemical intermediate manufacturing that minimizes waste and maximizes throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dicyclohexyl-18-Crown-6 via the hydrogenation of dibenzo-18-crown-6 has been plagued by poor stereoselectivity when using standard catalysts such as acidic Pichler Ruthenium. In these conventional processes, the resulting crude product typically contains a cis-isomer proportion ranging merely from one-third to two-thirds, creating a significant bottleneck for downstream processing. To achieve the requisite purity for effective strontium extraction, manufacturers are forced to employ labor-intensive purification techniques including column chromatography and repeated recrystallization in solvents like n-heptane. These additional steps not only drastically increase the consumption of solvents and energy but also lead to substantial product losses, thereby inflating the overall cost of goods sold. Furthermore, the variability in isomer ratios complicates quality control protocols, making it difficult to guarantee consistent performance in critical applications such as radioactive waste management.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN102040584A utilizes a uniquely prepared nano-ruthenium metal catalyst that fundamentally alters the reaction pathway to favor the formation of the cis-syn-cis isomer. By implementing this advanced catalytic system, the process achieves a cis-isomer content of over 80% directly from the reactor, effectively bypassing the need for rigorous purification steps like column chromatography. This leap in selectivity is achieved under relatively standard industrial hydrogenation conditions, utilizing solvents such as n-butanol, n-octanol, or toluene, which facilitates easy integration into existing manufacturing infrastructure. The ability to obtain high-purity high-purity dicyclohexyl-18-crown-6 without extensive post-reaction processing translates directly into improved operational efficiency and reduced environmental footprint. This novel approach not only simplifies the workflow but also ensures a more reliable supply of the active isomer required for high-performance separation tasks.
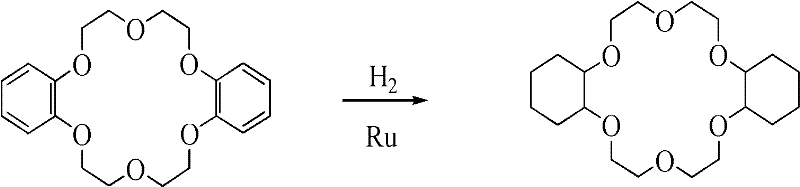
Mechanistic Insights into Nano-Ruthenium Catalyzed Hydrogenation
The core of this technological breakthrough lies in the specific preparation and activation of the nano-ruthenium catalyst, which dictates the stereochemical outcome of the hydrogenation. The patent details a multi-step catalyst synthesis involving the fusion of coarse ruthenium powder with potassium hydroxide and potassium nitrate, followed by precipitation and acidification. Crucially, the final activation step involves refluxing the dried metal powder in small molecule alcohols (C2-C4), a treatment that significantly reduces particle size and lowers surface oxygen content. Comparative data within the patent indicates that skipping this reflux step results in a drastic drop in cis-isomer selectivity, highlighting the importance of surface morphology in catalytic performance. This precise control over the catalyst's physical properties allows for a more uniform interaction with the aromatic rings of the dibenzo-18-crown-6 substrate, promoting the specific spatial arrangement required for the cis-configuration.
From an impurity control perspective, this mechanism offers a distinct advantage by minimizing the formation of the inactive trans-anti-cis isomer at the source rather than removing it later. In traditional catalytic cycles, non-selective hydrogenation leads to a statistical distribution of isomers that is thermodynamically difficult to separate due to similar physical properties. However, the nano-ruthenium system appears to kinetically favor the adsorption geometry that leads to the cis-product, thereby suppressing the formation of unwanted byproducts. This inherent selectivity reduces the complexity of the impurity profile, allowing for simpler analytical monitoring and easier compliance with stringent specifications required for specialty chemical applications. The robustness of this catalytic system also suggests potential for catalyst recovery and reuse, further enhancing the economic viability of the process.
How to Synthesize Dicyclohexyl-18-Crown-6 Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for scaling this reaction from laboratory benchtop to pilot plant operations with minimal friction. The process begins with the dissolution of the dibenzo-18-crown-6 starting material in a selected organic solvent, followed by the addition of the pre-activated nano-ruthenium catalyst under inert atmosphere conditions. The reaction mixture is then subjected to elevated hydrogen pressure and temperature in a stirred autoclave, where the hydrogenation proceeds to completion over a period of 2 to 24 hours depending on the specific batch parameters. Detailed standardized synthesis steps see the guide below.
- Dissolve dibenzo-18-crown-6 in a suitable organic solvent such as n-butanol or toluene and mix with the specialized nano-ruthenium metal catalyst.
- Conduct the catalytic hydrogenation reaction in an autoclave at temperatures between 100-200°C and hydrogen pressures ranging from 2-10 MPa for 2-24 hours.
- Separate the catalyst and solvent via centrifugation and concentration to isolate the crude product, which already possesses high cis-isomer content exceeding 80%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this nano-ruthenium catalyzed synthesis offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary value driver is the elimination of complex purification stages, which traditionally consume significant resources in terms of solvent procurement, waste disposal, and equipment occupancy time. By achieving high isomer purity directly from the reaction, manufacturers can drastically reduce the cycle time per batch, thereby increasing the overall throughput of the production facility without the need for capital expansion. This efficiency gain is particularly relevant for cost reduction in specialty chemical manufacturing, where margin pressures often dictate the feasibility of producing niche intermediates. Additionally, the simplified workflow reduces the dependency on specialized chromatography columns and large volumes of recrystallization solvents, mitigating supply chain risks associated with raw material availability.
- Cost Reduction in Manufacturing: The most significant financial impact stems from the removal of downstream purification steps such as column chromatography and repeated recrystallization, which are notoriously expensive and material-intensive. By producing a crude product with over 80% cis-isomer content, the process eliminates the need for these yield-eroding operations, leading to substantial savings in both raw material consumption and labor costs. Furthermore, the high conversion rates and selectivity minimize the generation of chemical waste, reducing the expenses associated with environmental compliance and hazardous waste disposal. This streamlined approach ensures that a higher percentage of the input material is converted into saleable product, optimizing the overall cost structure of the manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of the nano-ruthenium catalyst system contributes to a more stable and predictable production schedule, which is critical for maintaining continuity in the supply of critical intermediates. Since the reaction conditions are compatible with standard industrial hydrogenation equipment and common solvents like n-butanol or toluene, there is no need for exotic or hard-to-source reagents that could disrupt the supply chain. The potential for catalyst recovery and reuse further insulates the production process from fluctuations in precious metal prices, providing long-term cost stability. This reliability makes the manufacturer a more dependable partner for clients who require consistent quality and timely delivery for their own downstream applications.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving basic centrifugation and concentration, makes this process highly amenable to commercial scale-up from kilogram to multi-ton quantities. Unlike processes relying on delicate chromatographic separations that are difficult to scale linearly, this hydrogenation method can be easily adapted to larger reactors with standard agitation and pressure controls. Moreover, the reduction in solvent usage and waste generation aligns with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental burden of the facility. This scalability ensures that the technology can meet growing market demand for commercial scale-up of complex macrocyclic polyethers while maintaining a sustainable operational profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for Dicyclohexyl-18-Crown-6. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's potential. Understanding these details is essential for stakeholders assessing the feasibility of integrating this method into their existing production portfolios.
Q: Why is the cis-isomer content critical for Dicyclohexyl-18-Crown-6 applications?
A: The cis-syn-cis isomer exhibits superior extraction capabilities for Strontium-90 (Sr2+) in acidic solutions compared to the trans isomer, making high stereoselectivity essential for nuclear waste treatment efficiency.
Q: How does the nano-ruthenium catalyst preparation influence reaction selectivity?
A: The specific reflux treatment of the ruthenium powder in small molecule alcohols reduces particle size and surface oxygen content, which is the key mechanistic factor driving the high formation of the desired cis-isomer.
Q: Does this synthesis method require extensive downstream purification?
A: No, unlike conventional methods requiring column chromatography or repeated recrystallization, this patented process yields over 80% cis-isomer content directly, significantly simplifying the workup procedure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicyclohexyl-18-Crown-6 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in advanced separation materials and are well-positioned to leverage this patented technology for your specific needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Dicyclohexyl-18-Crown-6 meets the exacting standards required for nuclear and pharmaceutical applications. Our commitment to technical excellence allows us to deliver high-quality intermediates that empower your downstream processes with reliability and consistency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements and application constraints. We encourage potential partners to contact us directly to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us collaborate to bring this high-efficiency production method to your organization, securing a competitive advantage in the global market for specialty chemicals.
