Advanced Manufacturing of Dicyclohexyl-18-Crown-6: Technical Breakthroughs for Industrial Extraction
Introduction to Advanced Crown Ether Manufacturing
The field of macrocyclic polyethers, commonly known as crown ethers, represents a cornerstone of modern separation science and coordination chemistry. Specifically, Dicyclohexyl-18-Crown-6 (DCH18C6) has garnered immense attention for its exceptional capability in selectively extracting metal ions, including radioactive strontium-90 from nuclear waste streams and various alkali metals in analytical applications. However, the industrial viability of DCH18C6 has historically been constrained by complex stereochemical challenges. As detailed in patent CN1709888A, the efficacy of DCH18C6 as an extractant is not merely a function of its chemical formula but is critically dependent on its立体 configuration. The patent discloses a groundbreaking改进 synthesis process that addresses the longstanding issues of low yield and poor stereoisomer distribution, specifically targeting the enrichment of the active cis-syn-cis isomer. This technical evolution marks a significant shift from traditional methods that often resulted in intractable mixtures, offering a robust pathway for the reliable crown ether supplier market to meet the stringent purity demands of high-end extraction processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in earlier patents like ZL98125085.8, relied heavily on neutral Pichler-ruthenium catalysts operating under relatively mild conditions, typically around 70-80°C. While these conditions were manageable, they suffered from severe kinetic and thermodynamic limitations that hindered industrial scalability. The primary drawback was the inability to effectively control the stereochemical outcome of the hydrogenation reaction, leading to a product mixture where the ratio of the active cis-syn-cis isomer (Isomer A) to the inactive cis-anti-cis isomer (Isomer B) was often less than 2. This imbalance had catastrophic consequences for downstream processing; the high content of Isomer B prevented the product from crystallizing at room temperature, resulting instead in a viscous, sticky paste that was extremely difficult to filter, dry, or purify. Furthermore, the overall industrial yield for the precursor synthesis was stagnant at approximately 30%, creating significant waste and inflating the cost basis for manufacturers attempting to produce high-purity OLED material or pharmaceutical intermediates requiring similar macrocyclic scaffolds.
The Novel Approach
The innovative process outlined in CN1709888A introduces a multi-faceted strategy to overcome these bottlenecks, fundamentally altering both the precursor synthesis and the final hydrogenation steps. In the synthesis of the intermediate dibenzo-18-crown-6, the introduction of tetrabutylammonium bromide acts as a highly efficient phase transfer catalyst, which dramatically accelerates the macrocyclization reaction and boosts the industrial yield from roughly 30% to over 40%. 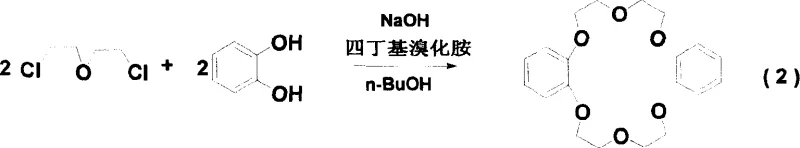 More critically, the final hydrogenation step employs a newly prepared and activated acidic Pichler-ruthenium catalyst with a pH range of 3-5. By strictly controlling the reaction temperature between 130-140°C and utilizing redistilled n-butanol as a solvent, this novel approach drives the reaction to completion within 3-5 hours. The result is a transformative increase in total yield to over 90% and, crucially, a stereoisomer ratio where Isomer A dominates, allowing the product to crystallize spontaneously upon cooling. This shift from a paste to a crystalline solid simplifies the entire manufacturing workflow, representing a major leap forward in the commercial scale-up of complex polymer additives and specialty chemicals.
More critically, the final hydrogenation step employs a newly prepared and activated acidic Pichler-ruthenium catalyst with a pH range of 3-5. By strictly controlling the reaction temperature between 130-140°C and utilizing redistilled n-butanol as a solvent, this novel approach drives the reaction to completion within 3-5 hours. The result is a transformative increase in total yield to over 90% and, crucially, a stereoisomer ratio where Isomer A dominates, allowing the product to crystallize spontaneously upon cooling. This shift from a paste to a crystalline solid simplifies the entire manufacturing workflow, representing a major leap forward in the commercial scale-up of complex polymer additives and specialty chemicals.
Mechanistic Insights into Acidic Pichler-Ruthenium Catalytic Hydrogenation
The core of this technological advancement lies in the precise manipulation of catalyst acidity and reaction thermodynamics to favor the formation of the desired stereoisomer. The patent elucidates that the activity of Pichler-ruthenium catalysts follows a distinct order: basic < neutral < acidic. By utilizing the acidic form, specifically adjusted to a pH of 3-5 through nitric acid treatment, the catalyst achieves maximum turnover frequency. This high activity allows the reaction to be conducted at elevated temperatures of 130-140°C, a range that is thermodynamically favorable for the formation of the cis-syn-cis conformation. 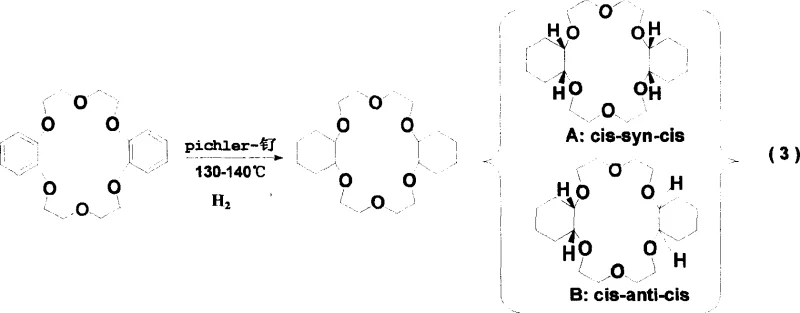 At these temperatures, the energy barrier for the hydrogenation of the aromatic rings is overcome efficiently, while the specific interaction between the acidic catalyst surface and the substrate appears to direct the addition of hydrogen in a manner that preserves the necessary symmetry for Isomer A. This mechanistic control is vital because the extraction performance of crown ethers is intimately linked to their molecular geometry; the cis-syn-cis isomer offers a planar cavity that optimally coordinates with guest cations, whereas other isomers distort this cavity, rendering them ineffective for high-precision separation tasks.
At these temperatures, the energy barrier for the hydrogenation of the aromatic rings is overcome efficiently, while the specific interaction between the acidic catalyst surface and the substrate appears to direct the addition of hydrogen in a manner that preserves the necessary symmetry for Isomer A. This mechanistic control is vital because the extraction performance of crown ethers is intimately linked to their molecular geometry; the cis-syn-cis isomer offers a planar cavity that optimally coordinates with guest cations, whereas other isomers distort this cavity, rendering them ineffective for high-precision separation tasks.
Furthermore, the process incorporates a robust impurity control mechanism through selective crystallization. Since the new method ensures that the content of Isomer A exceeds 89% directly from the reactor, the subsequent purification becomes a straightforward physical separation rather than a complex chemical resolution. The crude product, which is now a free-flowing crystal rather than a gum, can be recrystallized from C6-C8 alkanes such as n-heptane. 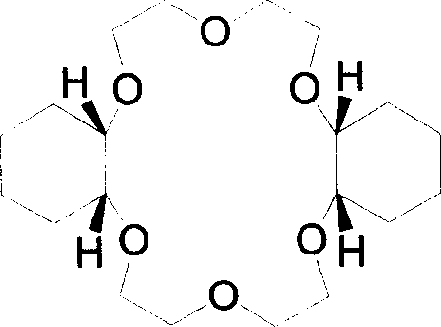 This step leverages the solubility differences between the highly symmetric Isomer A and the less symmetric Isomer B. As the solution cools, the planar Isomer A packs efficiently into the crystal lattice, excluding the distorted Isomer B, thereby elevating the purity to over 98%. This level of stereochemical purity is essential for applications in biochemistry and analytical chemistry where batch-to-batch consistency in extraction coefficients is non-negotiable, ensuring that the final high-purity dicyclohexyl-18-crown-6 meets the rigorous specifications required by global regulatory bodies.
This step leverages the solubility differences between the highly symmetric Isomer A and the less symmetric Isomer B. As the solution cools, the planar Isomer A packs efficiently into the crystal lattice, excluding the distorted Isomer B, thereby elevating the purity to over 98%. This level of stereochemical purity is essential for applications in biochemistry and analytical chemistry where batch-to-batch consistency in extraction coefficients is non-negotiable, ensuring that the final high-purity dicyclohexyl-18-crown-6 meets the rigorous specifications required by global regulatory bodies.
How to Synthesize Dicyclohexyl-18-Crown-6 Efficiently
The synthesis of Dicyclohexyl-18-Crown-6 via this improved protocol requires strict adherence to the specified reaction parameters to ensure safety and reproducibility. The process begins with the preparation of bis(2-chloroethyl)ether, followed by the macrocyclization to dibenzo-18-crown-6, and concludes with the critical stereoselective hydrogenation. Each step involves specific handling of reagents such as thionyl chloride and high-pressure hydrogen, necessitating specialized equipment and operational expertise. The detailed standardized synthesis steps, including exact molar ratios, temperature ramping rates, and workup procedures, are provided in the guide below to assist technical teams in replicating this high-yield pathway.
- Preparation of Bis(2-chloroethyl)ether via reaction of diethylene glycol with thionyl chloride at 110-120°C.
- Synthesis of Dibenzo-18-Crown-6 using catechol and the chloro-ether intermediate with tetrabutylammonium bromide as a phase transfer catalyst.
- Stereoselective hydrogenation of Dibenzo-18-Crown-6 using acidic Pichler-Ruthenium catalyst at 130-140°C to maximize cis-syn-cis isomer formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this improved synthesis route offers compelling economic and operational benefits that extend far beyond simple yield metrics. The transition from a process that generates viscous, unprocessable pastes to one that yields free-flowing crystals fundamentally alters the cost structure of production. By eliminating the need for extensive drying times, specialized pumping equipment for sludge, and complex chromatographic separations to remove sticky byproducts, the operational expenditure (OPEX) is significantly reduced. The ability to achieve yields exceeding 90% means that raw material consumption per kilogram of finished product is drastically lowered, directly impacting the cost reduction in extraction agent manufacturing. This efficiency gain allows suppliers to offer more competitive pricing without compromising on the quality margins that are critical for sensitive applications in the nuclear and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The implementation of tetrabutylammonium bromide in the precursor step and the switch to acidic catalysts in the hydrogenation step creates a synergistic effect on production costs. The higher reaction rates shorten the cycle time per batch, increasing the throughput of existing reactor assets without the need for capital expansion. Additionally, the elimination of the 'sticky paste' issue removes a major bottleneck in filtration and drying, reducing labor hours and energy consumption associated with solvent removal. These qualitative improvements translate into substantial cost savings, making the domestic production of this specialty chemical more viable against international competitors.
- Enhanced Supply Chain Reliability: One of the most significant risks in the supply of complex intermediates is batch failure due to poor crystallization or off-spec isomer ratios. The robust nature of this new process, which consistently delivers a crystalline product with high Isomer A content, mitigates this risk effectively. Manufacturers can guarantee delivery schedules with greater confidence, knowing that the likelihood of having to reprocess or scrap a batch due to physical form issues is minimal. This reliability is crucial for reducing lead time for high-purity dicyclohexyl-18-crown-6 derivatives, ensuring that downstream customers in the agrochemical and electronic materials sectors receive their materials on time, every time.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents like n-butanol and n-heptane which are readily available and easily recycled. The use of a heterogeneous-style acidic ruthenium catalyst that can be filtered and potentially regenerated aligns with green chemistry principles by minimizing heavy metal waste. Furthermore, the high atom economy of the hydrogenation step reduces the volume of organic waste generated per unit of product. This environmental profile simplifies the permitting process for scale-up and ensures long-term compliance with increasingly stringent environmental regulations, securing the supply chain against future regulatory shocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Dicyclohexyl-18-Crown-6 based on the patented technology. Understanding these nuances is essential for partners looking to integrate this material into their own formulations or separation processes. The answers provided are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industrial decision-makers.
Q: Why is the cis-syn-cis isomer of Dicyclohexyl-18-Crown-6 preferred for extraction?
A: The cis-syn-cis isomer possesses superior planarity and symmetry compared to other stereoisomers, which significantly enhances its ability to form stable host-guest complexes with metal cations like strontium-90 and alkali metals.
Q: How does the acidic Pichler-Ruthenium catalyst improve the process?
A: Unlike neutral catalysts used in previous methods, the acidic Pichler-Ruthenium catalyst (pH 3-5) exhibits higher activity, allowing the reaction to proceed efficiently at 130-140°C, resulting in yields over 90% and a dominant cis-syn-cis isomer ratio.
Q: What are the purification advantages of this new synthetic route?
A: The new process produces a product that crystallizes readily at room temperature due to the high isomer A content, eliminating the difficult-to-process viscous pastes associated with older methods and simplifying downstream purification via n-heptane recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicyclohexyl-18-Crown-6 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the acidic Pichler-Ruthenium catalytic route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chromatography and spectroscopy tools to verify the isomer ratio and metal content of every batch, guaranteeing that our Dicyclohexyl-18-Crown-6 performs consistently in your critical extraction applications.
We invite you to collaborate with us to optimize your supply chain for this critical specialty chemical. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By leveraging our optimized synthesis platform, we can help you secure a stable source of high-performance crown ethers. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical capabilities can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
