Advanced Catalytic Hydrogenation Route for High-Purity Ivabradine Intermediates and Commercial Scalability
Advanced Catalytic Hydrogenation Route for High-Purity Ivabradine Intermediates and Commercial Scalability
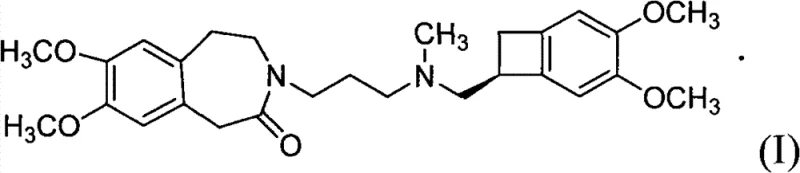
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for cardiovascular therapeutics like Ivabradine. Patent CN101768116B introduces a significant technological advancement in the preparation of S 16257-2, also known chemically as 3-{3-[{[(7S)-3,4-dimethoxy dicyclo [4.2.0] pungent-1,3,5-triolefin-7-yl]-methyl } (methyl) amino] propyl group }-7,8-dimethoxy-1,3,4,5-tetrahydrochysene-2H-3-benzazepine-2-ketone. This patent discloses a streamlined preparation method that fundamentally alters the manufacturing landscape by replacing complex purification steps with efficient catalytic hydrogenation and direct salt formation. The core innovation lies in the conversion of a specific unsaturated precursor into the saturated intermediate via catalytic hydrogenation, followed by a base-catalyzed coupling reaction that bypasses the need for column chromatography. This approach not only enhances the chemical purity of the final active pharmaceutical ingredient (API) but also addresses critical supply chain bottlenecks associated with traditional synthesis routes. By leveraging this technology, manufacturers can achieve superior process control and scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those detailed in European patent specification EP 0534859, have historically plagued the production of Ivabradine with significant inefficiencies that hinder large-scale commercialization. The most critical drawback of these conventional routes is the absolute reliance on column chromatography for the purification of key intermediates and the final product. In an industrial setting, column chromatography is notoriously resource-intensive, requiring vast quantities of silica gel and organic solvents, which drastically inflates production costs and generates substantial hazardous waste. Furthermore, the physical losses inherent in chromatographic separation lead to suboptimal overall yields, often rendering the process economically unviable for high-volume manufacturing. The complexity of these multi-step purifications also extends the production cycle time, creating vulnerabilities in the supply chain where delays in one purification stage can halt the entire downstream process. Consequently, there has been a persistent demand for a synthetic strategy that eliminates these chromatographic dependencies while maintaining stringent quality standards.
The Novel Approach
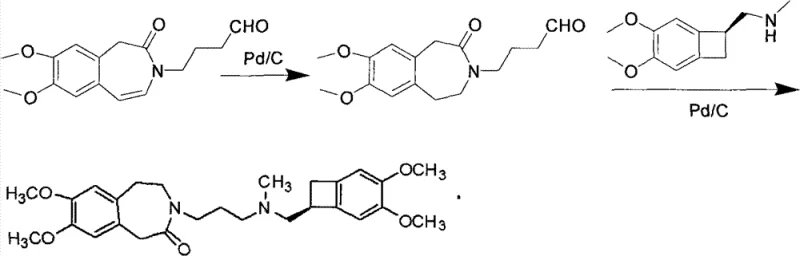
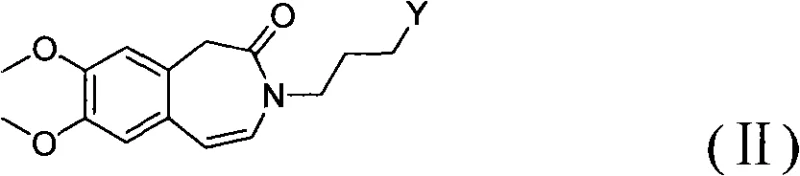
The methodology presented in CN101768116B offers a transformative solution by integrating catalytic hydrogenation with a direct telescoping alkylation strategy. As illustrated in the reaction scheme, the process begins with the catalytic hydrogenation of a Formula II compound, which contains an unsaturated bond within the benzazepine ring system. This step utilizes a heterogeneous catalyst, preferably palladium on carbon (Pd/C), under mild conditions to selectively reduce the double bond, yielding the saturated Formula III intermediate. Unlike previous methods that would isolate and purify this intermediate, the novel approach allows the crude Formula III compound to proceed directly to the next reaction stage. This intermediate then undergoes a condensation reaction with a Formula IV compound, specifically a cyclobutane amine salt, under base catalysis. The elimination of isolation steps between hydrogenation and alkylation represents a paradigm shift in process chemistry, effectively collapsing multiple unit operations into a more continuous and efficient workflow that maximizes throughput.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and Base-Mediated Alkylation
The success of this synthetic route hinges on the precise control of the catalytic hydrogenation mechanism and the subsequent nucleophilic substitution. In the first stage, the unsaturated benzazepine derivative (Formula II) is subjected to hydrogen gas in the presence of a Pd/C catalyst. The mechanism involves the adsorption of both the hydrogen molecules and the alkene substrate onto the surface of the palladium metal. The hydrogen molecules dissociate into atomic hydrogen, which then adds across the carbon-carbon double bond of the benzazepine ring in a syn-addition fashion. This reduction is highly chemoselective; the reaction conditions (20-60°C, 1-10 atm) are tuned to reduce the alkene without affecting the carbonyl group of the lactam or the methoxy substituents on the aromatic rings. The choice of solvent, typically methanol or ethanol, plays a crucial role in solubilizing the substrate while facilitating the interaction with the catalyst surface. The result is the clean formation of the saturated 3-(3-hydroxypropyl)-7,8-dimethoxy-4',5-dihydro-1H-benzazepine-2(3H)-one (Formula III), which serves as the pivotal scaffold for the final assembly.
Following hydrogenation, the reaction mixture transitions to the alkylation phase, where the secondary amine of the Formula IV compound acts as a nucleophile. This step is driven by a weak inorganic base, such as anhydrous potassium carbonate, which serves to deprotonate the amine or facilitate the leaving group departure from the propyl chain of the Formula III intermediate. The addition of a halide source, specifically potassium iodide (KI), acts as a catalyst for the nucleophilic substitution, likely through an in-situ Finkelstein-type activation where the hydroxyl or protected hydroxyl group is converted into a more reactive iodide species. This activation lowers the energy barrier for the SN2 attack by the cyclobutane amine, ensuring high conversion rates even in the presence of crude impurities from the previous hydrogenation step. The use of acetone as a solvent in this stage is strategic, as it dissolves the organic reactants while allowing the inorganic salts to remain largely insoluble or easily filterable, simplifying the downstream workup. This mechanistic synergy ensures that the final coupling proceeds with high fidelity, minimizing the formation of regioisomers or over-alkylated byproducts.
How to Synthesize Ivabradine Hydrochloride Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters to ensure reproducibility and safety on a commercial scale. The process begins with the suspension of the unsaturated precursor in an alcoholic solvent, followed by the introduction of the palladium catalyst and hydrogen gas. Maintaining the temperature between 30-40°C is critical to prevent thermal degradation while ensuring sufficient kinetic energy for the reduction. Once the hydrogenation is complete, the catalyst is removed via simple filtration, a unit operation that is far more scalable and cost-effective than chromatography. The filtrate, containing the crude saturated intermediate, is then concentrated and redissolved in acetone for the coupling reaction. The addition of the cyclobutane amine salt, base, and iodide promoter initiates the final bond formation. After refluxing to drive the reaction to completion, the mixture is worked up through standard acid-base extraction techniques to isolate the free base, which is subsequently converted to the hydrochloride salt via recrystallization. For a detailed, step-by-step breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the technical guide below.
- Perform catalytic hydrogenation on the unsaturated benzazepine precursor (Formula II) using Pd/C in an alcoholic solvent at 30-40°C to obtain the saturated intermediate (Formula III).
- React the crude Formula III intermediate with the cyclobutane amine salt (Formula IV) in the presence of anhydrous potassium carbonate and potassium iodide in acetone.
- Filter off the catalyst, concentrate the filtrate, and perform acid-base workup followed by recrystallization to isolate the pure Ivabradine hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN101768116B process offers tangible strategic benefits that extend beyond mere chemical elegance. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures and enhanced supply security. By eliminating the column chromatography step, the process removes a major bottleneck that typically consumes significant time, labor, and consumable resources. This streamlining allows for faster batch turnover times, enabling manufacturers to respond more agilely to market demand fluctuations. Furthermore, the use of common, commodity-grade reagents such as Pd/C, potassium carbonate, and acetone ensures that raw material sourcing remains stable and unaffected by the volatility often seen with specialized chiral reagents or exotic catalysts. The robustness of this route makes it an ideal candidate for reliable API intermediate supplier partnerships aiming for long-term cost stability.
- Cost Reduction in Manufacturing: The economic impact of removing column chromatography cannot be overstated, as this step is traditionally one of the most expensive components of fine chemical synthesis. Silica gel is a consumable cost that scales linearly with production volume, and the solvents required for elution represent a massive expense in both purchase and waste disposal. By replacing this with a filtration-based catalyst removal and a direct telescoping reaction, the new method significantly reduces the consumption of these high-cost materials. Additionally, the ability to use the crude intermediate without purification minimizes yield losses associated with isolation, thereby improving the overall mass balance of the process. This efficiency gain leads to substantial cost savings in the final cost of goods sold (COGS), making the end product more competitive in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the simplicity and robustness of the underlying chemistry. Complex purification steps introduce multiple points of failure where batches can be rejected due to purity issues, leading to unpredictable lead times. The novel hydrogenation-alkylation sequence is inherently more robust, with fewer unit operations that require precise manual intervention. The use of atmospheric pressure hydrogenation, as opposed to high-pressure systems, further reduces the engineering complexity and safety risks associated with the plant infrastructure. This simplicity ensures that production schedules are more predictable and that the risk of batch failure is minimized. For buyers, this translates to a more reliable supply of high-purity pharmaceutical intermediates, reducing the need for excessive safety stock and mitigating the risk of production stoppages due to material shortages.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns perfectly with the principles of Green Chemistry. The reduction in solvent usage and the elimination of silica gel waste significantly lower the environmental footprint of the manufacturing process. Waste streams are simpler to treat, primarily consisting of aqueous salt solutions and recoverable organic solvents, rather than complex mixtures of silica and diverse organics. This ease of waste management facilitates compliance with increasingly stringent environmental regulations, reducing the liability and permitting hurdles for manufacturing sites. Moreover, the process is highly scalable; the transition from kilogram to multi-ton scale is straightforward because the reaction conditions (temperature, pressure) are mild and the equipment requirements (standard reactors, filters) are ubiquitous in the fine chemical industry. This scalability ensures that the supply can grow in tandem with the commercial success of the final drug product.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common inquiries regarding the specific reaction conditions, impurity profiles, and scalability factors inherent to this patented method. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for technical decision-making. Whether you are assessing the feasibility of technology transfer or evaluating the quality of the supply, these answers clarify the operational realities of producing Ivabradine via this advanced route.
Q: How does this new method improve upon the traditional EP 0534859 synthesis route?
A: The traditional route described in EP 0534859 relies heavily on column chromatography for purification, which results in low yields and high operational costs. The novel method in CN101768116B eliminates the need for column chromatography entirely by utilizing a direct catalytic hydrogenation followed by a one-pot alkylation, significantly improving overall yield and industrial feasibility.
Q: What are the critical reaction conditions for the hydrogenation step?
A: The hydrogenation of the Formula II compound is optimally conducted at atmospheric pressure (1 atm) and a temperature range of 30-40°C using a palladium on carbon (Pd/C) catalyst in methanol. These mild conditions ensure high selectivity for the double bond reduction without affecting other sensitive functional groups.
Q: Why is the direct use of the crude Formula III intermediate advantageous?
A: Using the crude Formula III intermediate directly in the subsequent alkylation step avoids the time-consuming and yield-loss-prone isolation and purification processes. This telescoping of steps reduces solvent consumption, minimizes material handling, and accelerates the total production cycle time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ivabradine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising efficiencies of the CN101768116B route can be fully realized in a GMP-compliant environment. We understand that the production of cardiovascular intermediates demands rigorous adherence to quality standards; therefore, our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities at ppm levels. Our commitment to process optimization allows us to deliver high-purity Ivabradine intermediates that meet the exacting requirements of global regulatory bodies, providing a secure foundation for your drug development and commercialization efforts.
We invite you to leverage our technical proficiency to optimize your supply chain and reduce your overall manufacturing costs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced catalytic hydrogenation capabilities can serve as a cornerstone for your successful Ivabradine production strategy, ensuring a steady supply of high-quality materials for the global market.
