Advanced Manufacturing of Chiral Benzocyclobutane Derivatives for Cardiovascular Therapeutics
The pharmaceutical landscape for cardiovascular therapeutics has been significantly advanced by the development of Ivabradine, a selective inhibitor of the If current in the sinoatrial node. Central to the efficient manufacturing of this critical medication is the availability of high-quality chiral building blocks, specifically (1S)-4,5-dimethoxy-1-(methylaminomethyl)-benzocyclobutane. Patent CN1332933C discloses a groundbreaking synthetic methodology that addresses historical bottlenecks in producing this key API intermediate. Unlike traditional routes that struggled with poor stereochemical control and dismal mass balance, this innovative approach leverages a strategic resolution of primary amines to unlock substantial process improvements. For global reliable API intermediate supplier networks, adopting such refined methodologies is essential to ensure consistent drug quality and availability. The following analysis details how this specific chemical architecture supports the broader supply chain for heart failure medications.
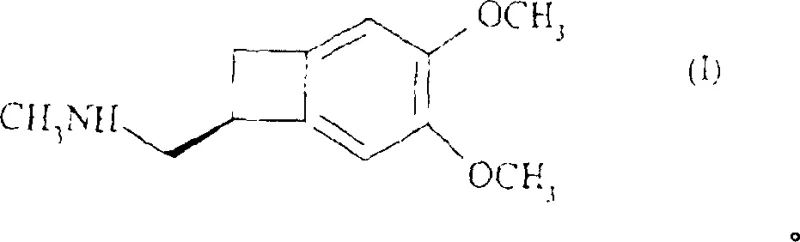
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral benzocyclobutane derivatives relied heavily on the resolution of secondary amine intermediates, a strategy fraught with significant inefficiencies that hampered large-scale production. Prior art methods, such as those utilizing camphorsulfonic acid for resolving secondary amines, suffered from catastrophic yield losses, often resulting in overall process yields as low as 2-3%. This inefficiency was primarily attributed to the poor selectivity during the crystallization of the secondary amine salts, where the desired enantiomer failed to precipitate effectively from the reaction mixture. Furthermore, the requirement for multiple protection and deprotection steps prior to the resolution added unnecessary complexity and waste generation to the manufacturing workflow. For procurement managers focused on cost reduction in pharmaceutical manufacturing, these legacy processes represented a significant financial burden due to the excessive consumption of raw materials and the high cost of waste disposal. The inability to efficiently recover the unwanted enantiomer further exacerbated the economic viability of these conventional routes.
The Novel Approach
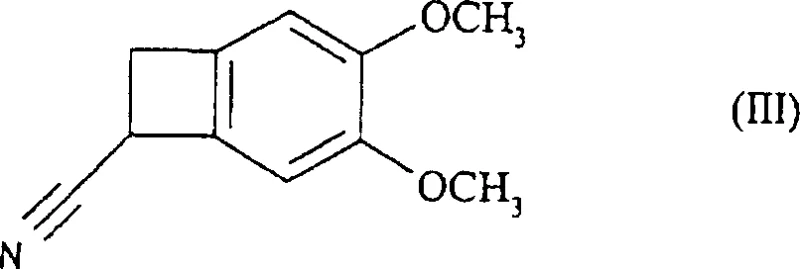
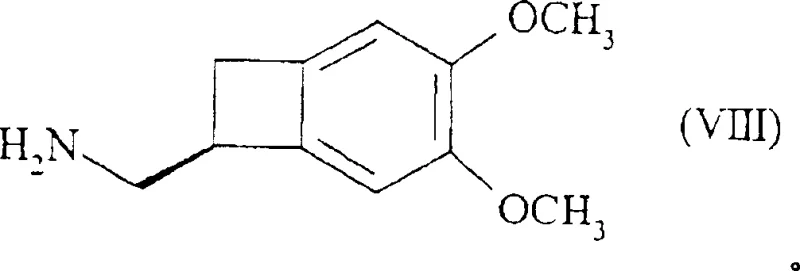
In stark contrast, the methodology described in CN1332933C introduces a paradigm shift by targeting the resolution of the primary amine precursor (Formula IV) rather than the secondary amine. By reacting the racemic primary amine with optically active diacidic compounds, specifically N-acetyl-L-glutamic acid, the process achieves a dramatic improvement in stereochemical separation efficiency. This strategic modification allows for the isolation of the desired (S)-configuration salt with exceptional selectivity, thereby boosting the overall yield from the nitrile starting material to approximately 30%. The elimination of early-stage methylation prior to resolution simplifies the molecular handling and reduces the steric hindrance that often plagues secondary amine crystallizations. This streamlined pathway not only enhances the commercial scale-up of complex chiral amines but also aligns with green chemistry principles by minimizing solvent usage and reaction steps. The robustness of this new route makes it an ideal candidate for industrial adoption by forward-thinking chemical manufacturers.
Mechanistic Insights into Diacidic Chiral Resolution
The core innovation of this synthesis lies in the specific interaction between the primary amine and the diacidic resolving agent, which facilitates a highly ordered crystal lattice formation favorable for the (S)-enantiomer. Unlike monobasic acids such as (R)-camphorsulfonic acid or various N-acetyl amino acids which yielded poor or negligible separation ratios (often near 50/50), the diacidic nature of N-acetyl-L-glutamic acid provides two carboxylic acid groups capable of forming stable salt bridges. This dual-point interaction significantly increases the energy difference between the diastereomeric salts, allowing the desired (S)/(R) ratio to reach levels as high as 93/7 in the initial precipitation. Subsequent recrystallization from alcoholic solvents like ethanol or ethanol/water mixtures further purifies the salt, pushing the enantiomeric excess to greater than 99%. For R&D Directors, understanding this mechanism is crucial for troubleshooting potential impurities and ensuring batch-to-batch consistency in high-purity cardiovascular intermediates. The choice of solvent system plays a pivotal role, as the solubility differential between the diastereomers is maximized in polar protic environments.
Following the successful resolution, the process employs a classic carbamate protection strategy to facilitate the final reduction to the secondary amine without racemization. The optically active primary amine is reacted with ethyl chloroformate to form a stable carbamate intermediate (Formula IX), which serves as a robust precursor for the subsequent hydride reduction. This step is critical because direct alkylation of the primary amine could lead to over-alkylation or loss of optical purity; the carbamate route ensures precise control over the nitrogen substitution. The reduction is typically carried out using powerful hydride sources like lithium aluminum hydride or sodium bis(2-methoxyethoxy)aluminum hydride, which cleanly convert the carbamate to the methylamine functionality. Finally, treatment with gaseous hydrogen chloride or HCl in ethyl acetate converts the free base into the stable hydrochloride salt, ensuring long-term storage stability and ease of handling for downstream API synthesis. This meticulous control over every functional group transformation guarantees the integrity of the chiral center throughout the entire synthetic sequence.
How to Synthesize (1S)-4,5-dimethoxy-1-(methylaminomethyl)-benzocyclobutane Efficiently
The practical execution of this synthesis requires careful attention to reaction conditions, particularly during the catalytic hydrogenation and resolution phases, to maximize throughput and quality. The process begins with the reduction of the nitrile starting material using Raney nickel under hydrogen pressure, a step that must be monitored to prevent over-reduction or catalyst poisoning. Once the racemic amine is obtained, the critical resolution step with N-acetyl-L-glutamic acid demands precise temperature control during reflux and cooling to optimize crystal growth and purity. Detailed standard operating procedures regarding solvent ratios, stirring rates, and filtration techniques are essential for replicating the high yields reported in the patent literature. For manufacturing teams looking to implement this technology, having access to standardized protocols is vital for reducing lead time for high-purity pharmaceutical intermediates.
- Reduce the racemic nitrile precursor (Formula III) using catalytic hydrogenation with Raney nickel or chemical reduction to obtain the primary amine.
- Perform chiral resolution of the primary amine using N-acetyl-L-glutamic acid in an alcoholic solvent to isolate the (S)-enantiomer salt with high selectivity.
- Convert the resolved amine to a carbamate intermediate, followed by hydride reduction and salt formation to yield the final high-purity hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers profound benefits that extend beyond mere chemical yield, directly impacting the bottom line and supply security for pharmaceutical companies. The transition from a low-yielding secondary amine resolution to a high-efficiency primary amine resolution fundamentally alters the cost structure of the intermediate, eliminating the need to purchase excessive quantities of starting materials to compensate for process losses. This inherent efficiency translates into significant raw material savings and a reduced environmental footprint, which is increasingly important for compliance with global sustainability standards. Furthermore, the reliance on commodity chemicals such as ethanol, methanol, and Raney nickel ensures that the supply chain is not vulnerable to the shortages or price volatility often associated with exotic chiral catalysts. For Supply Chain Heads, this robustness means greater predictability in delivery schedules and a lower risk of production stoppages due to material unavailability.
- Cost Reduction in Manufacturing: The drastic improvement in overall yield from roughly 2-3% to 30% implies a massive reduction in the cost of goods sold per kilogram of the final intermediate. By eliminating the wasteful resolution of secondary amines, manufacturers can avoid the expensive disposal of large volumes of mother liquor containing valuable chiral material. Additionally, the simplified process flow reduces utility consumption and labor hours required per batch, contributing to substantial operational expenditure savings. These efficiencies allow suppliers to offer more competitive pricing structures without compromising on quality margins, providing a clear economic advantage for long-term procurement contracts.
- Enhanced Supply Chain Reliability: The use of widely available reagents and standard unit operations such as filtration and crystallization ensures that production can be easily scaled across multiple manufacturing sites if necessary. Unlike processes dependent on single-source biocatalysts or precious metals, this chemical route utilizes resilient infrastructure that is common in fine chemical plants worldwide. This flexibility mitigates the risk of supply disruptions caused by geopolitical issues or vendor-specific production failures. Consequently, pharmaceutical buyers can secure a more stable supply of critical intermediates, ensuring continuity for their own finished dosage form production lines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and conditions that are manageable in large-scale reactors without requiring specialized high-pressure or cryogenic equipment. The ability to recrystallize intermediates to high purity levels reduces the burden on downstream purification steps, minimizing solvent waste and energy usage. This alignment with green chemistry principles facilitates easier regulatory approval and environmental permitting, accelerating the time to market for new generic or branded formulations relying on this intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality assurance of this chiral benzocyclobutane derivative. These insights are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding vendor qualification and process integration.
Q: Why is the primary amine resolution superior to secondary amine resolution for this intermediate?
A: Resolving the primary amine (Formula IV) using diacidic compounds like N-acetyl-L-glutamic acid avoids the extremely low yields (2-3%) associated with resolving the secondary amine, boosting overall process efficiency to approximately 30%.
Q: What level of enantiomeric purity can be achieved with this novel process?
A: The optimized resolution and recrystallization steps ensure an enantiomeric purity greater than 99%, meeting the stringent requirements for cardiovascular API manufacturing.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the process utilizes robust reagents like Raney nickel and standard solvents such as ethanol and methanol, making it highly suitable for commercial scale-up of complex chiral amines without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1S)-4,5-dimethoxy-1-(methylaminomethyl)-benzocyclobutane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the success of cardiovascular therapies depends on the unwavering quality and availability of their key precursors. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (1S)-4,5-dimethoxy-1-(methylaminomethyl)-benzocyclobutane meets the highest industry standards for enantiomeric and chemical purity. We are committed to being a strategic extension of your R&D and supply chain teams, offering the technical expertise required to navigate complex regulatory landscapes.
We invite you to collaborate with us to optimize your sourcing strategy for this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's economic and technical outcomes. Let us help you secure a reliable supply chain for your next generation of cardiovascular medicines.
