Advanced Racemization Technology For High-Purity Local Anesthetic Intermediates And Commercial Scale-Up
Advanced Racemization Technology For High-Purity Local Anesthetic Intermediates And Commercial Scale-Up
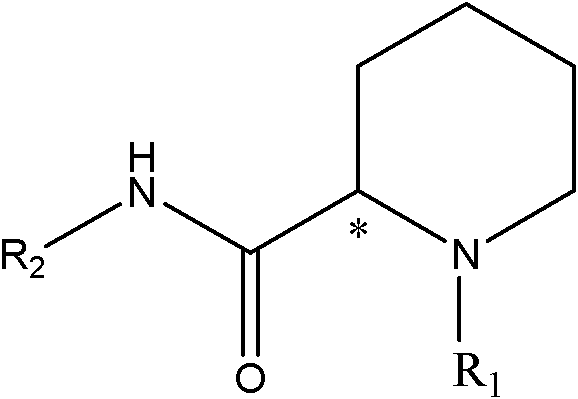
The pharmaceutical industry continuously demands more efficient pathways for producing single-enantiomer drugs, particularly for local anesthetics where the S-enantiomer exhibits superior safety profiles. Patent CN102093284A introduces a groundbreaking method for enriching piperidine-2-carboxanilide optically active compounds, specifically targeting key intermediates for ropivacaine and levobupivacaine. This technology addresses the critical bottleneck of low yield in traditional chiral resolution by implementing a robust racemization cycle that recycles the unwanted R-enantiomer. By shifting the paradigm from simple separation to dynamic kinetic resolution, this process offers a sustainable route for manufacturing high-purity pharmaceutical intermediates. The strategic integration of radical initiators and mercaptans allows for precise control over stereochemistry without compromising the integrity of the sensitive amide backbone. For global procurement teams, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at optimized costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer local anesthetics has been plagued by the inherent 50% yield limit of classical resolution techniques, where the undesired enantiomer is often discarded as waste. Conventional racemization methods typically rely on harsh acidic conditions at elevated temperatures exceeding 130°C, which poses severe risks of hydrolyzing the critical amide bond within the piperidine ring. This thermal degradation not only reduces the overall yield but also generates complex impurity profiles that are difficult and costly to remove during downstream purification. Furthermore, the high energy consumption associated with maintaining such extreme temperatures contradicts modern green chemistry principles and increases the carbon footprint of API manufacturing. The operational complexity of these legacy methods, often requiring inert gas protection and extended reaction times, further exacerbates production costs and limits scalability for commercial operations.
The Novel Approach
The innovative methodology described in the patent data utilizes a mild radical-mediated racemization process that operates effectively at temperatures between 70°C and 110°C, drastically reducing thermal stress on the molecule. By employing specific initiators such as diisopropyl azodicarboxylate alongside mercaptan stabilizers, the system achieves rapid equilibration of stereocenters without the need for corrosive strong acids. This approach not only preserves the structural integrity of the intermediate but also significantly shortens the reaction time to a window of 2 to 10 hours, enhancing throughput capacity. The ability to recycle the mother liquor containing the R-enantiomer back into the racemization loop transforms the theoretical yield limit, allowing for near-quantitative conversion of raw materials into the desired S-enantiomer. This closed-loop system minimizes waste generation and aligns perfectly with the cost reduction in API manufacturing goals of modern pharmaceutical enterprises.
Mechanistic Insights into Radical-Mediated Racemization and Chiral Resolution
The core of this technological advancement lies in the precise manipulation of radical chemistry to invert the configuration at the chiral center of the piperidine ring. The reaction mechanism involves the generation of carbon-centered radicals via the homolytic cleavage of the initiator, which is then facilitated by the hydrogen atom transfer from the mercaptan species. This reversible radical formation allows the stereocenter to flatten temporarily, enabling the reformation of the bond in either the R or S configuration with equal probability, thus achieving racemization. The careful selection of solvents like toluene or xylene ensures that the radical species remain stable and reactive throughout the process, preventing premature termination or side reactions. Understanding this mechanism is crucial for R&D directors aiming to replicate or scale this process, as it highlights the importance of maintaining strict stoichiometric ratios between the substrate, initiator, and mercaptan. The robustness of this radical pathway ensures that even sterically hindered intermediates can be efficiently racemized, broadening the scope of applicable substrates beyond simple analogs.
Following the racemization step, the separation of the desired S-enantiomer is achieved through diastereomeric salt formation using L-(-)-dibenzoyl tartaric acid (DBTA) as the resolving agent. The addition of ketone solvents such as acetone to the alcoholic solution induces selective precipitation of the S-type DBTA salt, leveraging the differences in solubility between the diastereomers. This crystallization step is critical for achieving high optical purity, often reaching an ee value of 100% after repeated recrystallization cycles. The mother liquor, which remains rich in the R-enantiomer, is not discarded but is instead subjected to alkalization and fed back into the racemization reactor. This iterative process ensures that the impurity profile remains controlled while maximizing the utilization of the chiral pool. For quality control teams, this mechanism provides a clear pathway to consistent high-purity local anesthetic intermediates, ensuring that the final API meets stringent regulatory specifications for enantiomeric excess.
How to Synthesize Pipecoloxylidide Efficiently
The synthesis of pipecoloxylidide and related intermediates using this patented method requires careful attention to the addition sequence of reagents and temperature control to ensure optimal radical generation. The process begins with the dissolution of the R-type starting material in an inert solvent, followed by the graded addition of the initiator to maintain a steady concentration of radical species throughout the reaction period. Detailed standardized synthesis steps see the guide below.
- Mix R-type ropivacaine or intermediates with inert solvent, initiator, and mercaptan, then heat to 70-110°C for 2-10 hours to achieve racemization.
- Perform acid-base extraction to isolate the racemic solid, then react with L-(-)-dibenzoyl tartaric acid (DBTA) in an alcoholic solution to precipitate the S-type enantiomer salt.
- Recycle the mother liquor containing R-type enantiomers by alkalization and repeat the racemization process to maximize overall yield and minimize waste.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers transformative benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for chiral intermediates. The ability to recycle the unwanted enantiomer fundamentally changes the economics of production, effectively doubling the yield from the same amount of starting material compared to traditional resolution methods. This dramatic improvement in atom economy translates directly into substantial cost savings, as the raw material cost per kilogram of the final product is significantly reduced. Furthermore, the lower operating temperatures and shorter reaction times reduce the energy load on manufacturing facilities, contributing to lower utility costs and a smaller environmental footprint. These efficiencies make the supply chain more resilient against fluctuations in raw material pricing, ensuring a stable and cost-effective supply of critical pharmaceutical building blocks for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of high-temperature acid hydrolysis steps removes the need for specialized corrosion-resistant equipment and reduces the consumption of expensive reagents. By recycling the R-enantiomer through the racemization loop, the process minimizes raw material waste, leading to a drastic simplification of the cost structure. The reduced formation of by-products also lowers the burden on purification systems, decreasing the consumption of solvents and chromatography media. These factors combine to deliver significant economic advantages without compromising the quality or purity of the final intermediate product.
- Enhanced Supply Chain Reliability: The use of common, commercially available solvents and reagents ensures that the supply chain is not vulnerable to shortages of exotic or specialized chemicals. The robustness of the radical racemization process allows for flexible manufacturing schedules, as the reaction times are predictable and easily scalable from pilot to commercial batches. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond quickly to market demands and regulatory changes. The consistent quality of the output ensures that downstream API synthesis proceeds without delays caused by out-of-specification starting materials.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing standard unit operations such as extraction, crystallization, and filtration that are easily implemented in existing facilities. The reduction in waste generation and the recycling of mother liquors align with strict environmental regulations, minimizing the cost and complexity of waste treatment. This environmental compliance is a key factor for multinational corporations seeking sustainable partners for the commercial scale-up of complex chiral compounds. The method's simplicity also reduces the risk of operational errors, ensuring safe and efficient production at multi-ton scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this racemization technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production lines. The information provided here serves as a foundational reference for further discussions on process optimization and technology transfer.
Q: How does this racemization method improve yield compared to traditional resolution?
A: Traditional resolution limits theoretical yield to 50%, whereas this method recycles the unwanted R-enantiomer through radical racemization, allowing for a theoretical yield approaching 100% of the desired S-enantiomer over multiple cycles.
Q: What are the specific temperature advantages of this process?
A: The process operates at 70-110°C, significantly lower than the 130°C required by conventional acid-catalyzed methods, reducing energy consumption and minimizing the risk of amide bond hydrolysis.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of common solvents like toluene and ethyl acetate, combined with simplified operational steps and reduced reaction times, makes this method highly scalable and compliant with industrial safety and environmental standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ropivacaine Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chiral technology application, leveraging advanced methodologies like the one described in CN102093284A to deliver superior value to our global partners. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can seamlessly transition this innovative racemization process from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ropivacaine or levobupivacaine intermediate meets the highest international standards. Our commitment to technical excellence allows us to offer customized solutions that address the specific needs of R&D and production teams, ensuring a smooth and efficient supply of critical materials.
We invite you to collaborate with us to explore how this technology can optimize your current manufacturing processes and reduce overall production costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your supply chain strategy. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable partner dedicated to driving innovation and efficiency in the pharmaceutical intermediate market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
