Advanced One-Step Synthesis of MAQ: Technical Breakthroughs for Commercial Scale-Up
Advanced One-Step Synthesis of MAQ: Technical Breakthroughs for Commercial Scale-Up
The global demand for high-efficiency, low-toxicity aryloxy propionic acid herbicides has necessitated a rigorous evolution in the synthesis of their key chiral intermediates. Patent CN112694403B introduces a transformative methodology for preparing (R)-(+)-2-(4-hydroxyphenoxy) methyl propionate, widely known in the industry as MAQ (CAS 96562-58-2). This compound serves as a critical building block for prominent herbicides such as diclofop-methyl and clodinafop-propargyl, which are essential for selective weed control in agriculture. The disclosed technology represents a paradigm shift from multi-step, equilibrium-limited processes to a streamlined, one-step condensation reaction. By leveraging chiral halopropionates and optimizing solvent systems, this invention addresses long-standing challenges regarding yield, optical purity, and environmental safety, positioning it as a cornerstone technology for any reliable agrochemical intermediate supplier aiming to enhance their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of MAQ has been plagued by inefficient synthetic routes that compromise both economic viability and product quality. Traditional methods, such as those described in earlier patent literature, often rely on the esterification of R-(+)-2-(4-hydroxyphenoxy) propionic acid with methanol. This approach is fundamentally flawed because esterification is a reversible reaction that generates water as a byproduct, necessitating complex azeotropic distillation procedures using solvents like toluene to continuously remove water and drive the equilibrium forward. Furthermore, alternative routes utilizing aqueous alkaline conditions frequently suffer from the hydrolysis of the ester bond, leading to the formation of unwanted carboxylic acid impurities and drastically reducing the overall yield. Other methods have employed highly toxic reagents like dimethyl sulfate, which poses severe safety hazards, generates difficult-to-treat acidic wastewater, and causes significant corrosion to reactor equipment, thereby inflating maintenance costs and operational risks for manufacturers.
The Novel Approach
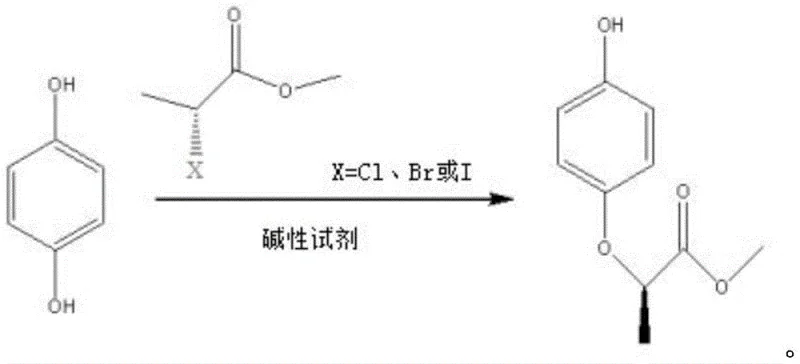
In stark contrast to these cumbersome legacy processes, the novel approach detailed in CN112694403B utilizes a direct nucleophilic substitution strategy that bypasses the need for pre-synthesized acids or toxic methylating agents. This method employs hydroquinone and chiral (S)-(-)-2-methyl halopropionate (where the halogen is chlorine, bromine, or iodine) as the primary starting materials. By conducting the reaction in a polar aprotic solvent system with an appropriate alkaline reagent, the process achieves a direct etherification in a single pot. This innovation not only eliminates the thermodynamic limitations of esterification but also avoids the hydrolysis pitfalls of aqueous systems. The result is a robust, high-yielding synthesis that delivers MAQ with exceptional optical purity (ee > 99%) and chemical purity (> 99.5%), effectively resolving the purity bottlenecks that have historically constrained the supply chain for high-performance herbicide intermediates.
Mechanistic Insights into Chiral SN2 Etherification
The core of this technological advancement lies in the precise control of stereochemistry through a bimolecular nucleophilic substitution (SN2) mechanism. In this reaction, the phenoxide anion, generated in situ by the deprotonation of hydroquinone by the alkaline reagent, acts as a potent nucleophile. It attacks the chiral carbon of the (S)-(-)-2-methyl halopropionate from the side opposite to the leaving halogen group. This backside attack forces a complete inversion of configuration, known as Walden inversion, transforming the (S)-configured reactant directly into the desired (R)-configured MAQ product. The choice of a polar aprotic solvent, such as N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), is critical; these solvents effectively solvate the cationic counter-ions of the base without heavily solvating the nucleophilic phenoxide anion, thereby maximizing its reactivity and ensuring the reaction proceeds rapidly at moderate temperatures of 70-100°C.
Furthermore, the mechanistic pathway inherently suppresses the formation of racemic impurities that often plague acid-catalyzed or high-temperature processes. By maintaining strict control over the reaction temperature and utilizing non-protic media, the integrity of the chiral center is preserved throughout the transformation. The subsequent workup involves a sophisticated purification strategy where the reaction mixture is cooled to precipitate inorganic salts, which are removed via filtration. The remaining liquid phase undergoes a two-stage reduced pressure distillation. The first stage removes the high-boiling polar solvent at temperatures below 140°C under vacuum (< 500 Pa), while the second stage isolates the target MAQ fraction at 150-160°C. This thermal separation is highly effective because the absence of water and acidic byproducts prevents thermal degradation or racemization during distillation, ensuring the final product meets the stringent specifications required for downstream herbicide synthesis.
How to Synthesize (R)-(+)-2-(4-hydroxyphenoxy) methyl propionate Efficiently
The operational protocol for this synthesis is designed for reproducibility and scalability, making it an ideal candidate for technology transfer into commercial manufacturing facilities. The process begins with the preparation of a homogeneous reaction mixture containing hydroquinone, a stoichiometric amount of an alkaline agent like potassium carbonate or cesium carbonate, and a polar aprotic solvent. This mixture is heated to an initial temperature of approximately 70-80°C to ensure complete dissolution and activation of the nucleophile. The chiral halopropionate is then added dropwise at a controlled rate to manage the exotherm, keeping the internal temperature within the optimal window of 70-100°C. Detailed standardized operating procedures regarding specific molar ratios, agitation speeds, and distillation parameters are essential for maintaining batch-to-batch consistency.
- Mix hydroquinone, an alkaline reagent such as potassium carbonate, and a polar aprotic solvent like DMF, then heat the mixture to initiate the reaction environment.
- Dropwise add (S)-(-)-2-methyl halopropionate while maintaining the system temperature between 70-100°C to control the exothermic condensation reaction.
- Upon completion, cool the system, filter off solids, and perform sequential reduced pressure distillation to isolate the high-purity MAQ fraction at 150-160°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that extend far beyond simple chemical yield improvements. The shift from multi-step, equilibrium-driven processes to a direct one-step condensation fundamentally alters the cost structure of MAQ manufacturing. By eliminating the need for expensive pre-formed chiral acids and toxic methylating agents, the raw material costs are significantly reduced. Moreover, the simplification of the process flow—removing the need for azeotropic water removal or complex neutralization steps—drastically reduces utility consumption and labor hours per kilogram of product. This efficiency translates directly into a more competitive pricing structure for the final intermediate, allowing downstream herbicide formulators to optimize their own cost bases without sacrificing quality.
- Cost Reduction in Manufacturing: The elimination of toxic reagents like dimethyl sulfate removes the substantial costs associated with specialized corrosion-resistant equipment and hazardous waste disposal. Additionally, the high conversion rate minimizes raw material waste, ensuring that every kilogram of hydroquinone and chiral halide contributes maximally to the final output, thereby driving down the unit cost of production significantly.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydroquinone and common polar solvents ensures a stable and resilient supply chain, free from the bottlenecks often associated with specialty chiral acids. The robustness of the reaction conditions allows for flexible scheduling and rapid scale-up, reducing lead times for high-purity agrochemical intermediates and ensuring consistent availability for global herbicide production lines.
- Scalability and Environmental Compliance: The process generates minimal wastewater and avoids the release of volatile organic compounds associated with azeotropic distillation, aligning perfectly with increasingly stringent global environmental regulations. The simplicity of the workup, involving basic filtration and distillation, facilitates easy scale-up from pilot plants to multi-tonne commercial reactors without the need for complex engineering modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy techniques in terms of safety, efficiency, and product quality. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does this new method improve upon traditional esterification routes for MAQ?
A: Unlike traditional esterification which requires water removal to drive equilibrium, this method utilizes a direct SN2 etherification that proceeds to completion without reversible side reactions, significantly simplifying the workflow.
Q: What ensures the high optical purity (ee > 99%) in the final product?
A: The use of chiral (S)-halopropionates in a strict SN2 mechanism guarantees a complete Walden inversion to the (R)-configuration, avoiding the racemization risks associated with acidic or high-temperature conditions in older methods.
Q: Is this process scalable for industrial production of agrochemical intermediates?
A: Yes, the process eliminates toxic reagents like dimethyl sulfate and uses standard polar aprotic solvents, making it highly suitable for safe, large-scale commercial manufacturing with simplified waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable MAQ Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN112694403B are fully realized in practical application. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of MAQ delivered meets the exacting standards required for the synthesis of next-generation herbicides. We are committed to bridging the gap between innovative patent chemistry and reliable industrial supply.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this superior method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and sustainability in your agrochemical manufacturing operations.
