Advanced Manufacturing of 2-(4-Hydroxyphenoxy)propionic Acid for High-Efficiency Herbicide Production
Introduction to Patent CN102775297A
The global demand for high-efficiency phenoxy herbicides, such as quizalofop-ethyl and diclofop-methyl, necessitates a robust and economically viable supply chain for their key precursors. Patent CN102775297A introduces a transformative manufacturing methodology for 2-(4-hydroxyphenoxy)propionic acid, often referred to as MAQ-acid, which serves as a critical building block in modern agrochemistry. This intellectual property outlines a sophisticated aqueous-phase synthesis that fundamentally shifts away from the hazardous and cost-prohibitive protocols of the past. By leveraging a strategic combination of S-methyl 2-chloropropionate and hydroquinone under controlled alkaline conditions, the invention achieves a delicate balance between reaction kinetics and selectivity. For R&D directors and procurement strategists, this patent represents a pivotal opportunity to optimize production costs while adhering to increasingly stringent environmental regulations regarding solvent usage and toxic reagent handling.
The significance of this technology extends beyond mere chemical synthesis; it addresses the core pain points of industrial scalability and waste management. Traditional routes often suffer from low atom economy and the generation of difficult-to-treat organic waste streams. In contrast, the disclosed method utilizes a water-based system that simplifies downstream processing and facilitates the recovery of valuable starting materials. The process is designed to operate at moderate temperatures, specifically between 70°C and 80°C, which reduces energy consumption compared to high-temperature reflux methods. Furthermore, the integration of a precise pH control strategy allows for the sequential isolation of intermediates and the recycling of unreacted hydroquinone, thereby enhancing the overall sustainability profile of the manufacturing operation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(4-hydroxyphenoxy)propionic acid has been plagued by significant technical and economic hurdles that hinder large-scale adoption. One prevalent conventional method relies on the condensation of alpha-brominated propionic ester with hydroquinone in dimethyl sulfoxide (DMSO) under alkaline conditions. While DMSO acts as a powerful polar aprotic solvent that accelerates nucleophilic substitution, it is notoriously difficult to remove completely from the final product and poses substantial challenges in wastewater treatment due to its high boiling point and stability. Additionally, the use of alpha-brominated esters introduces a considerable cost burden, as bromine-containing reagents are inherently more expensive than their chlorine counterparts. Another historical approach involves the use of diazomethane for esterification, a reagent known for its extreme toxicity and explosive potential, which necessitates specialized equipment and rigorous safety protocols that drive up capital expenditure.
Beyond safety and cost, selectivity remains a persistent issue in traditional syntheses. Hydroquinone possesses two phenolic hydroxyl groups that are chemically equivalent, making it prone to double substitution to form the undesired di-ether byproduct. To mitigate this, older methods often require a large excess of hydroquinone, sometimes in molar ratios as high as 2.5:1 relative to the halogenated ester. This stoichiometric imbalance leads to massive amounts of unreacted raw material that must be separated and disposed of or recycled through energy-intensive processes. The combination of expensive solvents, hazardous reagents, and poor atom economy results in a manufacturing process that is both financially inefficient and environmentally unsustainable, creating a strong market pull for the innovative solution presented in the patent data.
The Novel Approach
The novel approach detailed in the patent data circumvents these legacy issues by employing S-methyl 2-chloropropionate as the alkylating agent in conjunction with an aqueous sodium hydroxide system. This substitution of bromine for chlorine immediately lowers the raw material cost profile, as chloro-esters are commodity chemicals with stable supply chains. The reaction mechanism initiates with the in situ hydrolysis of the ester to form the sodium salt of 2-chloropropionic acid, which then undergoes nucleophilic attack on the hydroquinone nucleus. 
Crucially, the process avoids the use of DMSO, opting instead for a heterogeneous or semi-homogeneous aqueous environment that simplifies product isolation. The elimination of high-boiling polar solvents means that the final product can be precipitated directly via acidification, bypassing complex distillation or chromatographic purification steps.
Furthermore, the new method introduces a clever two-stage acidification protocol that maximizes yield and purity. After the initial coupling reaction, the mixture is treated with hydrochloric acid to adjust the pH to a weakly acidic range of 5 to 6. At this specific pH, any unreacted disodium hydroquinone is protonated back to neutral hydroquinone, which can then be extracted using an organic solvent like 4-methyl-2-pentanone. 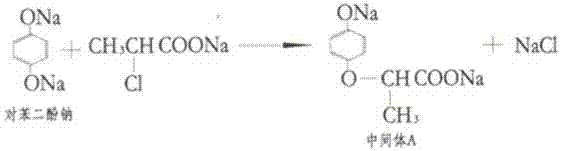
This recovery step not only purifies the aqueous phase containing the desired intermediate but also allows the expensive hydroquinone to be recycled back into the reactor. Finally, the aqueous phase is further acidified to pH 1 to 2, causing the target 2-(4-hydroxyphenoxy)propionic acid to precipitate out as a solid. This elegant sequence of pH swings ensures high purity without the need for extensive organic solvent washing, representing a paradigm shift in process efficiency.
Mechanistic Insights into Selective Nucleophilic Substitution
From a mechanistic perspective, the success of this synthesis hinges on the precise control of nucleophilicity and electrophilicity within the reaction matrix. The reaction begins with the deprotonation of hydroquinone by sodium hydroxide to form the disodium salt, which is a potent nucleophile. Simultaneously, the S-methyl 2-chloropropionate undergoes saponification to yield the sodium carboxylate, although the ester functionality can also participate directly in the substitution depending on the specific conditions. The core transformation is an SN2-type nucleophilic substitution where the phenoxide oxygen attacks the alpha-carbon of the chloropropionate moiety, displacing the chloride ion. However, the presence of two nucleophilic sites on the hydroquinone ring creates a competitive pathway leading to the di-substituted byproduct, Intermediate B. 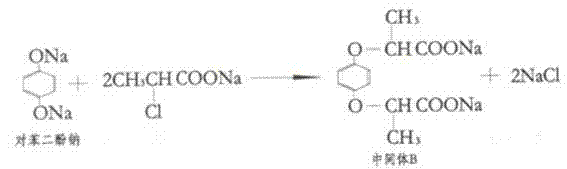
Understanding the kinetics of this competition is vital for R&D teams aiming to replicate or optimize the process at scale.
To suppress the formation of the di-substituted impurity, the process relies on kinetic control and stoichiometric balancing rather than just thermodynamic stability. By carefully managing the addition rate of the base and maintaining the reaction temperature between 70°C and 80°C for a duration of 8 hours, the system favors the mono-substitution product. The subsequent workup procedure acts as a chemical filter; the differential pKa values of the phenolic hydroxyl group versus the carboxylic acid group allow for selective protonation. When the pH is raised to 5-6, the carboxylic acid remains deprotonated (as a water-soluble salt), while the phenolic oxygen of any unreacted hydroquinone becomes protonated and extractable. This mechanistic nuance ensures that the final acidification step yields a product with minimal phenolic contamination, addressing a common purity bottleneck in the production of phenoxy herbicide intermediates.
How to Synthesize 2-(4-Hydroxyphenoxy)propionic Acid Efficiently
Implementing this synthesis requires strict adherence to the sequential addition of reagents and precise pH monitoring to ensure optimal yield and safety. The process begins with the charging of hydroquinone and S-methyl 2-chloropropionate into the reactor, followed by the controlled addition of sodium hydroxide to initiate the reaction cascade. Maintaining the temperature profile is critical, as deviations can lead to increased hydrolysis of the ester or accelerated formation of the di-ether byproduct. Following the reaction period, the careful addition of hydrochloric acid allows for the separation of the organic and aqueous phases, facilitating the recovery of raw materials. For a comprehensive, step-by-step operational guide including specific flow rates and quality control checkpoints, please refer to the standardized protocol below.
- Hydrolyze S-methyl 2-chloropropionate with sodium hydroxide to generate the reactive sodium salt in situ.
- React the sodium salt with hydroquinone under controlled alkaline conditions (70-80°C) to form the mono-substituted intermediate while minimizing di-substitution.
- Recover unreacted hydroquinone via pH adjustment and solvent extraction, then acidify the aqueous phase to precipitate the final high-purity acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend well beyond the laboratory bench. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing exotic and hazardous reagents like diazomethane and alpha-bromo esters with commodity chemicals like sodium hydroxide and chloro-esters, the supply chain becomes significantly more resilient and less prone to volatility. The elimination of DMSO as a solvent removes a major bottleneck in waste disposal, as aqueous waste streams are generally cheaper and easier to treat than those contaminated with high-boiling organic solvents. This reduction in environmental compliance complexity translates directly into lower operational expenditures and reduced risk of regulatory shutdowns.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the inherent cheapness of chlorine-based reagents compared to bromine-based alternatives, alongside the ability to recycle unreacted hydroquinone. By recovering and reusing the starting phenol through the pH-swing extraction technique, the effective consumption of raw materials per kilogram of product is significantly lowered. Furthermore, the avoidance of expensive solvent recovery systems for DMSO reduces both capital investment in distillation columns and the energy costs associated with their operation. These factors combine to create a manufacturing cost structure that is highly competitive in the global agrochemical market.
- Enhanced Supply Chain Reliability: Relying on widely available bulk chemicals such as hydroquinone, caustic soda, and hydrochloric acid ensures that production is not held hostage by the supply constraints of niche fine chemicals. The robustness of the aqueous chemistry means that the process is less sensitive to minor fluctuations in reagent quality, allowing for greater flexibility in sourcing. This stability is crucial for long-term supply agreements with major herbicide formulators who require consistent delivery schedules and uninterrupted production runs to meet seasonal agricultural demand.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which relies on precipitation and filtration rather than complex chromatography or multi-stage extractions, makes this process ideally suited for tonnage-scale production. The reduced generation of hazardous organic waste aligns with modern green chemistry principles, facilitating easier permitting and community acceptance of manufacturing facilities. As environmental regulations tighten globally, having a process that minimizes toxic effluent provides a distinct competitive advantage and future-proofs the manufacturing asset against stricter compliance regimes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific operational parameters and beneficial effects outlined in the patent documentation to assist decision-makers in evaluating feasibility. Understanding these nuances is essential for assessing the fit of this technology within existing manufacturing infrastructure.
Q: How does this process improve selectivity compared to traditional methods?
A: By utilizing a controlled stoichiometric ratio and a specific two-stage pH adjustment, the process effectively suppresses the formation of the di-substituted byproduct (Intermediate B), ensuring higher yield of the desired mono-ether.
Q: What are the primary cost drivers eliminated in this new method?
A: The method eliminates the need for expensive solvents like dimethyl sulfoxide (DMSO) and hazardous reagents such as diazomethane, replacing them with common aqueous sodium hydroxide and safer chloro-esters.
Q: Is the unreacted raw material recoverable?
A: Yes, the process includes a dedicated recovery step where unreacted hydroquinone is regenerated from its sodium salt via weak acidification and extracted with 4-methyl-2-pentanone for reuse, significantly reducing raw material waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Hydroxyphenoxy)propionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of 2-(4-hydroxyphenoxy)propionic acid meets the exacting standards required for downstream herbicide synthesis. Our commitment to quality assurance ensures that your final agrochemical products perform consistently in the field.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us help you secure a sustainable and cost-effective supply of this critical intermediate today.
