Advanced Synthetic Route for Ulipristal Acetate Intermediate: Enhancing Purity and Scalability
Advanced Synthetic Route for Ulipristal Acetate Intermediate: Enhancing Purity and Scalability
The pharmaceutical landscape for emergency contraception and progesterone receptor modulation has been significantly shaped by the introduction of Ulipristal Acetate, commercially known as Ella or EllaOne. As demand for this critical active pharmaceutical ingredient (API) continues to grow globally, the efficiency and robustness of its supply chain become paramount for multinational healthcare corporations. Patent CN103588845A discloses a groundbreaking synthetic methodology for producing the key intermediate of Ulipristal Acetate, specifically addressing the longstanding bottlenecks associated with stereochemical control and process safety. This innovation represents a pivotal shift from laboratory-scale curiosity to industrial viability, offering a pathway that drastically simplifies the production of complex steroid scaffolds. By leveraging a sequence of 17-position cyanation, strategic silyl protection, and precise Grignard addition, the disclosed method achieves exceptional purity profiles without the need for cumbersome purification techniques. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent regulatory standards while optimizing cost structures.
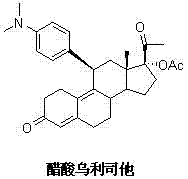
The structural complexity of Ulipristal Acetate, characterized by its 11β-(4-N,N-dimethylaminophenyl) substitution and specific diene system, necessitates a synthesis route that maintains rigorous stereochemical integrity throughout the transformation. The key intermediate, often referred to as Formula I in the patent literature, serves as the foundational building block upon which the final biological activity depends. Any deviation in the stereochemistry at the C17 position or instability in the C3 ketone protection can lead to inactive impurities that are difficult to remove in later stages. The novelty of CN103588845A lies not just in the chemical transformations themselves, but in the orchestration of reaction conditions that favor the desired isomer while suppressing degradation pathways. This level of control is what differentiates a viable commercial process from an academic exercise, ensuring that the resulting high-purity pharmaceutical intermediates are consistent batch after batch, a requirement that is non-negotiable for FDA and EMA compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
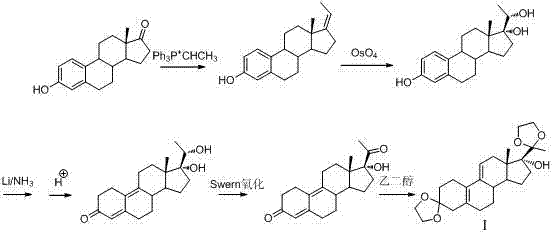
Historically, the synthesis of progesterone receptor modulators has been plagued by reliance on hazardous reagents and inefficient purification protocols that hinder scalability. Early methodologies, such as those described in U.S. Patent US4954490, relied heavily on osmium tetroxide (OsO4) for oxidative transformations, a reagent known for its extreme toxicity and high cost, posing significant environmental and safety liabilities for large-scale manufacturing facilities. Furthermore, alternative routes like those in U.S. Patent US5929262A utilized flammable metallic lithium and expensive chloromethyl chlorosilane, creating operational hazards that require specialized containment infrastructure and drive up capital expenditure. Other approaches, including Chinese Patent CN101466723A, introduced malodorous reagents such as thiophenyl chloride and trimethyl phosphite, which complicate waste management and worker safety protocols. Perhaps most critically, many of these legacy routes suffered from poor selectivity, often yielding complex mixtures that necessitated column chromatography for purification, a technique that is economically prohibitive and technically challenging to implement at the multi-ton scale required for global API supply.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the method disclosed in CN103588845A introduces a streamlined, high-yielding pathway that eliminates the need for toxic oxidants and hazardous metals. The core innovation involves a strategic protection-deprotection sequence that stabilizes the reactive centers of the steroid nucleus, allowing for clean transformations under mild conditions. By employing imidazole-mediated silylation, the process achieves near-quantitative conversion of the hydroxyl group, preventing the formation of unwanted isomers that typically plague carbonate-base mediated reactions. Subsequent Grignard addition is performed in a controlled manner to install the critical acetyl functionality with high stereoselectivity, followed by a robust ketalization step that secures the C3 position. This approach not only improves the overall yield but also simplifies the downstream processing, enabling purification through standard crystallization and extraction rather than chromatography. For supply chain leaders, this translates to a drastic reduction in cost reduction in pharmaceutical intermediates manufacturing, as the elimination of expensive reagents and complex purification steps directly lowers the cost of goods sold (COGS).
Mechanistic Insights into Imidazole-Mediated Silylation and Grignard Addition
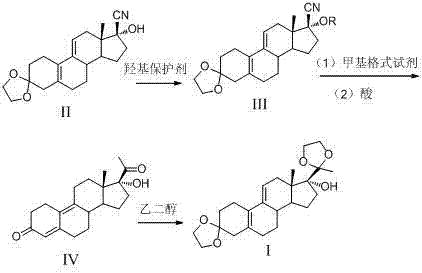
The mechanistic elegance of this synthesis lies in the precise manipulation of electronic and steric factors to guide the reaction trajectory. The initial step involves the conversion of the 17-hydroxyl group into a trimethylsilyl (TMS) ether using trimethylchlorosilane (TMSCl) in the presence of imidazole. Unlike weaker bases such as potassium carbonate, which were found to yield mixtures of cyanated products due to incomplete activation or competing side reactions, imidazole acts as a potent nucleophilic catalyst that facilitates the rapid and complete silylation. This protection is crucial because it prevents the hydroxyl group from interfering with subsequent nucleophilic attacks or undergoing elimination under the acidic conditions of later steps. The stability of the silyl ether allows the molecule to withstand the rigors of the Grignard reaction, where methylmagnesium bromide attacks the nitrile group at the C17 position. The steric bulk of the silyl group may also play a role in directing the approach of the Grignard reagent, favoring the formation of the desired 17-alpha configuration which is essential for the biological activity of the final Ulipristal Acetate molecule.
Following the Grignard addition, the intermediate undergoes acidic hydrolysis to reveal the ketone, which is subsequently converted to the enol ether or acetyl derivative depending on the specific workup conditions. The final step involves the protection of the C3 ketone as an ethylene ketal using ethylene glycol and triethyl orthoformate catalyzed by p-toluenesulfonic acid. This step is vital for masking the reactivity of the A-ring ketone, preventing it from participating in unwanted condensation reactions during the final coupling with the dimethylaminophenyl moiety in downstream synthesis. The choice of triethyl orthoformate as a water scavenger drives the equilibrium towards the ketal product, ensuring high conversion rates. The entire sequence is designed to minimize the exposure of sensitive functional groups to harsh conditions, thereby preserving the integrity of the steroid backbone. This meticulous attention to mechanistic detail ensures that the impurity profile of the intermediate remains within tight specifications, reducing the burden on quality control laboratories and facilitating faster regulatory approval for the final drug product.
How to Synthesize Ulipristal Acetate Intermediate Efficiently
The implementation of this synthetic route requires careful attention to reaction parameters such as temperature, stoichiometry, and solvent selection to maximize efficiency and safety. The process begins with the dissolution of the starting steroid in an anhydrous solvent like tetrahydrofuran, followed by the controlled addition of imidazole and TMSCl at low temperatures to manage exotherms. Subsequent steps involve the precise dosing of Grignard reagents and acid catalysts to ensure complete conversion without degrading the sensitive ketal protecting groups. While the general chemistry is robust, scaling these reactions from the gram scale to multi-kilogram batches requires optimized mixing and heat transfer capabilities to maintain the high yields reported in the patent. For technical teams looking to adopt this methodology, the detailed standardized operating procedures are critical for ensuring reproducibility and safety across different manufacturing sites.
- Perform selective silylation of the 17-hydroxyl group using TMSCl and imidazole to protect the functionality before nucleophilic attack.
- Execute stereoselective Grignard addition using methylmagnesium bromide followed by acidic hydrolysis to install the 17-alpha acetyl group.
- Finalize the synthesis by protecting the C3 ketone as an ethylene ketal using ethylene glycol and triethyl orthoformate under acidic catalysis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers transformative benefits for procurement managers and supply chain executives tasked with optimizing the sourcing of critical pharmaceutical ingredients. The primary advantage lies in the substantial simplification of the manufacturing process, which directly correlates to reduced operational expenditures and enhanced supply reliability. By eliminating the need for column chromatography, a resource-intensive and time-consuming purification method, manufacturers can significantly increase throughput and reduce cycle times. This efficiency gain allows for more flexible production scheduling and faster response to market demand fluctuations, which is crucial for emergency contraceptive products that may experience sudden spikes in usage. Furthermore, the replacement of toxic and hazardous reagents like osmium tetroxide and metallic lithium with safer, commodity chemicals reduces the regulatory burden associated with hazardous waste disposal and worker safety compliance, leading to long-term cost savings that extend beyond the immediate cost of raw materials.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and specialized reagents that characterized previous synthetic routes. The use of readily available reagents such as TMSCl, imidazole, and methylmagnesium bromide ensures a stable and predictable raw material supply chain, insulating manufacturers from price volatility associated with rare or specialty chemicals. Additionally, the high yields achieved at each step minimize material loss, meaning less starting material is required to produce the same amount of final intermediate, effectively lowering the unit cost of production. The avoidance of chromatographic purification further reduces costs by removing the need for large volumes of silica gel and organic solvents, as well as the associated labor and equipment maintenance costs. These cumulative savings contribute to a more competitive pricing structure for the final API, allowing pharmaceutical companies to improve their margins or pass savings on to healthcare providers.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness and simplicity of this synthetic method. The reliance on common, industrially produced chemicals reduces the risk of supply disruptions caused by the scarcity of exotic reagents. Moreover, the process tolerance to minor variations in reaction conditions makes it more forgiving in a large-scale manufacturing environment, reducing the likelihood of batch failures that can lead to costly delays. The ability to purify intermediates through crystallization rather than chromatography also simplifies the logistics of material handling and storage, as crystalline solids are generally more stable and easier to transport than oils or complex mixtures. This reliability ensures a continuous flow of high-quality intermediates to downstream API manufacturers, supporting uninterrupted production of the final drug product and safeguarding against stockouts in the global market.
- Scalability and Environmental Compliance: The environmental footprint of pharmaceutical manufacturing is an increasingly critical factor for corporate sustainability goals, and this route offers distinct advantages in terms of green chemistry principles. By avoiding toxic heavy metals and flammable alkali metals, the process generates less hazardous waste, simplifying effluent treatment and reducing the environmental liability of the manufacturing site. The high atom economy of the reactions, particularly the quantitative silylation and high-yield Grignard addition, ensures that a greater proportion of the input materials end up in the final product, minimizing waste generation. This alignment with environmental regulations facilitates smoother permitting processes for new manufacturing facilities and enhances the corporate social responsibility profile of the supply chain partners. The scalability of the process is further evidenced by the use of standard unit operations like extraction and crystallization, which are easily adapted from pilot plant to commercial scale without requiring bespoke engineering solutions.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis is vital for stakeholders evaluating the feasibility of integrating this intermediate into their supply chain. The following questions address common concerns regarding the practical implementation, safety, and quality implications of the patented method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a transparent view of the technology's capabilities and limitations. For procurement and technical teams, these answers serve as a foundation for risk assessment and vendor qualification discussions.
Q: Why is the imidazole-mediated silylation superior to carbonate bases in this synthesis?
A: Traditional carbonate bases often lead to mixture formation and lower yields during cyanation steps. The use of imidazole with TMSCl ensures quantitative conversion to the silyl ether, preventing side reactions and simplifying purification.
Q: How does this route address the toxicity issues of prior art methods?
A: Unlike earlier methods relying on toxic osmium tetroxide or flammable lithium metal, this novel pathway utilizes safer, industrially viable reagents like TMSCl and Grignard reagents, significantly reducing hazardous waste and safety risks.
Q: Is column chromatography required for purifying the intermediate?
A: No, a major advantage of this process is the elimination of column chromatography. The intermediates can be purified through standard extraction and crystallization techniques, which is critical for cost-effective large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ulipristal Acetate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Ulipristal Acetate depends on a partnership grounded in technical excellence and supply chain reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering stringent purity specifications that meet or exceed global regulatory requirements, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in steroid chemistry allows us to navigate the complexities of this synthesis with precision, guaranteeing a consistent supply of high-quality intermediates that empower your drug development and manufacturing timelines.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this methodology offers compared to your current supply sources. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing you to validate the quality and scalability of our offerings against your internal standards. Together, we can build a resilient supply chain that supports the global availability of this essential medication.
