Advanced Green Synthesis of 2-Methyl-1,4-Naphthoquinone for Commercial Vitamin K3 Production
The global demand for high-purity 2-methyl-1,4-naphthoquinone, a critical precursor for Vitamin K3 and essential feed additives, has necessitated a paradigm shift away from environmentally hazardous legacy processes. Patent CN111689844A discloses a groundbreaking three-step synthesis route that fundamentally restructures the supply chain logic for this key pharmaceutical intermediate. By utilizing o-cresol as the starting material and leveraging advanced microchannel reactor technology coupled with a novel DMSO-based catalytic oxidation system, this process addresses the twin challenges of environmental compliance and production scalability. Unlike traditional methods that rely on toxic chromium reagents or expensive meta-cresol derivatives, this innovation offers a streamlined pathway that significantly reduces the ecological footprint while maintaining rigorous quality standards suitable for commercial scale-up of complex quinones.
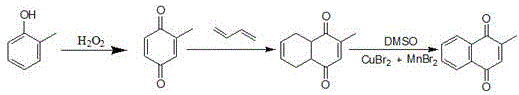
This technical breakthrough is particularly relevant for multinational corporations seeking a reliable pharmaceutical intermediates supplier capable of delivering consistent quality without the baggage of heavy metal contamination. The process begins with the continuous oxidation of o-cresol, proceeds through a Diels-Alder cycloaddition with butadiene, and culminates in a vacuum-assisted dehydrogenation. Each step has been meticulously optimized to maximize atom economy and minimize waste generation, positioning this technology as a cornerstone for sustainable cost reduction in vitamin K3 manufacturing. For procurement and R&D leaders, understanding the mechanistic advantages of this route is essential for evaluating long-term supply security and regulatory risk mitigation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-methyl-1,4-naphthoquinone has been plagued by significant technical and environmental bottlenecks. The most prevalent legacy method involves the oxidation of 2-methylnaphthalene using chromic anhydride or sodium dichromate in a glacial acetic acid medium. While chemically straightforward, this approach suffers from abysmal yields ranging typically between 30% and 50%, necessitating large volumes of raw material input for modest output. More critically, this process generates vast quantities of chromium-containing wastewater, which poses severe disposal challenges and escalating compliance costs under modern environmental regulations. Alternative routes utilizing m-cresol involve high-risk oxidation steps with poor selectivity and expensive starting materials, further exacerbating the economic inefficiency. These conventional pathways are increasingly untenable for modern supply chains that prioritize ESG (Environmental, Social, and Governance) criteria and operational safety.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111689844A introduces a robust alternative that circumvents these historical deficiencies by employing o-cresol, a readily available and cost-effective feedstock. The innovation lies not just in the choice of starting material, but in the integration of process intensification technologies. The initial oxidation step utilizes a microchannel reactor, which transforms a potentially hazardous batch operation into a safe, continuous flow process with superior thermal management. Furthermore, the final aromatization step replaces toxic chromium oxidants with a benign DMSO/copper-manganese bromide system. This shift eliminates the generation of heavy metal sludge entirely, simplifying downstream purification and waste treatment. The result is a process that is not only chemically superior with yields exceeding 90% in the final step but also economically advantageous due to reduced waste handling and raw material costs.
Mechanistic Insights into DMSO-Catalyzed Oxidative Dehydrogenation
The core chemical innovation of this process resides in the third step: the conversion of 2-methyl-1,4-tetrahydronaphthoquinone to the final aromatic product. This transformation is achieved through an oxidative dehydrogenation mechanism mediated by dimethyl sulfoxide (DMSO) in the presence of a dual-metal catalyst system comprising copper bromide and manganese bromide. Mechanistically, DMSO acts as both the solvent and the oxygen donor, facilitating the removal of hydrogen atoms from the tetrahydronaphthoquinone ring. The reaction produces water and dimethyl sulfide (DMS) as stoichiometric byproducts. In traditional batch setups, the accumulation of these byproducts can inhibit reaction progress or lead to side reactions. However, this patent specifies a critical operational parameter: the application of vacuum conditions ranging from -0.08MPa to -0.09MPa at temperatures between 85°C and 90°C.
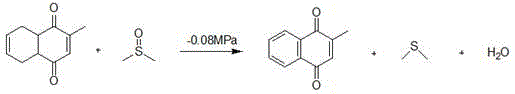
This vacuum application is not merely a procedural detail but a thermodynamic necessity that drives the equilibrium toward the desired product. By continuously stripping out the generated water and volatile dimethyl sulfide, the reaction is prevented from stalling, thereby achieving high conversion rates that are unattainable under atmospheric pressure. From an impurity control perspective, this mechanism is exceptionally clean. The absence of strong inorganic oxidants means there is no risk of over-oxidation or ring cleavage, which are common failure modes in chromium-based chemistry. The catalyst system, utilizing earth-abundant transition metals, ensures high selectivity, resulting in a crude product profile that requires minimal purification to meet high-purity 2-methyl-1,4-naphthoquinone specifications. This mechanistic elegance translates directly into operational reliability and consistent batch-to-batch quality.
How to Synthesize 2-Methyl-1,4-Naphthoquinone Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to fully realize its potential benefits. The process is divided into three distinct stages, each demanding specific engineering considerations regarding flow dynamics, pressure management, and thermal regulation. The transition from batch to continuous processing in the first step, followed by the pressurized cycloaddition and finally the vacuum-assisted oxidation, represents a sophisticated orchestration of unit operations.
- Oxidize o-cresol with hydrogen peroxide in a microchannel reactor using ethyl acetate as solvent to produce o-toluquinone.
- Perform a Diels-Alder addition reaction between o-toluquinone and 1,3-butadiene in a pressure reactor to form 2-methyl-1,4-tetrahydronaphthoquinone.
- Oxidize the tetrahydronaphthoquinone intermediate using DMSO and a Cu/Mn bromide catalyst system under vacuum (-0.08MPa) to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers tangible strategic advantages beyond mere chemical curiosity. The primary value driver is the substantial reduction in operational complexity and associated costs. By eliminating the need for chromium-based reagents, manufacturers avoid the高昂 costs associated with hazardous waste disposal, heavy metal testing, and regulatory reporting. This simplification of the waste stream directly contributes to cost reduction in vitamin K3 manufacturing, allowing for more competitive pricing structures in a margin-sensitive market. Furthermore, the use of o-cresol and butadiene ensures a stable supply of raw materials, as these are commodity chemicals with well-established global supply chains, unlike specialized oxidants that may be subject to supply volatility.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic chromium reagents removes a significant line item from the production budget. Additionally, the high yields achieved in the Diels-Alder step (approximately 98%) and the final oxidation step (over 90%) minimize raw material waste. The ability to recycle the aqueous catalyst phase in the first step further enhances atom economy. These factors combine to lower the overall cost of goods sold (COGS), providing a buffer against raw material price fluctuations and enabling more aggressive market positioning.
- Enhanced Supply Chain Reliability: The shift to continuous processing in the initial oxidation step significantly increases throughput capacity and reduces batch cycle times. Microchannel reactors allow for rapid scaling by numbering up rather than scaling up vessel size, which mitigates the technical risks often associated with plant expansion. This flexibility ensures that suppliers can respond quickly to surges in demand for pharmaceutical intermediates without the long lead times typically required for building new batch infrastructure. The robustness of the chemistry also reduces the likelihood of batch failures, ensuring consistent delivery schedules.
- Scalability and Environmental Compliance: As global regulations on heavy metal discharge tighten, processes that inherently avoid toxic pollutants future-proof the supply chain. This DMSO-based route generates benign byproducts that are easier to treat or recover, aligning with strict environmental standards in key markets like Europe and North America. The simplified downstream processing reduces the burden on effluent treatment plants, lowering capital expenditure for facility upgrades. This environmental compatibility facilitates smoother regulatory approvals and reduces the risk of production shutdowns due to compliance violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios.
Q: Why is the DMSO oxidation method superior to traditional chromium-based oxidation?
A: Traditional methods using chromic anhydride or sodium dichromate generate massive amounts of toxic chromium-containing wastewater, creating severe environmental compliance issues and high waste treatment costs. The patented DMSO oxidation method eliminates heavy metal pollutants entirely, producing only water and dimethyl sulfide as byproducts, which are easily removed under vacuum.
Q: What is the role of vacuum conditions in the final dehydrogenation step?
A: The oxidation of 2-methyl-1,4-tetrahydronaphthoquinone by DMSO generates water and dimethyl sulfide. Applying a vacuum of -0.08MPa to -0.09MPa continuously removes these byproducts from the reaction equilibrium, driving the reaction forward significantly and improving yield from typical low levels to over 90%.
Q: How does the use of a microchannel reactor benefit the initial oxidation step?
A: The oxidation of o-cresol with hydrogen peroxide is highly exothermic and poses safety risks in batch reactors. Microchannel technology provides superior heat transfer and mixing, allowing for precise temperature control (130-150°C), enhanced safety, and continuous production capability, which drastically improves efficiency compared to batch processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-1,4-Naphthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthesis routes is critical for the long-term viability of the fine chemical industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations like the o-cresol route are successfully translated into robust industrial realities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-methyl-1,4-naphthoquinone meets the exacting standards required for pharmaceutical and feed additive applications. Our commitment to process excellence allows us to offer products that are not only high in quality but also consistent in supply.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing technology can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this chromium-free process. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of our high-quality intermediates into your production workflow.
