Advanced Manufacturing of 2-Fluoro-5-Bromobenzonitrile: A Low-Toxicity Route for Global Pharma Supply Chains
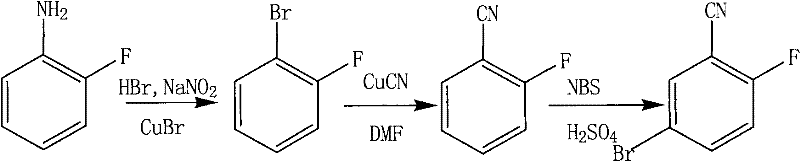
The pharmaceutical industry constantly seeks robust, scalable, and environmentally compliant routes for critical building blocks. Patent CN101898976A introduces a refined preparation method for 2-fluoro-5-bromobenzonitrile, a vital intermediate used in the synthesis of advanced therapeutic agents including kinase inhibitors and anti-inflammatory compounds. This technology utilizes o-fluoroaniline as the starting material, proceeding through a strategic three-step sequence: diazotization-bromination, cyanation, and selective bromination. Unlike traditional methods that often rely on highly hazardous reagents, this process emphasizes safety and environmental compatibility by substituting剧毒 alkali metal cyanides with cuprous cyanide and employing N-bromosuccinimide (NBS) for the final functionalization. The documented total yield reaches 39.7%, demonstrating a viable balance between efficiency and operational safety for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated benzonitriles has been plagued by significant safety and environmental challenges, particularly regarding the introduction of the cyano group. Conventional protocols frequently employ sodium cyanide (NaCN) or potassium cyanide (KCN) for nucleophilic substitution reactions. These reagents are acutely toxic, requiring stringent containment measures, specialized personal protective equipment, and complex wastewater treatment systems to neutralize free cyanide ions before discharge. Furthermore, the use of elemental bromine (Br2) in aromatic substitution often leads to poor regioselectivity and the generation of corrosive hydrogen bromide gas, necessitating expensive scrubbing systems. These factors collectively inflate the operational expenditure (OpEx) and introduce substantial supply chain risks related to regulatory compliance and worker safety.
The Novel Approach
The methodology outlined in CN101898976A addresses these痛点 by fundamentally altering the reagent profile. By utilizing cuprous cyanide (CuCN) in a polar aprotic solvent like DMF, the process mitigates the immediate toxicity risks associated with free cyanide ions, as the copper complex is more stable and manageable. Additionally, the replacement of elemental bromine with NBS in the final step allows for a much milder bromination environment. This shift not only improves the purity profile by minimizing over-bromination side products but also simplifies the work-up procedure. The result is a streamlined workflow that maintains high chemical efficiency while drastically lowering the barrier for safe industrial implementation, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to reduce liability.
Mechanistic Insights into CuCN-Mediated Cyanation and Regioselective Bromination
The core of this synthetic strategy lies in the efficient transformation of the aryl halide to the nitrile via a modified Rosenmund-von Braun reaction mechanism. In the second step, the 1-bromo-2-fluorobenzene intermediate reacts with CuCN in dimethylformamide (DMF) at elevated temperatures around 150°C. The mechanism involves the oxidative addition of the aryl bromide to the copper center, followed by reductive elimination to forge the carbon-nitrogen triple bond. The choice of DMF is critical here, as its high boiling point and polarity facilitate the solubility of the inorganic copper salt and stabilize the transition state, ensuring a consistent yield of approximately 60% for this specific transformation without the need for exotic ligands.
Following cyanation, the final step exploits the electronic properties of the cyano and fluoro groups to achieve precise regiocontrol. The electrophilic bromination using NBS in concentrated sulfuric acid targets the position para to the fluorine atom (which is ortho/para directing) and meta to the cyano group (which is meta directing). Since the 5-position satisfies both electronic directives, the reaction proceeds with high selectivity. The use of sulfuric acid as a solvent protonates the nitrile nitrogen slightly, deactivating the ring just enough to prevent poly-substitution while maintaining sufficient reactivity for the NBS electrophile. This mechanistic precision ensures that the impurity profile remains clean, reducing the burden on downstream purification units such as recrystallization or column chromatography.
How to Synthesize 2-Fluoro-5-Bromobenzonitrile Efficiently
Executing this synthesis requires careful control of temperature and stoichiometry across the three distinct stages to maximize the cumulative yield. The process begins with the diazotization of o-fluoroaniline in hydrobromic acid, followed by the immediate addition of cuprous bromide to secure the first bromine atom. Once the 1-bromo-2-fluorobenzene is isolated, it undergoes the high-temperature cyanation step, which is the most energy-intensive part of the sequence. Finally, the nitrile intermediate is subjected to controlled bromination. For detailed standard operating procedures, exact molar ratios, and specific work-up protocols derived directly from the patent examples, please refer to the technical guide below.
- Perform diazotization of o-fluoroaniline using HBr and NaNO2, followed by Sandmeyer bromination with CuBr to yield 1-bromo-2-fluorobenzene.
- Execute cyanation of the bromo-intermediate using cuprous cyanide (CuCN) in DMF at elevated temperatures (150°C) to form 2-fluorobenzonitrile.
- Conduct selective bromination of 2-fluorobenzonitrile using N-bromosuccinimide (NBS) in sulfuric acid to obtain the final 2-fluoro-5-bromobenzonitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented route offers tangible benefits beyond mere chemical yield. The primary advantage lies in the drastic reduction of hazardous material handling costs. By eliminating the need for bulk storage and transport of sodium cyanide, facilities can lower their insurance premiums and reduce the complexity of their environmental health and safety (EHS) audits. This translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the overhead associated with managing extreme hazards is significantly diminished. Furthermore, the use of commercially available and stable reagents like NBS ensures a more resilient supply chain, less susceptible to the regulatory fluctuations that often affect the availability of restricted toxic precursors.
- Cost Reduction in Manufacturing: The substitution of highly toxic cyanides with cuprous cyanide eliminates the need for specialized cyanide destruction units in wastewater treatment plants. This qualitative shift in process chemistry removes a major capital expenditure (CapEx) barrier for contract manufacturing organizations (CMOs). Additionally, the milder conditions of the NBS bromination step reduce energy consumption compared to harsher halogenation methods, leading to substantial cost savings in utility usage over long production campaigns.
- Enhanced Supply Chain Reliability: The reliance on o-fluoroaniline as a starting material leverages a widely available commodity chemical, ensuring consistent feedstock availability. Unlike processes dependent on custom-synthesized or highly regulated precursors, this route utilizes reagents that are standard in the fine chemical industry. This stability minimizes the risk of production stoppages due to raw material shortages, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like DMF and sulfuric acid which are easily recovered and recycled in industrial settings. The reduced toxicity profile facilitates easier regulatory approval for new manufacturing sites, allowing for rapid capacity expansion. The simplified purification steps, driven by the high selectivity of the NBS reaction, mean that less solvent is wasted on extensive chromatographic separations, aligning with modern green chemistry principles and reducing the overall environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-fluoro-5-bromobenzonitrile. These insights are derived from the specific experimental data and claims found in the underlying patent literature, providing clarity on yield expectations, safety protocols, and application suitability for potential partners evaluating this technology for their supply chains.
Q: Why is CuCN preferred over NaCN in this synthesis route?
A: While NaCN is highly toxic and poses severe safety risks, CuCN offers a significantly safer alternative for the Rosenmund-von Braun cyanation step, reducing hazardous waste disposal costs and improving workplace safety compliance without compromising the conversion to the nitrile group.
Q: What is the overall yield of the patented process?
A: According to patent CN101898976A, the optimized three-step process achieves a total yield of approximately 39.7%, with individual step yields ranging from 60% to over 80% depending on specific reaction conditions.
Q: How does the use of NBS improve the bromination step?
A: N-Bromosuccinimide (NBS) acts as a controlled source of bromine, allowing for milder reaction conditions compared to elemental bromine. This enhances regioselectivity for the 5-position and reduces the formation of poly-brominated byproducts, simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-5-Bromobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific thermal and safety requirements of the CuCN cyanation step, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to EHS standards, we guarantee a supply of high-purity intermediates that support your critical drug development timelines without compromise.
We invite you to leverage our technical expertise to optimize your sourcing strategy. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this low-toxicity synthesis can enhance your project's economic and operational viability.
