Advanced Manufacturing of 2-Fluoro-5-bromobenzonitrile: Technical Upgrades and Commercial Scale-Up Capabilities
Introduction to Advanced Synthetic Pathways for Fluorinated Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical intermediates. Patent CN101898976B introduces a refined preparation method for 2-fluoro-5-bromobenzonitrile, a pivotal building block in the synthesis of various bioactive compounds including anti-inflammatory agents, kinase inhibitors, and HIV therapeutics. This technology leverages a strategic three-step sequence starting from the readily available o-fluoroaniline, utilizing diazotization-bromination, cyanation, and selective bromination to achieve a total yield of 39.7%. The significance of this patent lies not only in its chemical efficiency but also in its deliberate shift towards greener chemistry principles, replacing hazardous reagents with safer alternatives without compromising output quality. For global procurement teams and R&D directors, understanding this pathway is essential for securing a stable supply of high-purity intermediates.
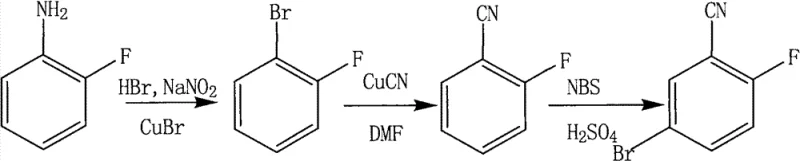
The core innovation involves the substitution of virulent alkali metal cyanides with cuprous cyanide and the use of N-bromosuccinimide (NBS) for the final halogenation step. This approach addresses long-standing concerns regarding operator safety and environmental impact, which are increasingly critical factors in modern chemical manufacturing compliance. By optimizing reaction conditions such as temperature control between 70-80°C for the initial step and utilizing dimethylformamide (DMF) as a solvent for the high-temperature cyanation at 150°C, the process ensures reproducibility. As a reliable pharmaceutical intermediate supplier, analyzing such patented methodologies allows us to align our production capabilities with the most advanced and sustainable industry standards, ensuring that our clients receive materials produced under the safest and most efficient protocols available.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated benzonitriles has been plagued by significant safety hazards and operational complexities. Traditional routes often rely on sodium cyanide or potassium cyanide for the introduction of the cyano group, reagents that are acutely toxic and require stringent containment measures, specialized waste treatment facilities, and extensive safety training for personnel. Furthermore, conventional bromination strategies frequently employ elemental bromine or hydrobromic acid under harsh conditions, which can lead to poor regioselectivity, generating difficult-to-remove isomeric impurities that compromise the purity profile required for pharmaceutical applications. These legacy methods also tend to generate substantial amounts of hazardous waste, increasing the environmental footprint and disposal costs for manufacturers. The reliance on such dangerous reagents creates bottlenecks in supply chains, as fewer facilities are licensed or equipped to handle them, leading to potential disruptions and inflated costs for downstream buyers seeking high-purity intermediates.
The Novel Approach
The methodology outlined in patent CN101898976B represents a paradigm shift by integrating safer reagents that maintain high reactivity while mitigating risk. By employing cuprous cyanide (CuCN) in the Rosenmund-von Braun reaction, the process eliminates the immediate lethality associated with free cyanide ions, thereby simplifying safety protocols and reducing the burden on effluent treatment systems. Additionally, the adoption of N-bromosuccinimide (NBS) or dibromohydantoin for the final bromination step offers a controlled release of bromine species, enhancing regioselectivity towards the 5-position relative to the fluoro and cyano groups. This precision minimizes the formation of by-products, streamlining the purification process and improving the overall mass balance of the synthesis. The use of concentrated sulfuric acid as a solvent in the final step further facilitates the reaction kinetics at mild temperatures (20-30°C), preserving the integrity of the sensitive nitrile functionality. This novel approach effectively balances chemical efficacy with operational safety, making it an optimal choice for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into CuCN-Mediated Cyanation and Electrophilic Bromination
The mechanistic foundation of this synthesis rests on well-established yet carefully optimized organometallic and electrophilic substitution principles. The initial conversion of o-fluoroaniline to 1-bromo-2-fluorobenzene proceeds via a classic Sandmeyer-type reaction mechanism. In this sequence, the amine group is first diazotized using sodium nitrite and hydrobromic acid to form a reactive diazonium salt intermediate. The subsequent addition of cuprous bromide (CuBr) facilitates the replacement of the diazonium group with a bromine atom through a radical or organocopper intermediate pathway, ensuring the retention of the ortho-fluorine substituent which is crucial for the biological activity of downstream derivatives. The careful control of pH and temperature during this exothermic process is vital to prevent the decomposition of the diazonium species, which could lead to phenol formation and reduced yields.
Following the initial bromination, the cyanation step utilizes a nucleophilic aromatic substitution mechanism mediated by copper. At elevated temperatures of 150°C in a polar aprotic solvent like DMF, the carbon-bromine bond undergoes oxidative addition to the copper center, followed by reductive elimination to forge the carbon-cyanide bond. This transition metal-catalyzed process is far superior to direct nucleophilic displacement for aryl halides, which typically requires extremely harsh conditions. Finally, the introduction of the second bromine atom is achieved through electrophilic aromatic substitution. The cyano group acts as a meta-directing deactivator, while the fluorine atom is an ortho/para director; however, the strong activation by the solvent system and the specific reactivity of NBS ensure substitution occurs selectively at the position para to the fluorine and meta to the nitrile group. This precise control over electronic effects is what enables the production of high-purity 2-fluoro-5-bromobenzonitrile with minimal isomeric contamination.
How to Synthesize 2-Fluoro-5-bromobenzonitrile Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process begins with the preparation of the diazonium salt under acidic conditions, followed by the critical copper-mediated transformations. Operators must monitor reaction temperatures closely, particularly during the cyanation phase where thermal stability is key. The workup procedures, involving steam distillation and recrystallization from ethanol, are designed to remove residual copper salts and organic impurities effectively. For detailed operational parameters, stoichiometry, and safety guidelines, please refer to the standardized protocol below which outlines the exact execution of this three-step transformation.
- Perform diazotization of o-fluoroaniline followed by bromination using HBr, NaNO2, and CuBr to obtain 1-bromo-2-fluorobenzene.
- Execute cyanation of 1-bromo-2-fluorobenzene using cuprous cyanide (CuCN) in DMF at 150°C to yield 2-fluorobenzonitrile.
- Conduct selective bromination of 2-fluorobenzonitrile using N-bromosuccinimide (NBS) in concentrated sulfuric acid to finalize 2-fluoro-5-bromobenzonitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of safety compliance and waste management. By eliminating the need for handling bulk quantities of sodium cyanide, facilities can reduce their insurance premiums, lower the costs associated with hazardous material storage, and minimize the risk of regulatory shutdowns due to safety violations. This inherent safety feature enhances supply chain reliability, as production is less susceptible to interruptions caused by safety audits or accidents. Furthermore, the use of commercially available and inexpensive starting materials like o-fluoroaniline ensures that raw material costs remain stable and predictable, shielding buyers from volatile market fluctuations often seen with exotic reagents.
- Cost Reduction in Manufacturing: The substitution of toxic reagents with safer alternatives like CuCN and NBS leads to significant operational savings. Without the need for specialized cyanide destruction units or complex scrubbing systems for bromine gas, the capital expenditure for setting up production lines is substantially lowered. Additionally, the improved selectivity of the NBS bromination reduces the load on purification columns and crystallization steps, resulting in higher throughput and lower energy consumption per kilogram of product. These efficiencies compound to offer a more competitive pricing structure for the final intermediate, allowing pharmaceutical companies to optimize their bill of materials without sacrificing quality.
- Enhanced Supply Chain Reliability: Sourcing intermediates produced via this method mitigates the risk of supply disruptions common with highly regulated toxic chemicals. Since the reagents used are standard industrial commodities rather than controlled substances, the supply chain is more resilient to logistical bottlenecks. The robustness of the three-step sequence, with yields consistently demonstrated across multiple embodiments in the patent, ensures that manufacturers can meet large-volume orders with confidence. This reliability is crucial for drug developers who require uninterrupted access to key building blocks to maintain their clinical trial timelines and commercial launch schedules.
- Scalability and Environmental Compliance: The mild reaction conditions, particularly the low-temperature bromination step, make this process highly amenable to scale-up from pilot plant to multi-ton commercial production. The reduced generation of hazardous by-products aligns with modern green chemistry initiatives, helping corporate clients meet their sustainability goals and carbon reduction targets. Facilities adopting this route can operate with a smaller environmental footprint, avoiding the heavy fines and reputational damage associated with non-compliance. This forward-thinking approach ensures long-term viability of the supply source, making it a sustainable partner for the future growth of the pharmaceutical pipeline.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-fluoro-5-bromobenzonitrile. These answers are derived directly from the technical specifications and advantageous effects described in the underlying patent literature, providing clarity on process safety, yield expectations, and purity standards. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader manufacturing strategies.
Q: How does this synthesis method improve safety compared to traditional cyanation routes?
A: This method replaces highly toxic sodium cyanide or potassium cyanide with cuprous cyanide (CuCN), significantly reducing toxicity risks and simplifying waste treatment protocols in industrial settings.
Q: What is the overall yield efficiency of the patented 2-fluoro-5-bromobenzonitrile process?
A: The patented three-step finishing synthesis achieves a total yield of 39.7%, demonstrating robust efficiency for commercial production while maintaining high purity standards through recrystallization.
Q: Why is NBS preferred over elemental bromine for the final bromination step?
A: N-Bromosuccinimide (NBS) offers milder reaction conditions and better selectivity compared to elemental bromine or HBr, reducing side reactions and environmental pollution while ensuring consistent product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-5-bromobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has thoroughly analyzed the synthetic pathways described in patent CN101898976B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering 2-fluoro-5-bromobenzonitrile that meets stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific requirements of fluorinated chemistry, ensuring that the delicate balance of reactivity and safety is maintained throughout the manufacturing process.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized production capabilities, we can help you reduce lead time for high-purity pharmaceutical intermediates and secure a stable supply for your R&D and commercial needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to excellence can support your project milestones.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
