Advanced Synthetic Route for Scutellarin: Enhancing Purity and Commercial Scalability
Introduction to Patent CN103374050A
The pharmaceutical industry continuously seeks robust and scalable methods for producing bioactive flavonoids, particularly Scutellarin (5,6,4'-trihydroxyflavone-7-O-D-glucuronic acid), a critical active ingredient derived from Erigeron breviscapus. Patent CN103374050A, published in late 2013, introduces a groundbreaking synthetic methodology that addresses the longstanding limitations of both natural extraction and previous chemical synthesis routes. This innovation is pivotal for manufacturers aiming to secure a reliable supply of high-purity pharmaceutical intermediates without relying on the fluctuating availability of botanical sources. The disclosed method employs a novel four-step sequence involving acylation, glycosylation, and a unique two-stage hydrolysis process, effectively bypassing the need for toxic heavy metal catalysts traditionally used in glycosylation reactions.
For R&D directors and procurement specialists, this patent represents a significant shift towards greener and more cost-effective manufacturing paradigms. By replacing expensive silver or mercury salts with benign potassium halides, the process not only lowers the barrier to entry for production but also simplifies the downstream purification workflow. The technical depth of this disclosure provides a clear pathway for scaling up production from laboratory grams to multi-ton commercial quantities, ensuring that the supply chain for cardiovascular and cerebrovascular therapeutics remains resilient against raw material shortages. This report analyzes the mechanistic advantages and commercial implications of adopting this specific synthetic route for Scutellarin production.
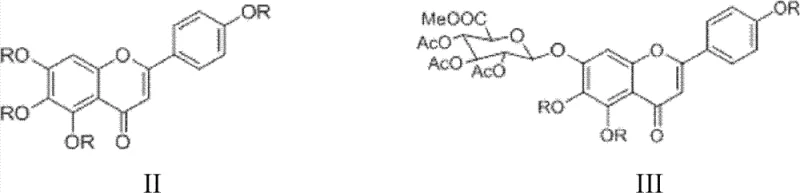
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of Scutellarin has been dominated by extraction from plant materials, a process inherently plagued by low yields and high variability due to agricultural factors. When chemical synthesis was attempted to overcome these supply bottlenecks, the existing literature and prior patents relied heavily on the use of silver salts (such as silver carbonate) or mercury salts to promote the critical glycosylation step. These heavy metal reagents are not only prohibitively expensive, driving up the cost of goods sold (COGS), but they also introduce severe environmental and safety liabilities. The removal of trace heavy metals from the final API requires additional, costly purification steps, and the disposal of metal-laden waste streams complicates regulatory compliance. Furthermore, traditional synthetic routes often involved lengthy protection and deprotection sequences that resulted in cumulative yield losses, making the final product economically unviable for mass-market pharmaceutical applications.
The Novel Approach
The methodology outlined in CN103374050A disrupts this status quo by introducing a streamlined, four-step synthetic strategy that eliminates the dependency on precious metal catalysts. Instead of silver or mercury, the process utilizes inexpensive and readily available potassium iodide or potassium bromide as promoters for the glycosylation reaction. This substitution drastically reduces raw material costs and removes the toxicity profile associated with heavy metals. Additionally, the route employs a strategic acylation step using pivaloyl or benzoyl chlorides to protect the flavone core, followed by a selective acid hydrolysis that cleverly differentiates between the protecting groups on the flavone ring and those on the sugar moiety. This selectivity allows for a shorter reaction sequence with fewer isolation steps, directly translating to higher overall throughput and reduced operational expenditure for manufacturing facilities.
Mechanistic Insights into KI/KBr-Catalyzed Glycosylation
The core innovation of this patent lies in the glycosylation mechanism, where the choice of catalyst plays a decisive role in reaction efficiency and stereo-selectivity. In the presence of potassium iodide or bromide, the alpha-brominated triacetoxy glucuronic acid methyl ester undergoes an in-situ halogen exchange or activation that enhances its electrophilicity towards the phenolic oxygen of the protected flavone. This catalytic system facilitates the formation of the beta-glycosidic bond with high fidelity, minimizing the formation of alpha-anomers which are difficult to separate and represent a significant impurity burden. The reaction proceeds smoothly in polar aprotic solvents like DMF or acetone, allowing for excellent solubility of both the flavone substrate and the sugar donor. The use of mild bases such as potassium carbonate further ensures that the sensitive flavone skeleton remains intact during the coupling process, preventing degradation or side reactions that could compromise the structural integrity of the final molecule.
Following glycosylation, the patent describes a sophisticated two-step hydrolysis protocol that demonstrates exceptional chemoselectivity. The first hydrolysis step utilizes mineral acids like sulfuric or nitric acid in methanol to selectively cleave the bulky acyl protecting groups (pivaloyl or benzoyl) from the 5, 6, and 4' positions of the flavone ring, while leaving the acetyl groups on the glucuronic acid moiety untouched. This is a critical differentiation, as it preserves the ester functionality required for the subsequent step. The second hydrolysis step employs a mineral base under oxygen-free conditions to saponify the remaining acetyl groups on the sugar and hydrolyze the methyl ester to the free carboxylic acid. This sequential deprotection strategy ensures that the final product is obtained with high purity (>98% by HPLC) and minimizes the generation of partially deprotected intermediates that are common pitfalls in flavonoid glycoside synthesis.

How to Synthesize Scutellarin Efficiently
The synthesis of Scutellarin via this patented route offers a reproducible framework for manufacturing teams looking to establish a robust production line. The process begins with the protection of 5,6,7,4'-tetrahydroxy flavone, followed by the key glycosylation event, and concludes with the sequential removal of protecting groups to reveal the active pharmacophore. Each step has been optimized for yield and purity, with specific attention paid to reaction temperatures and stoichiometry to ensure consistency. For detailed operational parameters, including exact molar ratios, solvent volumes, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process parameters defined in the patent documentation.
- Perform acylation of 5,6,7,4'-tetrahydroxy flavone using pivaloyl chloride or acetic anhydride to protect hydroxyl groups.
- Conduct glycosylation with alpha-brominated triacetoxy glucuronic acid methyl ester using potassium iodide or bromide as a catalyst.
- Execute selective acid hydrolysis to remove flavone protecting groups while retaining sugar esters, followed by base hydrolysis to finalize the glucuronic acid structure.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthetic route offers profound strategic advantages for procurement managers and supply chain leaders tasked with securing cost-effective and reliable sources of pharmaceutical intermediates. The elimination of silver and mercury salts from the process inventory immediately translates to a significant reduction in raw material procurement costs, as potassium halides are commodity chemicals with stable pricing and abundant global supply. Furthermore, the removal of heavy metals simplifies the quality control landscape, reducing the analytical burden required to certify that the final API meets stringent residual metal specifications. This streamlined purification process not only accelerates batch release times but also reduces the consumption of specialized scavenging resins or activated carbon, contributing to a leaner and more efficient manufacturing operation.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with inexpensive potassium salts fundamentally alters the cost structure of Scutellarin production. By avoiding the high capital and operational expenses associated with handling and disposing of toxic heavy metals, manufacturers can achieve substantial cost savings per kilogram of product. Additionally, the high yields reported in the patent examples across all four steps mean that less starting material is wasted, further optimizing the material balance and improving the overall economic viability of the process for large-scale commercialization.
- Enhanced Supply Chain Reliability: Reliance on botanical extraction subjects the supply chain to risks such as crop failure, seasonal variations, and geopolitical instability in sourcing regions. In contrast, this fully synthetic route relies on petrochemical-derived starting materials and common reagents that are available from multiple global suppliers. This diversification of the supply base ensures continuity of supply and protects against market volatility, allowing pharmaceutical companies to maintain consistent production schedules for their finished dosage forms without fear of raw material shortages.
- Scalability and Environmental Compliance: The process conditions described, such as moderate temperatures and the use of standard organic solvents, are inherently scalable from pilot plant to full commercial production. The absence of toxic heavy metals significantly reduces the environmental footprint of the manufacturing process, easing the burden on wastewater treatment facilities and ensuring compliance with increasingly strict environmental regulations. This 'green chemistry' aspect not only mitigates regulatory risk but also aligns with the sustainability goals of modern pharmaceutical enterprises, enhancing the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic method. They are derived from the specific technical disclosures and advantageous effects highlighted in the patent data, providing clarity on how this technology compares to existing standards in the industry. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement teams negotiating supply agreements based on this specific manufacturing process.
Q: What are the advantages of using KI/KBr catalysts over silver salts in Scutellarin synthesis?
A: Using potassium iodide or bromide eliminates the need for expensive and toxic silver or mercury salts, significantly reducing raw material costs and environmental disposal burdens while maintaining high reaction yields.
Q: How does this synthetic route improve product purity compared to plant extraction?
A: Chemical synthesis allows for precise control over reaction conditions and purification steps, consistently achieving purity levels above 98% via HPLC, whereas plant extraction often suffers from low yields and complex impurity profiles.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes readily available reagents, avoids extreme conditions, and features a short synthetic route with high yields at each step, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Scutellarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for complex pharmaceutical intermediates like Scutellarin. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the highest standards for identity, assay, and impurity profiles, giving you confidence in the material you integrate into your final drug products.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this heavy-metal-free process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your development timeline, ensuring a seamless transition from R&D to commercial supply.
