Advanced Enzymatic Synthesis of Lesinurad Intermediate for Commercial Scale-up
The pharmaceutical landscape for gout management has evolved significantly with the introduction of URAT1 inhibitors, creating a robust demand for high-quality active pharmaceutical ingredients and their precursors. Patent CN106187927B, published in early 2020, discloses a groundbreaking preparation method for methyl 2-((4-(4-cyclopropylnaphthalen-1-yl)-4H-1,2,4-triazol-3-yl)thio)acetate, a critical building block in the value chain of Lesinurad. This specific intermediate serves as the direct precursor for the final drug substance, and its efficient synthesis is paramount for ensuring a stable supply of this essential medication. The structural integrity and purity of this molecule are non-negotiable, as they directly impact the efficacy and safety profile of the final therapeutic agent. By leveraging a novel three-step synthetic strategy, this technology addresses long-standing inefficiencies in heterocyclic chemistry, offering a pathway that balances high yield with rigorous safety standards.
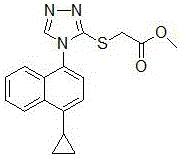
For procurement specialists and supply chain directors, the implications of this patent extend far beyond the laboratory bench. The shift towards enzymatic catalysis and the elimination of hazardous reagents represent a strategic pivot in cost reduction in pharmaceutical intermediate manufacturing. Traditional routes often rely on volatile and toxic substances that necessitate expensive containment systems and waste treatment protocols. In contrast, the methodology outlined in CN106187927B utilizes readily available starting materials such as 4-methyl-4H-3-mercapto-1,2,4-triazole and methyl bromoacetate. This accessibility ensures a reliable pharmaceutical intermediate supplier can maintain continuity of supply without being bottlenecked by the scarcity or regulatory restrictions associated with controlled chemicals. Furthermore, the simplified operational parameters reduce the barrier to entry for commercial scale-up, allowing manufacturers to respond agilely to market fluctuations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Lesinurad intermediates was plagued by significant technical and safety hurdles that hindered efficient commercial scale-up of complex pharmaceutical intermediates. As illustrated in the reaction schemes from earlier patents like US2012164222A1, traditional pathways often involved the use of concentrated nitric acid and thiophosgene. Thiophosgene is a highly toxic, volatile liquid that poses severe risks during transportation, storage, and handling, requiring specialized infrastructure that drives up capital expenditure. Moreover, the cumulative yields of these legacy processes were abysmal; for instance, the route described in US2012164222A1 achieved a total yield of merely 13.22%. Such low efficiency results in massive waste generation and inflated raw material costs, making the final product economically unviable in a competitive generic market. Other methods, such as those disclosed in WO2014198241A8, utilized carbon disulfide, an extremely flammable and explosive solvent, further compounding the safety liabilities for production facilities.
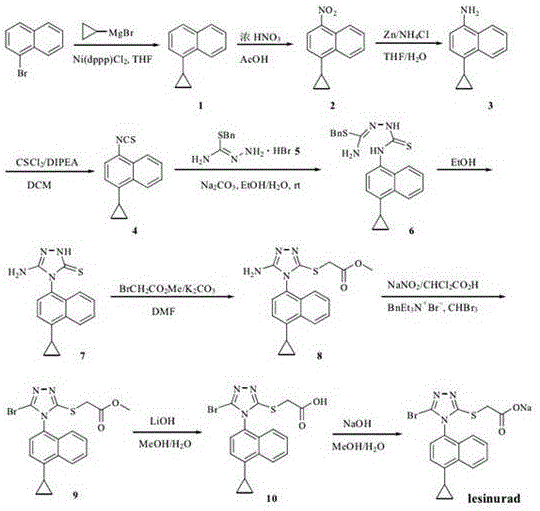
The Novel Approach
The methodology presented in CN106187927B fundamentally reengineers the synthetic logic to bypass these historical bottlenecks. Instead of relying on harsh chemical oxidants or toxic sulfur sources, the new process introduces a biocatalytic step for N-demethylation, a transformation that is typically difficult to achieve with high selectivity using traditional chemistry. This approach not only eliminates the need for dangerous reagents like thiophosgene and carbon disulfide but also streamlines the purification process. The reaction conditions are remarkably mild, operating at temperatures between 25°C and 50°C, which significantly lowers energy consumption compared to high-temperature reflux or high-pressure hydrogenation methods found in other prior art. By achieving a total process yield of 67.11% and a product purity of 99.82%, this novel route demonstrates that safety and efficiency are not mutually exclusive. It provides a robust framework for reducing lead time for high-purity pharmaceutical intermediates by minimizing the number of purification cycles and maximizing the throughput of each batch.
Mechanistic Insights into Enzymatic N-Demethylation and Triazole Functionalization
The core innovation of this synthesis lies in the strategic application of N-demethylase enzymes, specifically nicotine N-demethylase or erythromycin N-demethylase, to effect the removal of the methyl group from the triazole ring. In traditional organic synthesis, N-demethylation often requires harsh conditions that can degrade sensitive functional groups or lead to over-oxidation. However, the enzymatic mechanism operates under physiological pH and moderate temperatures, utilizing a methanol/potassium dihydrogen phosphate buffer system to maintain optimal enzyme activity. This biocatalytic step ensures exceptional regioselectivity, preventing side reactions on the sulfur atom or the ester moiety. The subsequent N-arylation step, where the demethylated triazole reacts with 1-bromo-4-cyclopropylnaphthalene, proceeds smoothly in the presence of potassium carbonate in DMF. The mechanistic elegance of this sequence ensures that the cyclopropyl group, which is susceptible to ring-opening under acidic or radical conditions, remains intact throughout the synthesis.
From an impurity control perspective, this mechanism offers distinct advantages for meeting stringent regulatory requirements. The high specificity of the enzymatic step drastically reduces the formation of structural analogs and by-products that are difficult to separate. In the experimental examples provided, the intermediate obtained after the S-alkylation step showed an HPLC purity of 99.75%, and the final product consistently exceeded 99.8% purity. This level of cleanliness is critical for a high-purity pharmaceutical intermediate, as it simplifies the downstream processing of the final API. The absence of heavy metal catalysts, which are often required for C-N bond formation in cross-coupling reactions, further simplifies the impurity profile. This allows manufacturers to avoid the costly and time-consuming heavy metal scavenging steps that are mandatory when using transition metal catalysts, thereby enhancing the overall process mass intensity (PMI) and environmental footprint.
How to Synthesize Methyl 2-((4-(4-cyclopropylnaphthalen-1-yl)-4H-1,2,4-triazol-3-yl)thio)acetate Efficiently
The synthesis protocol detailed in the patent provides a clear, reproducible roadmap for laboratory and pilot-scale production. The process begins with the S-alkylation of the triazole thiol, followed by the critical enzymatic demethylation, and concludes with the N-arylation to install the naphthalene scaffold. Each step is optimized for maximum conversion and ease of isolation, utilizing standard workup procedures such as filtration and crystallization rather than complex chromatography.
- S-Alkylation of 4-methyl-4H-3-mercapto-1,2,4-triazole with methyl bromoacetate using potassium carbonate in DMF.
- Enzymatic N-demethylation using nicotine N-demethylase or erythromycin N-demethylase in a methanol/buffer system.
- N-Arylation of the demethylated triazole with 1-bromo-4-cyclopropylnaphthalene under basic conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational resilience, the adoption of this synthetic route offers compelling economic arguments rooted in process chemistry rather than speculative financial modeling. The elimination of thiophosgene and carbon disulfide removes the need for specialized hazardous material handling permits and expensive scrubbing systems, leading to substantial cost savings in facility operations and compliance. Furthermore, the dramatic improvement in yield—from roughly 13% in older methods to over 67% in this new process—means that significantly less raw material is required to produce the same amount of product. This efficiency gain directly translates to a lower cost of goods sold (COGS) and reduced waste disposal fees, aligning with modern green chemistry principles.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents with commodity chemicals like methyl bromoacetate and potassium carbonate significantly lowers the direct material costs. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high yield minimizes the loss of valuable starting materials, collectively driving down the manufacturing overhead.
- Enhanced Supply Chain Reliability: By avoiding reagents that are subject to strict regulatory controls or supply volatility, such as thiophosgene, manufacturers can secure a more stable supply chain. The use of common solvents like DMF and methanol ensures that production is not halted due to solvent shortages, providing a reliable foundation for long-term supply agreements with API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simplicity and lack of extreme conditions (e.g., high pressure or cryogenic temperatures). The reduction in toxic waste generation facilitates easier compliance with increasingly stringent environmental regulations, reducing the risk of production shutdowns due to environmental violations and enhancing the company's sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the safety advantages of this new Lesinurad intermediate synthesis route?
A: Unlike conventional methods that utilize highly toxic thiophosgene or explosive carbon disulfide, this novel process employs mild enzymatic catalysis and standard alkylating agents, significantly reducing occupational hazards and environmental pollution risks.
Q: How does the yield of this enzymatic method compare to prior art?
A: The total yield of this process reaches approximately 67.11%, which represents a substantial improvement over previous methods reported in US2012164222A1 (13.22%) and WO2014008295A (20.32%), leading to better atom economy.
Q: Is this process suitable for large-scale GMP manufacturing?
A: Yes, the reaction conditions are mild (25-50°C), do not require high-pressure hydrogenation equipment, and utilize common solvents like DMF and methanol, making it highly adaptable for multi-kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lesinurad Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity levels demonstrated in CN106187927B can be reliably replicated on an industrial scale. We understand that for a critical gout medication intermediate, consistency is key. Our rigorous QC labs and stringent purity specifications guarantee that every batch of Lesinurad intermediate meets the exacting standards required for global regulatory submissions, providing our partners with peace of mind and supply security.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized version of this enzymatic route, we can help you achieve significant efficiencies in your supply chain. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the pharmaceutical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
