Revolutionizing Valienamine Production: Solid Catalyst Technology for Commercial Scale-up
The pharmaceutical industry is constantly seeking more efficient pathways for synthesizing critical intermediates, and the production of valienamine stands as a prime example of this technological evolution. Patent CN100347149C introduces a transformative preparation method for valienamine utilizing solid catalysts, marking a significant departure from conventional biological and liquid-phase chemical synthesis. This innovation addresses the longstanding challenges of low yield and complex purification that have historically plagued the manufacturing of this key precursor for anti-diabetic agents like voglibose and acarbose. By leveraging heterogeneous catalysis, specifically through strong acidic cation exchange resins or zeolites, the process achieves selective hydrolysis of acarbose and validamycin derivatives with remarkable efficiency. For R&D directors and procurement strategists, this patent represents a viable route to secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials with reduced environmental impact. The shift from time-consuming bacterial fermentation to a robust chemical catalysis system underscores a major leap in process intensification, promising better control over reaction parameters and product consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the commercial-scale production of valienamine has been constrained by two primary methodologies, both of which exhibit significant operational drawbacks that hinder cost-effective manufacturing. The first approach involves the direct bacterial hydrolysis of validamycin, a process that is inherently slow and biologically variable, often requiring fermentation times extending from 24 to 200 hours to achieve modest conversion rates. This biological dependency introduces risks related to strain stability and contamination, leading to inconsistent batch quality and extended lead times for high-purity pharmaceutical intermediates. The second conventional method relies on total synthesis starting from glucose or chemical hydrolysis using homogeneous liquid acids such as sulfuric or hydrochloric acid. While chemical hydrolysis can be faster, it frequently suffers from insufficient selectivity, affecting only terminal groups while leaving the core structure intact, or conversely, causing degradation of the sensitive valienamine moiety. Furthermore, the use of liquid acids generates substantial quantities of waste acid and saline wastewater, creating severe environmental compliance burdens and escalating the cost reduction in API manufacturing due to expensive waste treatment protocols.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes heterogeneous solid catalysts to drive the selective hydrolysis of pseudotrisaccharides like acarbose and validamycin. This method employs robust materials such as strong acid cation exchange resins (e.g., Amberlyst 131) or zeolites (e.g., ZSM-5) which function effectively at temperatures ranging from 40°C to 150°C. The heterogeneity of the catalyst allows for a clean separation of the reaction mixture via simple filtration, eliminating the need for complex neutralization steps and significantly reducing the formation of by-products like pseudo-sugars. This technological pivot not only enhances the yield to an impressive range of 75% to 90% but also streamlines the downstream processing by enabling the adsorption of the target valienamine directly onto the catalyst matrix. For supply chain heads, this translates to a more predictable and scalable process, facilitating the commercial scale-up of complex pharmaceutical intermediates without the bottlenecks associated with biological fermentation cycles or corrosive liquid acid handling.
Mechanistic Insights into Solid Catalyst-Mediated Selective Hydrolysis
The core of this technological advancement lies in the precise mechanistic action of the solid acid or base catalysts on the glycosidic bonds of the substrate molecules. When substrates such as acarbose derivatives are introduced to the solid catalyst surface, the active sites—typically sulfonic acid groups on resin matrices or acidic sites on zeolite frameworks—protonate the oxygen atoms in the glycosidic linkages. This protonation weakens the bond, facilitating a nucleophilic attack by water molecules that results in the cleavage of the specific alpha-glycosidic bond connecting the valienamine moiety to the sugar units. Unlike liquid acids which permeate the entire solution and may cause non-specific hydrolysis or degradation, the solid catalyst provides a controlled microenvironment that favors the formation of valienamine while minimizing side reactions. The reaction conditions are finely tuned, with cation exchange resins operating optimally between 80°C and 120°C, ensuring that the thermal energy is sufficient to overcome the activation barrier without compromising the structural integrity of the heat-sensitive carbocyclic sugar. This selectivity is crucial for maintaining the stereochemistry required for the biological activity of the final drug product.
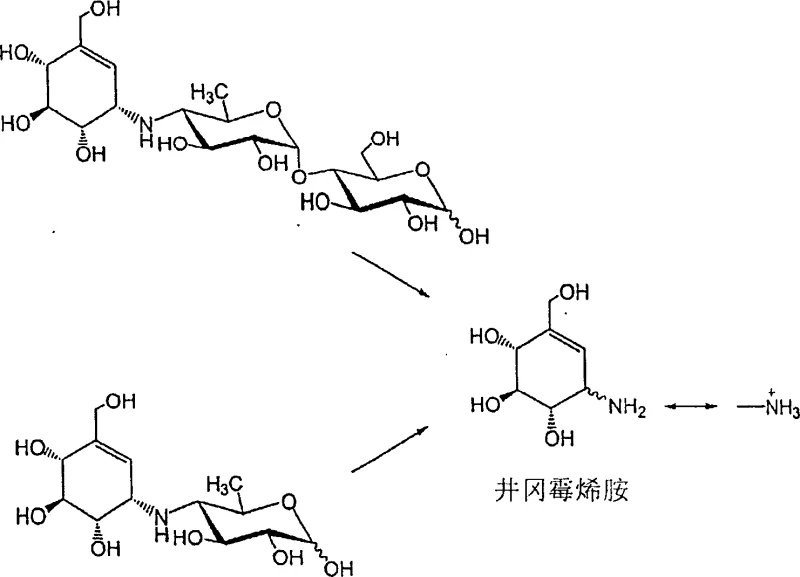
Furthermore, the purification mechanism integrated into this process offers a distinct advantage in impurity control. Upon completion of the hydrolysis, the valienamine product is selectively adsorbed onto the porous matrix of the catalyst resin or zeolite via interaction with the functional groups, while smaller by-products like monosaccharides and disaccharides remain in the supernatant or are washed away. This in-situ capture mechanism acts as a primary purification step, drastically reducing the load on subsequent chromatographic columns. The adsorbed valienamine is then eluted using a mild amine solution, typically ammonia water at concentrations between 0.5% and 20%, which displaces the product from the acidic sites without inducing degradation. This elegant interplay between reaction and separation ensures that the final crystalline product obtained after recrystallization is of exceptional purity, meeting the stringent specifications required for regulatory submission and clinical application.
How to Synthesize Valienamine Efficiently
The synthesis of valienamine via this solid catalyst method is designed for operational simplicity and robustness, making it highly suitable for industrial adoption. The process begins with the preparation of an aqueous slurry containing the substrate, such as acarbose or validamycin derivatives, and the selected solid catalyst, with catalyst loading optimized between 50 to 150 parts per 100 parts of substrate. The reaction mixture is then heated in a pressure reactor to the specified temperature range, typically maintained for 2 to 24 hours depending on the specific catalyst activity and desired conversion rate. Following the reaction, the solid catalyst is separated by filtration, and the adsorbed product is recovered through elution, followed by a final polishing step using a weak acid cation exchange resin column to remove any residual impurities. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the substrate (acarbose or validamycin derivatives) with a solid catalyst such as Amberlyst 131 or ZSM-5 zeolite in an aqueous medium.
- Heat the mixture to a temperature between 80°C and 120°C (depending on catalyst type) and maintain reaction for 2 to 24 hours to ensure complete selective hydrolysis.
- Filter the solid catalyst, elute the adsorbed valienamine using an amine solution, and perform final purification via a weak acid cation exchange resin column to obtain crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this solid catalyst technology offers profound strategic benefits that extend beyond mere technical feasibility. The transition from biological fermentation to chemical catalysis fundamentally alters the cost structure and reliability of the supply chain for valienamine. By eliminating the need for sterile fermentation facilities and the associated long incubation periods, manufacturers can achieve a drastic simplification of the production infrastructure, leading to substantial cost savings in capital expenditure and operational overhead. The ability to run reactions in standard chemical reactors rather than bioreactors allows for greater flexibility in production scheduling and capacity utilization, ensuring a more consistent flow of materials to downstream formulation plants. Moreover, the reduction in waste generation aligns with increasingly strict global environmental regulations, mitigating the risk of production shutdowns due to compliance issues and enhancing the overall sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The implementation of solid catalysts eliminates the recurring costs associated with purchasing and disposing of large volumes of liquid mineral acids, which are not only expensive but also hazardous to handle. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to by-products, thereby improving the overall material efficiency and reducing the cost of goods sold. The catalyst itself, being a solid heterogeneous material, can potentially be regenerated and reused multiple times, further amortizing the cost of consumables over a larger production volume. This economic efficiency is critical for maintaining competitiveness in the generic pharmaceutical market where margin pressure is intense.
- Enhanced Supply Chain Reliability: Unlike bacterial fermentation which is susceptible to biological variability and contamination events that can halt production for weeks, the chemical catalytic process offers a deterministic and reproducible outcome. The reaction kinetics are well-defined, allowing for precise prediction of batch completion times and reliable inventory planning. This predictability is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers receive their raw materials on schedule. The robustness of the solid catalysts also means that the process is less sensitive to minor fluctuations in raw material quality, providing an additional layer of security against supply disruptions.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst system facilitates straightforward scale-up from laboratory to commercial production without the complex mass transfer limitations often encountered in aerobic fermentations. The process generates significantly less saline wastewater and no waste acid streams, simplifying the effluent treatment plant requirements and reducing the environmental footprint of the manufacturing site. This green chemistry approach not only lowers compliance costs but also enhances the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical value chain, appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the solid catalyst production method for valienamine, derived directly from the insights provided in patent CN100347149C. These answers are intended to clarify the operational advantages and feasibility of implementing this technology in a commercial setting. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or procurement partnerships.
Q: How does the solid catalyst method improve upon traditional bacterial hydrolysis for valienamine?
A: Traditional bacterial hydrolysis requires lengthy fermentation periods ranging from 24 to 200 hours and often suffers from low yields. The solid catalyst method described in patent CN100347149C significantly reduces reaction time to as little as 2 hours while achieving yields between 75% and 90%, drastically improving throughput.
Q: What specific catalysts are recommended for this hydrolysis process?
A: The patent identifies strong acid cation exchange resins like Amberlyst 131, strong base anion exchange resins like Amberjet 4400OH, and zeolite-based catalysts such as ZSM-5 as highly effective. These heterogeneous catalysts allow for easy separation and potential reuse, unlike homogeneous liquid acids.
Q: Does this method generate significant hazardous waste compared to liquid acid hydrolysis?
A: No, this method is designed as a purification technology that mitigates environmental issues associated with traditional homogeneous liquid acid catalysis. By using solid catalysts, the process eliminates the generation of large volumes of waste acid and simplifies wastewater treatment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valienamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications, and we are committed to bridging the gap between innovative patent technologies and commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in laboratory settings are faithfully reproduced at an industrial scale. We operate state-of-the-art rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of valienamine meets the exacting standards required by global regulatory bodies. Our expertise in handling complex carbohydrate chemistry and solid catalyst systems positions us as a preferred partner for companies seeking to optimize their supply chain for anti-diabetic APIs.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our solid catalyst-derived valienamine. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your drug development pipeline.
