Advanced Vortioxetine Manufacturing: Overcoming Legacy Toxicity and Yield Barriers
Advanced Vortioxetine Manufacturing: Overcoming Legacy Toxicity and Yield Barriers
The pharmaceutical landscape for treating Major Depressive Disorder (MDD) has been significantly reshaped by the introduction of Vortioxetine (Lu AA 21004), a multimodal antidepressant with a unique receptor activity profile. However, the commercial viability of such complex small molecules often hinges on the robustness of their synthetic routes. Patent CN105622546B, published in August 2020, presents a pivotal advancement in the preparation method of Vortioxetine, addressing critical bottlenecks found in earlier methodologies. This technical disclosure outlines a streamlined four-step synthesis that replaces hazardous ferrocene-based chemistry with efficient nucleophilic substitutions and palladium-catalyzed couplings. For R&D directors and procurement strategists, this patent represents a pathway to not only higher purity profiles but also a fundamentally safer and more cost-effective supply chain for this high-value API intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically referenced in Chinese patent CN2819025, relied heavily on cumbersome and environmentally taxing chemistries that posed severe risks for industrial scale-up. The traditional route utilized ferrocene complexes of o-dichlorobenzene alongside resin-protected piperazine derivatives. This approach suffered from intrinsic inefficiencies, most notably a disastrously low yield of merely 14% in the final decomplexation and resin fracture step. Furthermore, the reliance on ferrocene introduces significant toxicity concerns and complicates waste stream management, creating substantial downstream costs for environmental compliance. The multi-step nature of protecting and deprotecting resins also elongates the production cycle, increasing the risk of impurity accumulation and reducing overall throughput capacity for manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN105622546B adopts a direct and convergent strategy that bypasses the need for transition metal complexes and solid-phase resins entirely. The new route initiates with a straightforward alkaline condensation between 2-bromophenylthiol and N-methylpiperazine, achieving a robust yield of 83.6% for the thiophenol intermediate. This is followed by a palladium-catalyzed cross-coupling with 2,4-dimethyliodobenzene, which constructs the core diaryl sulfide scaffold with a respectable 70.3% yield. By shifting to solution-phase chemistry with readily available liquid reagents, the process drastically simplifies unit operations and enhances the safety profile of the manufacturing facility.
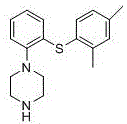
The strategic advantage of this novel approach lies in its modularity and the use of standard organic transformations that are well-understood in process chemistry. The elimination of the resin fracture step alone removes a major source of yield loss and particulate contamination. Moreover, the final deprotection step utilizes mild basic conditions to remove the phenyl formate group, delivering the final Vortioxetine product in an impressive 86.6% yield. This dramatic improvement in step-wise efficiency translates directly into reduced raw material consumption and a lower cost of goods sold (COGS), making it an attractive option for generic API manufacturers seeking to enter the market with a competitive price point.
Mechanistic Insights into Pd-Catalyzed C-S Bond Formation
The cornerstone of this synthesis is the formation of the carbon-sulfur bond between the piperazine-substituted thiophenol and the dimethyl-iodobenzene moiety. This transformation is achieved using a palladium catalyst system, specifically employing Pd2(dba)3 and rac-BINAP as the ligand in the presence of sodium tert-butoxide. The mechanism likely proceeds through a classic catalytic cycle involving oxidative addition of the aryl iodide to the Pd(0) species, followed by coordination and deprotonation of the thiol to form a Pd-thiolate complex. Subsequent reductive elimination releases the desired diaryl sulfide product and regenerates the active Pd(0) catalyst. The choice of rac-BINAP is critical here, as bulky bidentate phosphine ligands facilitate the reductive elimination step, which is often the rate-determining step in C-S bond formations, thereby ensuring high conversion rates even with sterically hindered substrates.
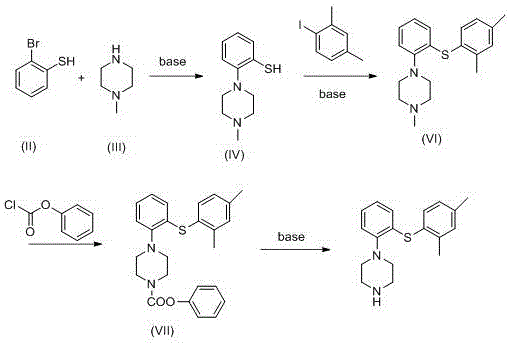
Impurity control is meticulously managed through the introduction of a phenyl chloroformate protection step prior to the final deprotection. By converting the secondary amine of the piperazine ring into a carbamate (Intermediate VII), the process prevents potential N-arylation side reactions or oxidation of the nitrogen center during subsequent handling. The final step involves a simple hydrolysis or alcoholysis of this carbamate using potassium carbonate in toluene. This deprotection strategy is superior to acid-labile groups because it avoids the use of strong acids that could potentially degrade the sensitive thioether linkage or cause demethylation of the piperazine ring. The result is a final product with a cleaner impurity profile, reducing the burden on downstream purification processes such as crystallization or chromatography.
How to Synthesize Vortioxetine Efficiently
The synthesis of Vortioxetine described in this patent offers a practical blueprint for process chemists aiming to replicate these results in a pilot or production plant. The protocol relies on standard reflux conditions in common solvents like toluene and DMF, eliminating the need for specialized cryogenic equipment. The procedure emphasizes the importance of stoichiometric balance, particularly in the Pd-catalyzed step where the ratio of ligand to metal and base to substrate dictates the reaction kinetics. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures required to achieve the reported yields, please refer to the standardized synthesis guide below.
- Condense 2-bromophenylthiol with N-methylpiperazine under alkaline conditions to form 2-(1-methylpiperazinyl)thiophenol.
- Perform Pd-catalyzed cross-coupling with 2,4-dimethyliodobenzene to establish the diaryl sulfide core.
- Protect the piperazine nitrogen with phenyl chloroformate, followed by base-mediated deprotection to yield final Vortioxetine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to the synthesis route outlined in CN105622546B offers tangible strategic benefits beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain for raw materials. By removing the dependency on specialized ferrocene complexes and functionalized resins, manufacturers can source starting materials like 2-bromophenylthiol and 2,4-dimethyliodobenzene from a broader base of commodity chemical suppliers. This diversification mitigates the risk of supply disruptions and provides greater leverage in price negotiations, leading to significant cost reduction in Vortioxetine manufacturing.
- Cost Reduction in Manufacturing: The elimination of toxic ferrocene reagents and resin supports removes the need for expensive hazardous waste disposal protocols and complex filtration units required for resin handling. Furthermore, the substantial increase in yield at every stage—particularly the jump from 14% to 86.6% in the final step—means that less raw material is wasted per kilogram of final API produced. This efficiency gain directly lowers the variable cost of production, allowing for more competitive pricing in the generic antidepressant market without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on robust, solution-phase chemistry ensures that production timelines are predictable and consistent. Unlike solid-phase synthesis which can suffer from swelling issues or incomplete reactions due to diffusion limitations, this liquid-phase process allows for real-time monitoring via HPLC or GC. The use of stable intermediates like the phenyl carbamate (VII) also allows for potential stockpiling of key precursors, decoupling the production of the final API from the immediate availability of all starting materials and enhancing overall supply continuity.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is far superior. The removal of heavy metal ferrocene complexes reduces the heavy metal load in the effluent, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The process operates at atmospheric pressure and moderate reflux temperatures, reducing energy consumption compared to high-pressure hydrogenation or cryogenic steps. This 'green chemistry' alignment not only reduces operational costs but also future-proofs the manufacturing site against tightening regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Vortioxetine synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the operational benefits for potential manufacturing partners.
Q: How does this new process improve upon previous ferrocene-based methods?
A: The legacy method described in CN2819025 relied on toxic ferrocene complexes and resin protection, resulting in a critically low final step yield of only 14%. The new process eliminates these hazardous reagents, utilizing standard alkaline condensation and Pd-catalysis to achieve significantly higher yields (up to 86.6% in the final step) and safer operational conditions.
Q: What represents the critical quality control point in this synthesis?
A: The introduction of the phenyl formate protecting group (Intermediate VII) is crucial for purity. It prevents side reactions during the thioether formation and allows for a clean deprotection step using potassium carbonate, ensuring the final API meets stringent impurity specifications required for antidepressant medications.
Q: Is this route suitable for large-scale commercial production?
A: Yes, the process utilizes robust, scalable chemistry such as reflux in toluene and DMF, avoiding cryogenic conditions or exotic catalysts. The elimination of resin fracture steps and the use of common bases like potassium carbonate make it highly amenable to multi-kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vortioxetine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antidepressants like Vortioxetine requires more than just a patent; it demands a partner with deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor train is seamless. We are equipped with rigorous QC labs and advanced analytical instrumentation to meet stringent purity specifications, guaranteeing that every batch of Vortioxetine intermediate or API meets the highest global pharmacopeial standards.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to leverage this optimized synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to obtain specific COA data for our pilot batches and comprehensive route feasibility assessments, and let us help you secure a reliable, cost-effective supply of this critical mental health medication.
