Advanced Synthesis of Posaconazole Intermediates: A Safer, Scalable Route for Global API Manufacturing
The pharmaceutical landscape for broad-spectrum antifungal agents continues to evolve, driven by the critical need for safer and more efficient manufacturing processes for complex Active Pharmaceutical Ingredients (APIs). Patent CN108586280B introduces a transformative methodology for synthesizing N'-[(2S,3S)-2-(benzyloxy)pentan-3-yl]formylhydrazine, a pivotal chiral intermediate in the production of Posaconazole. This advanced triazole antifungal, originally developed to treat invasive Aspergillus and Candida infections, demands high stereochemical purity to ensure therapeutic efficacy. The disclosed innovation addresses significant bottlenecks in traditional synthesis by replacing hazardous reagents with a more benign, operationally simple protocol. By shifting away from moisture-sensitive aluminum hydrides to a sodium-mediated reduction strategy, this patent offers a compelling value proposition for reliable posaconazole intermediate suppliers aiming to optimize their supply chains. The structural complexity of the final drug, as illustrated below, necessitates precise control over every synthetic step to maintain the integrity of the chiral centers.
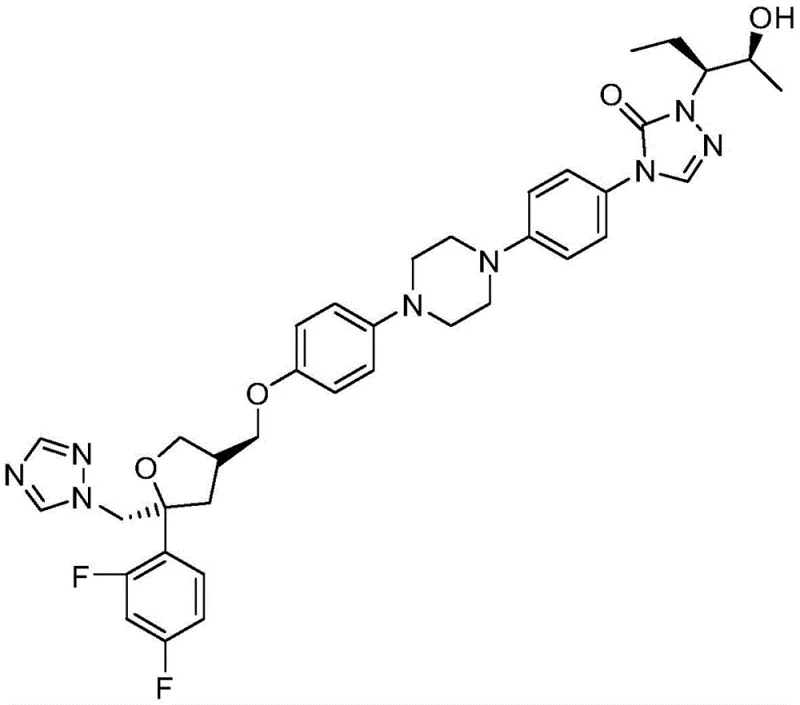
Furthermore, the strategic importance of this intermediate cannot be overstated, as it forms the lipophilic side chain essential for the drug's bioavailability. The ability to produce this fragment efficiently directly correlates to the cost reduction in antifungal API manufacturing, a key metric for procurement teams managing global budgets. This report delves deep into the mechanistic advantages and commercial implications of adopting this novel pathway, providing R&D directors and supply chain heads with the technical intelligence needed to make informed sourcing decisions. The transition from legacy methods to this improved process represents a significant leap forward in green chemistry principles applied to fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically referenced in international patent application WO 2013042138, outlines a synthesis route that, while functional, suffers from severe operational and safety drawbacks that hinder large-scale adoption. The conventional pathway typically involves a five-step sequence that relies heavily on Diisobutylaluminum hydride (DIBAL-H) for the critical reduction of an ester or nitrile to the corresponding aldehyde. DIBAL-H is notoriously pyrophoric, meaning it can ignite spontaneously upon contact with air, posing extreme safety risks in a manufacturing environment. Moreover, reactions involving DIBAL-H mandate strictly anhydrous conditions, requiring expensive solvent drying systems and inert atmosphere protocols that drastically increase capital expenditure and operational overhead. The lengthy five-step process also inherently accumulates impurities, leading to lower overall yields and complicating the purification of the final high-purity pharmaceutical intermediates. These factors combined create a fragile supply chain vulnerable to disruptions and cost volatility.
The Novel Approach
In stark contrast, the methodology disclosed in CN108586280B streamlines the synthesis into a robust four-step sequence that fundamentally alters the risk profile of the operation. The core innovation lies in the replacement of the hazardous DIBAL-H reduction with a metallic sodium-mediated reduction in ethanol. This switch not only eliminates the fire hazard associated with organo-aluminum compounds but also relaxes the stringent requirement for anhydrous conditions, allowing for more flexible and cost-effective processing. The new route begins with the condensation of (S)-2-benzyloxypropionic acid and ethylenediamine, followed by the key sodium reduction to generate the aldehyde in situ. This is subsequently reacted with formylhydrazine and subjected to a Grignard addition to install the ethyl group with high stereocontrol. The visual representation of this optimized pathway highlights the logical flow and reduced complexity compared to legacy methods.
![Novel 4-step synthetic route for N'-[(2S,3S)-2-(benzyloxy)pentan-3-yl]formylhydrazine avoiding DIBAL-H](/insights/img/posaconazole-intermediate-synthesis-pharma-supplier-20260308154355-04.webp)
By condensing the synthetic steps and utilizing readily available reagents like metallic sodium and ethyl bromide, the novel approach significantly enhances the commercial scale-up of complex pharmaceutical intermediates. The post-treatment procedures are notably simplified, often requiring only precipitation and filtration rather than complex chromatographic separations. This efficiency translates directly into higher throughput and reduced waste generation, aligning with modern environmental compliance standards. For procurement managers, this means a more stable supply of critical materials with reduced lead times, as the reliance on specialized, hazardous reagents is minimized. The robustness of this chemistry ensures consistent quality, which is paramount for regulatory approval and patient safety in the final antifungal medication.
Mechanistic Insights into Sodium-Mediated Reduction and Grignard Addition
The heart of this technological advancement lies in the mechanistic elegance of the sodium-mediated reduction of the imidazoline intermediate. In this step, metallic sodium acts as a single-electron donor in an alcoholic medium, facilitating the cleavage of the imidazoline ring to reveal the aldehyde functionality with remarkable chemoselectivity. Unlike hydride reductions which can be prone to over-reduction to alcohols if not carefully controlled, this method leverages the specific reactivity of the imidazoline scaffold to stop precisely at the aldehyde stage. The reaction is conducted at mild temperatures, typically between -5°C and 5°C, which helps suppress side reactions such as aldol condensations or racemization of the sensitive chiral center adjacent to the benzyloxy group. This precise control is critical for maintaining the optical purity required for the downstream biological activity of Posaconazole. The use of ethanol as a proton source ensures a clean conversion, and the subsequent workup with oxalic acid effectively quenches excess sodium and facilitates the isolation of the aldehyde.
Following the formation of the hydrazone intermediate, the process employs a Grignard reaction to introduce the ethyl side chain, establishing the second chiral center at the 3-position. The use of ethyl magnesium bromide (prepared in situ from ethyl bromide and magnesium) allows for a nucleophilic attack on the hydrazone double bond. The stereochemical outcome of this addition is governed by the existing chirality at the 2-position, leading to the desired (2S,3S) diastereomer through a highly organized transition state. The inclusion of N,O-bis(trimethylsilyl)acetamide (BSA) in the reaction mixture serves to activate the hydrazone by silylation, enhancing its electrophilicity and ensuring complete conversion. This mechanistic nuance prevents the formation of unreacted starting material and minimizes the generation of diastereomeric impurities. The result is a process that delivers high-purity pharmaceutical intermediates with a defined impurity profile, simplifying the regulatory filing process for the final API manufacturer.
How to Synthesize N'-[(2S,3S)-2-(benzyloxy)pentan-3-yl]formylhydrazine Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and purity. The initial condensation step utilizes a molar ratio of acid to diamine optimized to drive the equilibrium towards the imidazoline while minimizing polymerization. Subsequent reduction steps must be monitored closely to prevent exotherms, although the sodium/ethanol system is inherently more manageable than DIBAL-H additions. The final Grignard step benefits from the in situ preparation of the reagent, ensuring high reactivity and minimizing the storage risks associated with pre-formed organometallics. Detailed standard operating procedures for each stage, including specific quenching and extraction protocols, are essential for technology transfer.
- Condense (S)-2-benzyloxypropionic acid with ethylenediamine to form the imidazoline intermediate.
- Perform a chemoselective reduction using metallic sodium in ethanol to generate the key aldehyde precursor.
- React the aldehyde with formylhydrazine followed by stereoselective Grignard addition to finalize the chiral side chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the raw material portfolio. By eliminating the need for Diisobutylaluminum hydride, a reagent that often commands a premium price due to its hazardous nature and specialized packaging requirements, manufacturers can achieve cost reduction in antifungal API manufacturing. Furthermore, the removal of strict anhydrous requirements reduces the energy consumption associated with solvent drying and nitrogen purging, contributing to a lower carbon footprint and reduced utility costs. The simplified workup procedures, which rely on precipitation and filtration rather than column chromatography, significantly decrease solvent usage and waste disposal costs, enhancing the overall economic viability of the process.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous aluminum hydrides directly lowers the Bill of Materials (BOM) cost. Additionally, the reduction in step count from five to four decreases labor hours, equipment occupancy time, and solvent consumption. The use of commodity chemicals like metallic sodium and ethyl bromide ensures stable pricing and availability, shielding the supply chain from the volatility often seen with specialty reagents. This structural cost advantage allows for more competitive pricing strategies in the global market for antifungal intermediates.
- Enhanced Supply Chain Reliability: Reliance on pyrophoric reagents often introduces logistical bottlenecks, as their transport and storage require specialized infrastructure and permits. By switching to safer, non-pyrophoric reagents, the supply chain becomes more resilient and less prone to regulatory delays. The robustness of the sodium-mediated reduction means that production can continue with fewer interruptions due to safety incidents or equipment failures related to moisture ingress. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: The milder reaction conditions and simplified purification steps make this process ideally suited for commercial scale-up of complex pharmaceutical intermediates. The reduced generation of aluminum-containing waste streams simplifies effluent treatment and aligns with increasingly stringent environmental regulations. The ability to scale from kilogram to multi-ton quantities without significant process re-engineering provides a clear path for meeting growing global demand for Posaconazole. This scalability ensures long-term supply security for partners investing in this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is vital for R&D teams evaluating technology transfer and procurement teams assessing supplier capabilities. The answers are derived directly from the experimental data and beneficial effects described in patent CN108586280B, ensuring accuracy and relevance to real-world manufacturing scenarios.
Q: Why is the new synthesis route safer than the conventional method?
A: The conventional route relies on Diisobutylaluminum hydride (DIBAL-H), a pyrophoric and moisture-sensitive reagent requiring strict anhydrous conditions. The novel method replaces this with metallic sodium in ethanol, significantly reducing fire hazards and operational complexity.
Q: How does this process impact the cost of Posaconazole production?
A: By eliminating expensive aluminum reagents and reducing the step count from five to four, the process lowers raw material costs and simplifies purification, leading to substantial overall manufacturing savings.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the reaction conditions are milder and do not require extreme anhydrous environments, making the process highly robust and easily scalable for industrial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N'-[(2S,3S)-2-(benzyloxy)pentan-3-yl]formylhydrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is critical for the future of pharmaceutical manufacturing. Our team of expert chemists has extensively analyzed the methodology disclosed in CN108586280B and possesses the technical capability to implement this advanced process at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate meets the high standards required for API synthesis. We are committed to delivering value through technical excellence and operational reliability.
We invite you to collaborate with us to leverage this innovative synthesis route for your Posaconazole projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can optimize your bottom line. Please contact us to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure a supply chain that is not only cost-effective but also built on the foundation of safety, sustainability, and scientific rigor.
