Advanced Synthesis of Posaconazole Intermediates: A Safe and Scalable Commercial Route
The pharmaceutical industry constantly seeks robust synthetic pathways for high-value antifungal agents, and the synthesis of Posaconazole intermediates remains a critical focus area for process chemists. Patent CN109796368B introduces a transformative methodology for producing N'-[(2S,3S)-2-(benzyloxy)pentan-3-yl]formylhydrazine, a key chiral building block in the assembly of this broad-spectrum triazole antifungal. This innovation addresses long-standing challenges in the field by replacing hazardous stoichiometric reductants with a catalytic hydrogenation approach, specifically utilizing a Rosenmund reduction strategy. By shifting away from dangerous reagents like diisobutylaluminum hydride, this technology not only enhances operational safety but also aligns with green chemistry principles through improved atom economy and catalyst recyclability. For global procurement teams and R&D directors, this represents a significant opportunity to optimize the supply chain for complex pharmaceutical intermediates while mitigating production risks associated with pyrophoric materials.
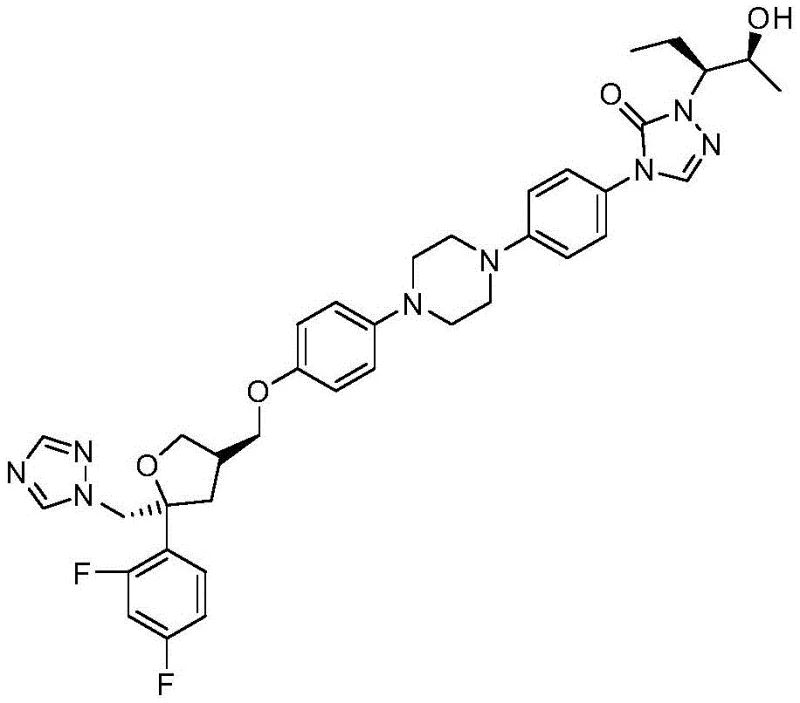
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific chiral hydrazine intermediate has been plagued by inefficient multi-step sequences that rely on hazardous chemistry. As illustrated in prior art such as international patent application WO 2013042138, traditional routes often involve up to five distinct synthetic steps, leading to cumulative yield losses and increased waste generation. A major bottleneck in these conventional processes is the reliance on diisobutylaluminum hydride (DIBAL-H) for the reduction of esters to aldehydes. DIBAL-H is not only expensive but also highly pyrophoric, requiring rigorous anhydrous conditions and specialized handling equipment to prevent fires during quenching and workup. Furthermore, these older methods frequently necessitate complex purification techniques, such as column chromatography, which are impractical and cost-prohibitive at the metric-ton scale required for commercial API production.
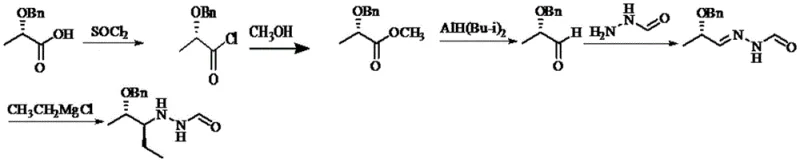
The Novel Approach
The methodology disclosed in CN109796368B offers a streamlined alternative that fundamentally restructures the synthetic logic to prioritize safety and efficiency. Instead of the lengthy ester reduction pathway, this novel approach employs a direct acylation followed by a catalytic Rosenmund reduction to generate the crucial aldehyde intermediate. This shift allows for the use of molecular hydrogen as the reductant, mediated by a palladium barium sulfate catalyst, which is significantly safer and more cost-effective than stoichiometric aluminum hydrides. The subsequent condensation with formylhydrazine and stereoselective Grignard addition completes the carbon skeleton construction in fewer overall operations. By simplifying the workflow and eliminating the most dangerous step of the traditional route, this new process drastically reduces the barrier to entry for safe, large-scale manufacturing of this high-value pharmaceutical intermediate.
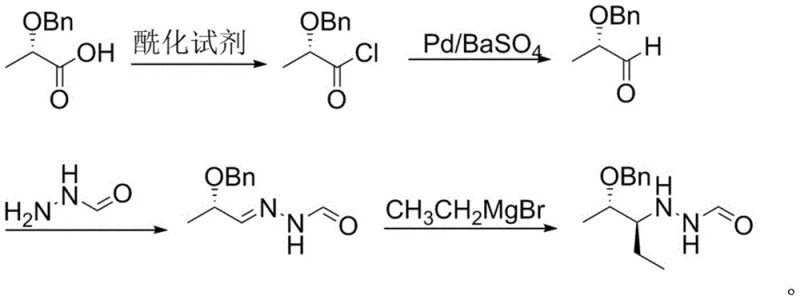
Mechanistic Insights into Rosenmund Reduction and Grignard Addition
The core chemical innovation lies in the application of the Rosenmund reduction to a benzyloxy-substituted acid chloride. In this mechanism, the acid chloride, generated in situ from (S)-2-benzyloxypropionic acid using standard acylating agents like thionyl chloride, undergoes hydrogenolysis over a poisoned palladium catalyst. The barium sulfate support serves to moderate the activity of the palladium, preventing over-reduction of the aldehyde to the corresponding alcohol, a common side reaction in uncatalyzed hydrogenations. This selectivity is paramount for maintaining the integrity of the chiral center adjacent to the carbonyl group. The reaction proceeds under mild hydrogen pressure (0.05-0.15 MPa) in o-xylene or toluene, ensuring that the sensitive benzyloxy protecting group remains intact while efficiently converting the acid chloride to the aldehyde with high fidelity.
Following the formation of the hydrazone intermediate, the stereochemistry is established through a nucleophilic attack by an ethyl Grignard reagent. The patent specifies the use of ethyl magnesium bromide, prepared in situ from bromoethane and magnesium turnings in methyl tert-butyl ether. The presence of the bulky benzyloxy group and the hydrazone moiety directs the incoming ethyl group to the desired face of the molecule, preserving the (2S, 3S) configuration essential for the biological activity of the final Posaconazole API. The use of N,O-bis(trimethylsilyl)acetamide (BSA) as an additive likely facilitates the solubility of the hydrazone and activates the imine bond for nucleophilic attack, ensuring high diastereoselectivity. This mechanistic precision allows for the production of high-purity intermediates without the need for extensive downstream purification.
How to Synthesize N'-[(2S,3S)-2-(benzyloxy)pentan-3-yl]formylhydrazine Efficiently
The execution of this synthesis requires careful control of reaction parameters, particularly during the hydrogenation and Grignard steps, to ensure optimal yield and safety. The process begins with the activation of the carboxylic acid, followed by the critical catalytic reduction which sets the stage for the final carbon-carbon bond formation. Operators must maintain strict temperature controls during the exothermic Grignard addition to prevent racemization or side reactions. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and workup procedures derived from the patent examples, are outlined below to guide process implementation.
- React (S)-2-benzyloxypropionic acid with an acylating agent like thionyl chloride to form the acid chloride.
- Perform Rosenmund reduction using a Pd/BaSO4 catalyst under hydrogen atmosphere to obtain the aldehyde.
- Condense the resulting aldehyde with formylhydrazine to generate the hydrazone intermediate.
- React the hydrazone with ethyl magnesium bromide (Grignard reagent) to finalize the chiral pentyl chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis route offers substantial strategic benefits beyond mere chemical elegance. The elimination of DIBAL-H removes a significant logistical burden, as this reagent requires special shipping classifications and storage protocols due to its pyrophoric nature. By substituting it with hydrogen gas and a heterogeneous catalyst, the facility requirements for production are simplified, leading to lower capital expenditure on safety infrastructure. Furthermore, the ability to recycle the palladium catalyst multiple times without loss of activity directly impacts the cost of goods sold, providing a buffer against fluctuations in precious metal prices. This stability is crucial for long-term supply contracts where price predictability is a key negotiation factor.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive stoichiometric reductants with catalytic hydrogenation. Diisobutylaluminum hydride is a high-cost reagent that generates significant aluminum waste, whereas hydrogen is inexpensive and the palladium catalyst is recoverable. Additionally, the simplified workup procedures, which rely on filtration and extraction rather than chromatography, reduce solvent consumption and labor hours. These factors combine to lower the overall manufacturing cost, allowing for more competitive pricing in the global market for pharmaceutical intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized hazardous reagents. By utilizing commodity chemicals like thionyl chloride, bromoethane, and magnesium, this synthesis route decouples production from the supply constraints of niche reagents. The robustness of the Rosenmund reduction also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent batch-to-batch performance. This reliability is essential for maintaining the uninterrupted production schedules required by downstream API manufacturers, reducing the risk of stockouts or delays in the final drug product launch.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with increasingly stringent regulatory frameworks regarding waste disposal and worker safety. The avoidance of aluminum waste streams simplifies effluent treatment, and the use of recyclable catalysts minimizes heavy metal discharge. The process is designed for scalability, with reaction conditions that are easily transferable from pilot plant to commercial scale reactors. The mild reaction temperatures and pressures further enhance the safety profile, making it easier to obtain regulatory approvals for new manufacturing sites. This environmental and operational compatibility ensures long-term viability and reduces the risk of production shutdowns due to compliance issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this route for industrial production.
Q: How does this new method improve safety compared to traditional DIBAL-H routes?
A: The patented method eliminates the use of diisobutylaluminum hydride (DIBAL-H), a pyrophoric reagent that poses significant fire risks and requires strict anhydrous conditions. Instead, it utilizes a catalytic hydrogenation process which is inherently safer and easier to manage on an industrial scale.
Q: Is the palladium catalyst used in this process recyclable?
A: Yes, the patent explicitly demonstrates that the palladium barium sulfate (Pd/BaSO4) catalyst can be recovered via filtration and reused multiple times without a significant loss in catalytic activity, thereby reducing raw material costs.
Q: What are the purification advantages of this synthetic route?
A: Unlike prior art methods that often require complex column chromatography or difficult recrystallizations, this route relies on simple workup procedures such as filtration, solvent removal, and liquid-liquid extraction, making it highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N'-[(2S,3S)-2-(benzyloxy)pentan-3-yl]formylhydrazine Supplier
At NINGBO INNO PHARMCHEM, we understand that the successful commercialization of complex antifungal agents depends on the reliability and quality of their key intermediates. Our technical team has extensively analyzed the pathway described in CN109796368B and possesses the expertise to execute this advanced synthesis with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N'-[(2S,3S)-2-(benzyloxy)pentan-3-yl]formylhydrazine meets the exacting standards required for GMP API synthesis.
We invite you to collaborate with us to leverage this safer and more efficient synthetic route for your Posaconazole projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and security into your supply chain.
