Scalable Manufacturing of Key Paxlovid Intermediate Formula 1 Using Advanced Sulfur Ylide Chemistry
Scalable Manufacturing of Key Paxlovid Intermediate Formula 1 Using Advanced Sulfur Ylide Chemistry
The global demand for effective antiviral therapeutics has placed immense pressure on the supply chains of critical pharmaceutical intermediates. Patent CN115784968A, published in March 2023, introduces a transformative synthetic methodology for producing (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3,1,0]hexane-2-carboxylic acid methyl ester hydrochloride, widely recognized in the industry as Formula 1. This compound serves as the pivotal chiral scaffold for the synthesis of PF-07321332, the active ingredient in Paxlovid, as well as Boceprevir. The disclosed innovation replaces hazardous legacy chemistries with a robust sulfur ylide-mediated cyclopropanation strategy, offering a pathway that is not only safer but also delivers exceptional yields exceeding 93% overall. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in process chemistry, addressing long-standing issues regarding safety, cost, and scalability in the production of complex bicyclic amino acid derivatives.
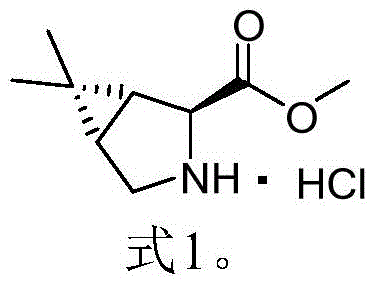
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of Formula 1 relied heavily on phosphorus ylide chemistry or dangerous diazo compounds, presenting severe operational hazards and economic inefficiencies. Traditional routes, such as those described in earlier patent applications like CN202111547312.0, necessitated the use of n-butyllithium, a pyrophoric reagent that poses significant fire risks and requires specialized cryogenic equipment and rigorous safety protocols during large-scale manufacturing. Furthermore, alternative methods utilizing 2,2-dibromopropane suffered from poor atom economy due to the excess bromine content and higher raw material costs compared to mono-brominated analogs. These legacy processes often involved multi-step sequences with cumulative yield losses, where the cyclopropanation step typically hovered around 90-92%, and the overall process was plagued by the complexity of ligand preparation when transition metal catalysts like cobalt dibromide were employed. The reliance on solid-liquid two-phase reactions with zinc powder further complicated reactor design and mixing efficiency, creating bottlenecks for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
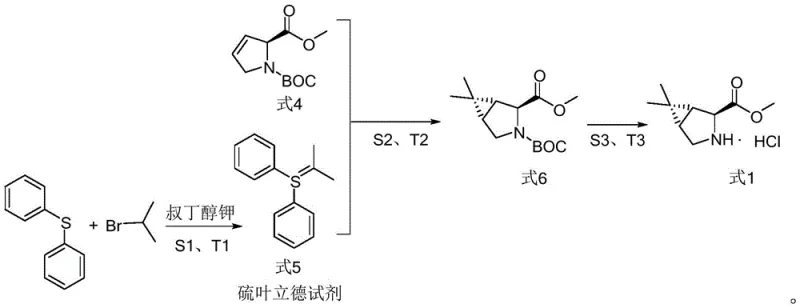
The methodology disclosed in CN115784968A fundamentally reengineers the cyclopropanation step by substituting phosphorus ylides with a sulfur ylide reagent generated in situ under mild conditions. This novel approach utilizes diphenyl sulfide and 2-bromopropane to construct the sulfur ylide precursor (Formula 5), which then reacts with the alkene substrate (Formula 4) to form the bicyclic core (Formula 6). By eliminating the need for n-butyllithium and expensive transition metal catalysts, the new route drastically simplifies the operational workflow and enhances workplace safety. The reaction conditions are optimized to run between -25°C and -20°C, a range that is far more energy-efficient and easier to maintain industrially than the cryogenic temperatures often required for lithiation reactions. Moreover, the final deprotection step utilizes hydrogen chloride gas in methyl tert-butyl ether, achieving yields of over 97%, thereby ensuring that the high purity required for API synthesis is maintained without extensive purification burdens.
Mechanistic Insights into Sulfur Ylide-Mediated Cyclopropanation
The core of this technological breakthrough lies in the generation and reactivity of the sulfur ylide species. In the first stage, diphenyl sulfide undergoes nucleophilic substitution with 2-bromopropane to form a sulfonium salt, which is subsequently deprotonated by a strong base such as potassium tert-butoxide. This generates the reactive sulfur ylide (Formula 5), a neutral species possessing a carbanion character adjacent to a positively charged sulfur atom. Unlike phosphorus ylides which can be unstable or require harsh conditions to generate, this sulfur ylide is formed smoothly in tetrahydrofuran (THF) at controlled low temperatures. The mechanistic advantage here is the precise control over the ylide concentration, which minimizes side reactions such as dimerization or decomposition, thereby preserving the integrity of the chiral centers in the substrate.
Following ylide generation, the cyclopropanation proceeds via a concerted [2+1] cycloaddition mechanism where the carbanion of the ylide attacks the electron-deficient double bond of the pyrroline derivative (Formula 4). The stereochemical outcome is rigorously controlled by the existing chirality of the proline scaffold and the steric bulk of the sulfur substituents, ensuring the formation of the desired (1R,2S,5S) configuration with high diastereoselectivity. The subsequent elimination of dimethyl sulfide drives the equilibrium towards the formation of the cyclopropane ring, resulting in Formula 6. This mechanism avoids the formation of heavy metal waste streams associated with cobalt or copper-catalyzed carbene transfers, aligning with green chemistry principles. The final conversion to Formula 1 involves the acid-mediated removal of the BOC protecting group, where the use of gaseous HCl in MTBE ensures complete deprotection and simultaneous salt formation, crystallizing the product directly from the reaction mixture.
How to Synthesize (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3,1,0]hexane-2-carboxylic acid methyl ester hydrochloride Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding solvent choice and temperature control to maximize the yield advantages demonstrated in the patent data. The process begins with the preparation of the sulfur ylide reagent, where the molar ratio of diphenyl sulfide to 2-bromopropane is critical; experimental data suggests a ratio of 1:2 is optimal to drive the alkylation to completion while minimizing excess reagent waste. The subsequent cyclopropanation step must be conducted in dichloromethane or THF at temperatures strictly maintained between -25°C and -20°C to prevent ylide decomposition and ensure the high yield of over 96% reported in the preferred embodiments. Detailed standardized operating procedures for each reaction stage, including workup and purification protocols, are essential for technology transfer.
- Generate the sulfur ylide reagent (Formula 5) by reacting diphenyl sulfide with 2-bromopropane followed by treatment with potassium tert-butoxide in THF at -25°C to -20°C.
- Perform the cyclopropanation reaction by adding the alkene precursor (Formula 4) to the sulfur ylide solution, maintaining temperatures between -25°C and -20°C to achieve over 96% yield.
- Deprotect the resulting bicyclic ester (Formula 6) using hydrogen chloride gas in methyl tert-butyl ether at 40°C to 50°C to obtain the final hydrochloride salt (Formula 1).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from phosphorus-based to sulfur-based chemistry offers tangible strategic benefits beyond mere yield improvements. The primary advantage is the substantial reduction in raw material costs and hazard management expenses. By replacing expensive and atom-inefficient reagents like 2,2-dibromopropane with commodity chemicals like 2-bromopropane and diphenyl sulfide, the direct material cost of goods sold (COGS) is significantly lowered. Furthermore, the elimination of pyrophoric reagents like n-butyllithium removes the need for specialized storage facilities and complex safety infrastructure, leading to reduced capital expenditure and lower insurance premiums for manufacturing sites.
- Cost Reduction in Manufacturing: The new process eliminates the requirement for expensive transition metal catalysts such as cobalt dibromide and complex ligands, which are not only costly to purchase but also require rigorous removal steps to meet residual metal specifications in pharmaceutical products. By utilizing a metal-free organocatalytic approach for the key bond-forming step, the downstream purification process is drastically simplified, reducing solvent consumption and waste disposal costs. The high overall yield of over 93% means that less starting material is required to produce the same amount of final API intermediate, directly enhancing the economic efficiency of the production line and allowing for more competitive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals like diphenyl sulfide and 2-bromopropane mitigates the risk of supply disruptions often associated with specialized fine chemical reagents. Since these precursors are produced on a massive scale for various industrial applications, their availability is stable, and price volatility is minimal compared to niche pharmaceutical building blocks. Additionally, the robustness of the reaction conditions, which tolerate a broader range of temperatures and solvent grades compared to sensitive organolithium reactions, ensures consistent production output even when minor variations in raw material quality occur, thereby securing the continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is vastly superior for large-scale operations. The absence of heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing facility, aiding in compliance with increasingly stringent global environmental regulations. The use of common solvents like THF, DCM, and MTBE allows for efficient solvent recovery and recycling systems to be implemented, further reducing the volume of hazardous waste generated. The mild reaction temperatures reduce energy consumption for cooling, contributing to a more sustainable manufacturing profile that aligns with the corporate social responsibility goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and comparative analysis provided in patent CN115784968A, offering clarity on how this method compares to established industry standards. Understanding these nuances is critical for technical teams evaluating process feasibility and for commercial teams assessing the long-term viability of sourcing intermediates produced via this technology.
Q: Why is the sulfur ylide method superior to traditional phosphorus ylide routes for Formula 1?
A: The sulfur ylide method eliminates the need for dangerous pyrophoric reagents like n-butyllithium required for phosphorus ylide generation. It operates under milder conditions (-25°C vs cryogenic) and uses cheaper, more atom-economical starting materials like 2-bromopropane instead of expensive 2,2-dibromopropane.
Q: What is the overall yield of the new synthesis method described in CN115784968A?
A: The optimized process achieves a total yield of over 93% for Formula 1. Specifically, the cyclopropanation step yields over 96%, and the final deprotection step yields over 97%, significantly improving upon prior art methods.
Q: Is this process suitable for large-scale commercial production of antiviral intermediates?
A: Yes, the process is designed for industrial scalability. It avoids hazardous reagents that complicate safety protocols at scale, utilizes common solvents like THF and MTBE, and demonstrates robust yield consistency across different temperature and solvent optimization studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3,1,0]hexane-2-carboxylic acid methyl ester hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is paramount for the future of antiviral drug manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN115784968A are fully realized in practical, GMP-compliant manufacturing environments. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature controls required for sulfur ylide chemistry, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of Formula 1 meets the exacting standards required for Pfizer's Paxlovid and other critical antiviral therapies.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic impact of switching to this sulfur ylide route for your specific volume requirements. We encourage you to contact us today to discuss route feasibility assessments and to obtain specific COA data for our pilot batches, ensuring that your project timelines are met with the highest quality intermediates available in the market.
