Advanced Catalytic Synthesis of Bioactive Chiral Chroman Compounds for Commercial Drug Development
Advanced Catalytic Synthesis of Bioactive Chiral Chroman Compounds for Commercial Drug Development
The pharmaceutical industry is constantly seeking robust and scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN112920173A introduces a significant breakthrough in the asymmetric synthesis of chiral chroman compounds, a privileged structural motif found in numerous bioactive natural products and synthetic drugs. These compounds, characterized by the general structure shown in Formula 1, have demonstrated potent cytotoxic activity against a spectrum of aggressive adenocarcinoma cell lines, including HepG2, MDA-MB-231, and SGC-7901. 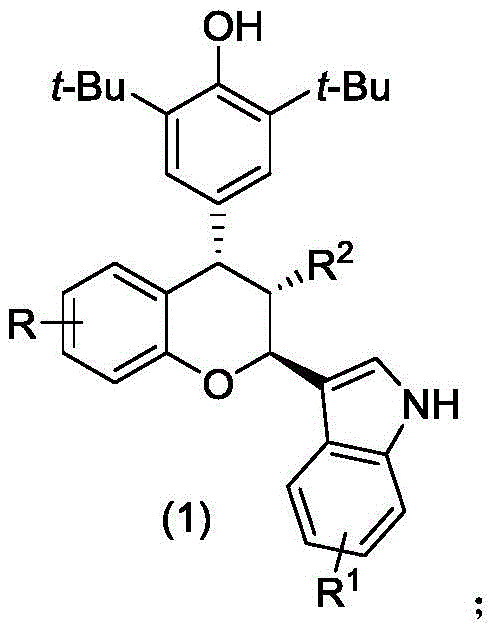 . For R&D directors and procurement specialists, this patent represents a vital opportunity to access high-purity intermediates for oncology pipelines through a process that balances stereochemical precision with operational simplicity.
. For R&D directors and procurement specialists, this patent represents a vital opportunity to access high-purity intermediates for oncology pipelines through a process that balances stereochemical precision with operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral chroman frameworks has relied heavily on transition metal catalysis or stoichiometric chiral auxiliaries, which often present significant drawbacks for industrial application. Traditional routes frequently require harsh reaction conditions, such as elevated temperatures or strongly acidic environments, which can lead to the decomposition of sensitive functional groups and the formation of complex impurity profiles. Furthermore, many legacy processes suffer from poor atom economy and generate substantial heavy metal waste, creating bottlenecks in purification and increasing the environmental burden of manufacturing. The inability to consistently achieve high enantiomeric excess without extensive recycling or resolution steps has long been a pain point for supply chain managers aiming to reduce cost of goods sold (COGS) while maintaining rigorous quality standards.
The Novel Approach
The methodology disclosed in CN112920173A offers a transformative alternative by employing chiral phosphoric acid organocatalysis to drive the [4+2] cycloaddition between p-methylene benzoquinones and 3-indolenines. This approach operates under remarkably mild conditions, typically utilizing dichloromethane as a solvent at temperatures as low as -30°C, thereby preserving the integrity of delicate substrates. 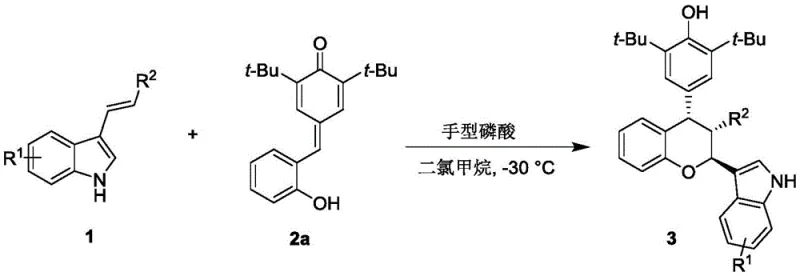 . The versatility of this protocol is evidenced by its tolerance to a wide array of substituents on both the quinone and indole rings, allowing for the rapid generation of diverse chemical libraries. By eliminating the need for transition metals, this novel route simplifies the downstream processing workflow, removing the costly and time-consuming steps associated with metal scavenging and residual metal testing.
. The versatility of this protocol is evidenced by its tolerance to a wide array of substituents on both the quinone and indole rings, allowing for the rapid generation of diverse chemical libraries. By eliminating the need for transition metals, this novel route simplifies the downstream processing workflow, removing the costly and time-consuming steps associated with metal scavenging and residual metal testing.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cycloaddition
The success of this synthesis hinges on the unique ability of the chiral phosphoric acid catalyst to function as a dual hydrogen-bond donor and acceptor, creating a highly organized chiral environment within the transition state. The catalyst, often a BINOL-derived phosphoric acid substituted with bulky groups like 9-anthracenyl (as depicted in the catalyst structure), engages the carbonyl oxygen of the p-methylene benzoquinone and the nitrogen or pi-system of the 3-indolenine simultaneously. 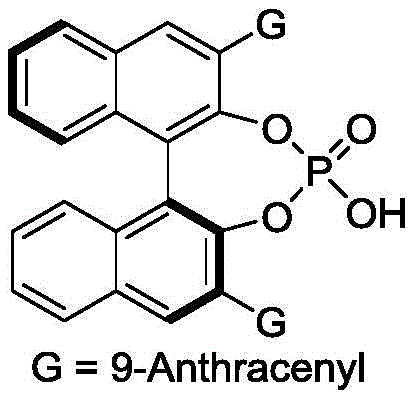 . This bifunctional activation lowers the energy barrier for the cycloaddition while rigidly locking the substrates into a specific orientation that favors the formation of one enantiomer over the other. The steric bulk of the catalyst's 3,3'-substituents plays a critical role in shielding one face of the reactive intermediate, ensuring that the nucleophilic attack occurs with high facial selectivity.
. This bifunctional activation lowers the energy barrier for the cycloaddition while rigidly locking the substrates into a specific orientation that favors the formation of one enantiomer over the other. The steric bulk of the catalyst's 3,3'-substituents plays a critical role in shielding one face of the reactive intermediate, ensuring that the nucleophilic attack occurs with high facial selectivity.
From an impurity control perspective, this mechanism is exceptionally clean because it avoids radical pathways or high-energy intermediates that typically lead to polymerization or side reactions. The reaction proceeds through a concerted or stepwise ionic pathway that is tightly controlled by the catalyst's chiral pocket, resulting in products with excellent diastereomeric ratios (dr) often exceeding 85:15 and enantiomeric ratios (er) reaching up to 88:12 in optimized examples. This high level of stereocontrol minimizes the formation of unwanted stereoisomers, significantly reducing the burden on purification teams and ensuring that the final active pharmaceutical ingredient (API) precursor meets stringent regulatory specifications for chiral purity without the need for preparative chiral HPLC.
How to Synthesize Chiral Chroman Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and temperature controls to maximize yield and selectivity. The process begins with the activation of molecular sieves to ensure anhydrous conditions, followed by the sequential addition of the p-methylene benzoquinone and 3-indolenine substrates in dichloromethane. The reaction is initiated by the addition of the chiral phosphoric acid catalyst (typically 10 mol%) and maintained at -30°C with vigorous stirring to ensure homogeneous mixing and heat transfer.
- Prepare the reaction mixture by combining p-methylene benzoquinone and 3-indolene substrates in dichloromethane solvent with activated molecular sieves.
- Add the chiral phosphoric acid catalyst (e.g., BINOL derivative) and stir the reaction at -30°C until TLC indicates completion.
- Filter the mixture to remove desiccants, concentrate the filtrate, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route offers compelling economic and logistical benefits that directly impact the bottom line. The shift away from precious metal catalysts to organic phosphoric acids eliminates the volatility risks associated with rhodium, palladium, or iridium pricing, stabilizing raw material costs over the long term. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint and aligning with modern sustainability goals.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavengers and the associated analytical testing for residual metals, which are costly and time-intensive steps in traditional API manufacturing. The simplified workup procedure, involving basic filtration and concentration, drastically reduces solvent usage and labor hours compared to multi-step extraction protocols. Furthermore, the high atom economy of the cycloaddition reaction ensures that a greater proportion of starting materials are converted into the desired product, minimizing waste disposal costs and maximizing material efficiency.
- Enhanced Supply Chain Reliability: The substrates required for this synthesis, such as substituted phenols and indoles, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, leading to more consistent batch-to-batch performance and reliable delivery schedules. This stability allows for better inventory planning and reduces the likelihood of production delays caused by failed batches or difficult purifications.
- Scalability and Environmental Compliance: The use of dichloromethane, while requiring proper handling, is a well-understood solvent in the industry with established recovery and recycling infrastructure, facilitating easy scale-up from gram to ton quantities. The absence of heavy metals simplifies the regulatory filing process and wastewater treatment requirements, as the effluent does not require specialized treatment for toxic metal removal. This environmental compatibility accelerates the timeline for regulatory approval and supports the company's commitment to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these chiral chroman derivatives. Understanding these details is crucial for integrating this technology into existing drug discovery and development workflows effectively.
Q: What is the biological significance of these chiral chroman compounds?
A: According to patent CN112920173A, these compounds exhibit strong cytotoxic activity against various adenocarcinoma cell lines, including HepG2 liver cancer, MDA-MB-231 breast cancer, and SGC-7901 gastric cancer cells, making them valuable leads for oncology drug discovery.
Q: How does the chiral phosphoric acid catalyst improve the synthesis?
A: The chiral phosphoric acid acts as a bifunctional organocatalyst, activating both the electrophile and nucleophile through hydrogen bonding networks. This dual activation ensures high stereocontrol, resulting in excellent diastereomeric ratios (dr) and enantiomeric ratios (er) without the need for toxic transition metals.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the method utilizes mild reaction conditions (-30°C), common solvents like dichloromethane, and avoids hazardous reagents. The simple workup procedure involving filtration and standard chromatography makes it highly amenable to commercial scale-up and industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Chroman Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the race to develop new oncology therapies. Our team of expert chemists has extensively analyzed the methodology described in CN112920173A and is fully prepared to support your projects from milligram-scale screening to commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the enantiomeric excess required for clinical trials.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis to your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new analogues, we are ready to provide the data-driven insights you need. Partner with us to leverage this advanced catalytic technology and accelerate your path to market with confidence.
