Advanced Fluorinated Iridium Complexes: Revolutionizing OLED Phosphorescent Material Manufacturing
Advanced Fluorinated Iridium Complexes: Revolutionizing OLED Phosphorescent Material Manufacturing
The landscape of organic light-emitting diode (OLED) technology is continuously evolving, driven by the relentless demand for higher efficiency and longer device lifetimes. A pivotal advancement in this domain is documented in Chinese Patent CN103450891A, which discloses a novel class of iridium complex phosphorescent materials utilizing fluorinated pyridine carboxylic acid as an auxiliary ligand. This innovation addresses critical bottlenecks in electrophosphorescent material design by enhancing quantum efficiency and thermal stability through strategic ligand modification. By replacing traditional auxiliary ligands with fluorinated derivatives, the invention achieves a significant boost in luminescent performance while maintaining a chemically stable octahedral structure centered on the iridium atom. For industry leaders seeking a reliable OLED material supplier, understanding the structural nuances and synthetic advantages of these (C^N)2IrLX type complexes is essential for next-generation display development.
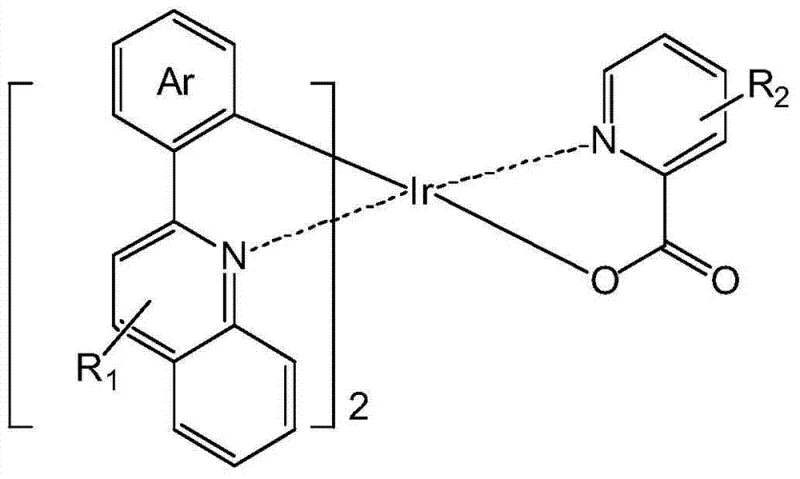
The general formula presented in the patent illustrates the versatility of this chemical platform, where the auxiliary ligand LX incorporates fluorine atoms on the pyridine ring. This specific structural modification is not merely cosmetic; it fundamentally alters the electronic environment of the metal center, leading to improved intersystem crossing rates and reduced non-radiative decay pathways. The ability to tune emission colors across the visible spectrum by varying the aryl groups on the main ligand, combined with the efficiency gains from the fluorinated auxiliary ligand, positions this technology as a cornerstone for high-performance full-color display applications. As we delve deeper into the technical specifics, it becomes clear why this approach represents a substantial leap forward in electronic chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of phosphorescent iridium complexes has been hindered by the trade-off between synthetic complexity and optoelectronic performance. Traditional neutral complexes such as Ir(C^N)3, while efficient, often suffer from structural rigidity and difficult preparation protocols that limit their scalability. Furthermore, earlier iterations of (C^N)2IrLX complexes utilizing standard picolinic acid or acetylacetone auxiliary ligands frequently exhibited suboptimal quantum efficiencies and thermal instability under device operating conditions. Literature precedents indicate that modifying the main ligand with bulky groups often resulted in decreased photon quantum efficiency, while attempts to modify auxiliary ligands with methyl or phenyl groups showed negligible improvements. Additionally, ionic iridium complexes, another alternative, were plagued by high melting points that made vacuum deposition—a standard process in OLED fabrication—extremely difficult, thereby restricting their commercial viability and application scope in mass production environments.
The Novel Approach
The methodology outlined in CN103450891A circumvents these historical limitations by strategically incorporating fluorine atoms into the auxiliary ligand structure. This novel approach leverages the strong electron-withdrawing nature of fluorine to stabilize the excited states of the iridium complex, resulting in a marked increase in fluorescence intensity and phosphorescence quantum efficiency. Unlike previous attempts where ligand modifications led to efficiency drops, the introduction of fluorinated picolinic acid derivatives has been shown to elevate quantum efficiency values significantly, outperforming both non-fluorinated analogues and standard reference materials like Ru(bpy)3. This strategy not only enriches the library of available phosphorescent materials but also simplifies the synthetic route by utilizing commercially available fluorinated precursors. Consequently, this method offers a robust pathway for cost reduction in electronic chemical manufacturing by minimizing synthetic steps and maximizing yield purity without compromising on the critical photophysical properties required for high-end display technologies.
Mechanistic Insights into Fluorinated Ligand Coordination
The core of this technological breakthrough lies in the precise coordination chemistry between the iridium center and the fluorinated auxiliary ligands. The synthesis proceeds through a well-defined two-step mechanism that ensures high regioselectivity and product purity. Initially, the cyclometalation of the 2-arylquinoline derivative with iridium chloride forms a stable dichloro-bridged dimer intermediate. This step is crucial as it establishes the primary coordination sphere and defines the geometric framework of the final complex. The subsequent ligand exchange reaction involves the displacement of the chloride bridges by the bidentate fluorinated picolinic acid ligand. The presence of fluorine atoms on the pyridine ring enhances the acidity of the carboxylic acid group and modifies the electron density distribution, facilitating a more favorable coordination equilibrium. This mechanistic pathway ensures that the final (C^N)2IrLX complex maintains a rigid octahedral geometry, which is instrumental in suppressing non-radiative vibrational relaxation and thus enhancing the overall phosphorescence lifetime and efficiency.
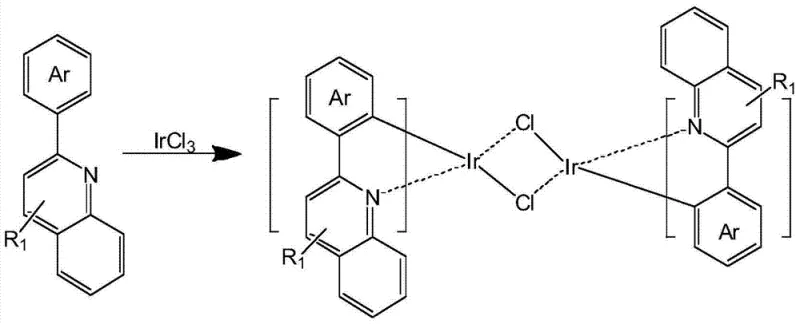
Furthermore, the impurity control mechanism inherent in this synthetic route is highly effective for industrial applications. The use of specific solvent systems, such as ethylene glycol ether during the dimer formation and dichloromethane/ethanol mixtures during the ligand exchange, allows for precise control over reaction kinetics and solubility profiles. This control minimizes the formation of side products such as homoleptic species or partially substituted intermediates, which are common pitfalls in organometallic synthesis. The patent data indicates that the resulting complexes exhibit sharp emission peaks and high color purity, attributes that are directly linked to the homogeneity of the molecular structure achieved through this controlled coordination process. By understanding these mechanistic details, R&D teams can better optimize reaction conditions to further push the boundaries of efficiency and stability in OLED emitter design.
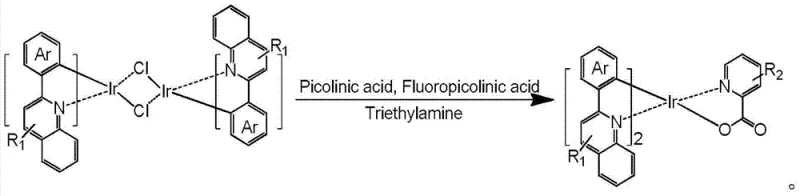
How to Synthesize (DPQ)2Ir(5FPic) Efficiently
The practical implementation of this patented technology involves a streamlined protocol designed for reproducibility and scalability. The synthesis targets specific high-performance emitters such as (DPQ)2Ir(5FPic), where DPQ stands for 2,4-diphenylquinoline. The process begins with the rigorous preparation of the iridium dimer precursor, followed by a mild ligand exchange under basic conditions. This approach avoids the harsh conditions often required for C-H activation, thereby preserving the integrity of sensitive functional groups on the ligands. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below, which outline the exact molar ratios, temperature controls, and purification techniques necessary to achieve the high purity specifications demanded by the optoelectronics industry.
- Synthesize the iridium dichloro-bridged dimer by reacting IrCl3 with 2-arylquinoline derivatives in ethylene glycol ether at 110°C under nitrogen protection.
- React the purified dichloro-bridged dimer with fluorinated picolinic acid auxiliary ligands in dichloromethane and ethanol.
- Add triethylamine as a base and stir at room temperature for 12 hours, followed by solvent removal and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this fluorinated iridium complex technology offers distinct strategic advantages over legacy materials. The primary benefit stems from the simplification of the raw material sourcing strategy. Since the key auxiliary ligands—fluorinated pyridine carboxylic acids—are commercially available off-the-shelf, manufacturers can bypass the capital-intensive and time-consuming processes associated with custom ligand synthesis. This direct sourcing capability drastically reduces the lead time for high-purity electronic chemicals and mitigates the risks associated with supply chain disruptions for specialized intermediates. Moreover, the synthetic route described in the patent generates fewer by-products, which translates to simplified downstream processing and reduced waste disposal costs. These factors collectively contribute to a more resilient and cost-effective supply chain model for OLED material production.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the elimination of complex ligand synthesis steps. By utilizing commercially purchasable fluorinated precursors, the overall bill of materials is optimized, and the labor hours required for multi-step organic synthesis are substantially reduced. Additionally, the high selectivity of the reaction minimizes the loss of expensive iridium metal to side products, ensuring that the precious metal utilization rate is maximized. This efficiency gain is critical for maintaining competitive pricing in the volatile market of display materials, allowing manufacturers to achieve substantial cost savings without sacrificing the quality or performance of the final phosphorescent emitter.
- Enhanced Supply Chain Reliability: Reliance on commodity-grade fluorinated building blocks rather than bespoke synthesized ligands strengthens the supply chain against potential bottlenecks. The availability of these precursors from multiple global suppliers ensures continuity of supply, which is paramount for meeting the rigorous production schedules of panel manufacturers. Furthermore, the robustness of the synthetic method, which operates under relatively mild conditions and uses common organic solvents, reduces the dependency on specialized equipment or hazardous reagents that might face regulatory scrutiny. This operational flexibility ensures that production can be scaled up rapidly to meet surging demand, providing a reliable OLED material supplier with the agility needed in a fast-paced market.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing process is favorably low compared to traditional methods. The reduction in synthetic steps inherently lowers the consumption of energy and solvents, aligning with green chemistry principles. The ease of purification via standard column chromatography or recrystallization techniques means that solvent recovery and recycling can be implemented effectively, further reducing waste generation. As regulatory pressures on chemical manufacturing intensify globally, adopting a process that naturally generates fewer hazardous by-products and utilizes stable, manageable reagents positions companies favorably for long-term compliance. This scalability ensures that the transition from laboratory gram-scale to commercial ton-scale production can be executed smoothly and sustainably.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of fluorinated iridium complexes in OLED architectures. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these aspects helps stakeholders evaluate the feasibility of integrating these materials into existing production lines and product roadmaps. The answers reflect the consensus on performance metrics and process reliability observed during the development of these advanced phosphorescent systems.
Q: How does fluorine substitution affect the quantum efficiency of iridium complexes?
A: According to patent CN103450891A, introducing fluorine atoms onto the pyridine carboxylic acid auxiliary ligand significantly enhances the room temperature phosphorescence quantum efficiency, reaching up to 0.15 compared to 0.033 for non-fluorinated analogues.
Q: What is the thermal stability profile of these new iridium materials?
A: Thermogravimetric analysis indicates that these fluorinated iridium complexes exhibit excellent thermal stability, with significant mass loss typically occurring only around 300°C, making them suitable for vacuum deposition processes.
Q: Are the auxiliary ligands commercially available for large-scale production?
A: Yes, the patent highlights that the fluorinated pyridine carboxylic acids used as auxiliary ligands are commercially purchasable, which eliminates complex synthesis steps for the ligand itself and simplifies the overall supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iridium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the fluorinated iridium complex technology described in CN103450891A for the future of display manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to mass market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for electronic grade materials. We understand that consistency is key in the semiconductor and display industries, and our dedicated process engineering team works tirelessly to optimize every batch for maximum quantum efficiency and thermal stability, delivering the high-purity OLED material performance your devices demand.
We invite you to collaborate with us to leverage these advanced synthetic routes for your next-generation products. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to deep technical expertise and a supply chain committed to excellence. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can accelerate your time-to-market while optimizing your overall production costs.
