Advanced Manufacturing of (S)-4-(4-Aminobenzyl)-2-Oxazolidinone for Zomitriptan Production
Advanced Manufacturing of (S)-4-(4-Aminobenzyl)-2-Oxazolidinone for Zomitriptan Production
The pharmaceutical landscape for migraine treatment relies heavily on the efficient production of key chiral intermediates, specifically (S)-4-(4-aminobenzyl)-2-oxazolidinone, which serves as the critical backbone for the synthesis of Zomitriptan. As detailed in patent CN101289430A, a novel synthetic methodology has been developed that fundamentally restructures the production pathway to enhance safety, yield, and environmental sustainability. This technical breakthrough addresses the longstanding industry challenge of balancing high-volume manufacturing with rigorous safety standards, particularly by eliminating the reliance on hazardous reagents like phosgene. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this four-step sequence is essential for securing a stable supply chain. The process begins with the abundant and cost-effective starting material, L-Phenylalanine, and proceeds through a series of highly controlled transformations including reduction, cyclization, nitration, and final hydrogenation. By integrating strict crystallization protocols at every stage, this method ensures that the final product possesses the optical purity and chemical integrity required for sensitive downstream API synthesis, thereby mitigating the risks associated with impurity propagation in complex drug manufacturing.
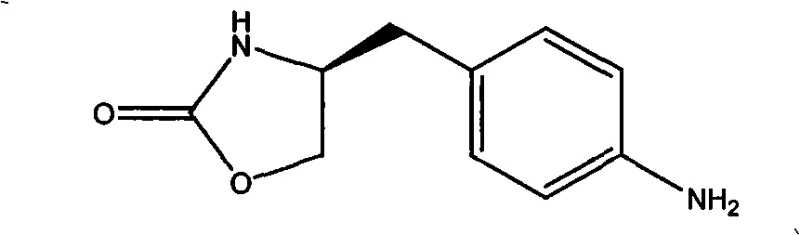
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of this oxazolidinone derivative has been plagued by significant safety hazards and inefficient material throughput. The pioneering route disclosed by Wellcome (WO 91/18897), while chemically valid, necessitates the use of phosgene gas, a substance classified as extremely toxic and requiring specialized, high-cost containment infrastructure to handle safely. Furthermore, this legacy process involves a cumbersome five-step sequence starting from L-Phenylalanine, which inherently compounds yield losses at each transformation stage. Documentation indicates that the cumulative yield of this traditional pathway is dismally low, reported at approximately 16%, rendering it economically unviable for large-scale commercial operations where margin compression is a constant threat. Additionally, alternative methods such as those reported by AstraZeneca (WO 97/06162) attempted to streamline the process but often resulted in 'one-pot' reactions that compromised the purity profile of the intermediate. These purity issues are critical because residual impurities can interfere with the subsequent coupling reactions required to build the final Zomitriptan molecule, leading to costly rework or batch rejection. The reliance on hazardous reagents and the inability to consistently deliver high-purity material without extensive purification makes these conventional methods unsuitable for modern, green chemistry-focused manufacturing environments.
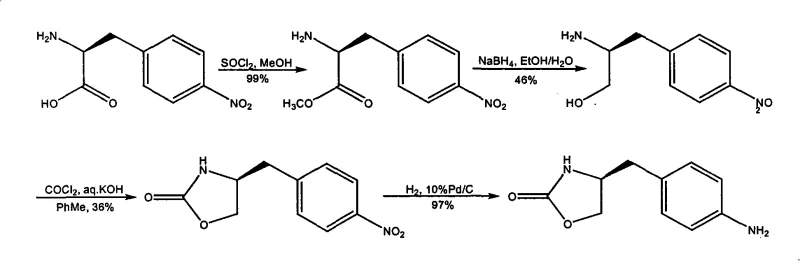
The Novel Approach
In stark contrast to the hazardous legacy pathways, the innovative method described in CN101289430A introduces a streamlined four-step protocol that prioritizes operator safety and process efficiency without sacrificing yield. The most significant advancement is the complete elimination of phosgene; instead, the cyclization step utilizes vinyl chloroformate, a significantly safer and more manageable reagent that facilitates the formation of the oxazolidinone ring under mild conditions. This strategic substitution not only reduces the regulatory burden and safety equipment costs associated with toxic gas handling but also simplifies the reactor setup, allowing for more flexible commercial scale-up of complex pharmaceutical intermediates. Moreover, the new route incorporates a rigorous purification strategy where the product of each step is isolated via crystallization before proceeding to the next reaction. This 'stop-and-purify' approach effectively breaks the chain of impurity carryover, ensuring that the final amine product is of exceptional quality. By reducing the step count from five to four and dramatically improving the yield at each stage—achieving up to 95% in the reduction step and 89% in the cyclization—the overall material efficiency is vastly superior. This approach represents a paradigm shift towards sustainable manufacturing, aligning perfectly with the goals of cost reduction in API manufacturing by minimizing waste generation and maximizing the output per kilogram of raw material input.
Mechanistic Insights into the Four-Step Synthetic Sequence
The success of this novel synthesis lies in the precise control of reaction conditions and the selection of reagents that favor high chemoselectivity. The first step involves the reduction of L-Phenylalanine to L-phenylpropyl amino alcohol using sodium borohydride activated by sulfuric acid in a THF solvent system. This reduction is conducted at a tightly controlled temperature of 0 ± 3 °C to prevent side reactions, followed by a hydrolysis step at 90–100 °C to decompose excess borohydride and facilitate product isolation. The second step, the formation of the oxazolidinone core, employs vinyl chloroformate in the presence of sodium methylate. This reaction proceeds through a carbamate intermediate which spontaneously cyclizes upon heating to 85 ± 5 °C, releasing acetaldehyde as a benign byproduct rather than toxic gases. The third step introduces the necessary functionality for the final drug molecule via electrophilic aromatic substitution, using 95% nitric acid to nitrate the para-position of the benzyl ring. Finally, the nitro group is reduced to the primary amine using catalytic hydrogenation with 5% Pd/C in methanol. This final reduction is clean and high-yielding (85-95%), completing the transformation with high stereochemical fidelity. The mechanistic elegance of this route ensures that the chiral center derived from the natural amino acid is preserved throughout the sequence, delivering the required (S)-enantiomer with high optical purity.
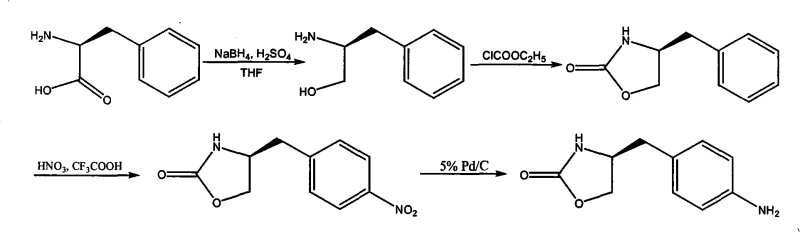
Impurity control is arguably the most critical aspect of this process for ensuring downstream success in Zomitriptan synthesis. In many 'one-pot' methodologies, byproducts from early stages, such as unreacted starting materials or over-reduced species, can persist and complicate the final purification. The patented method mitigates this risk by enforcing a crystallization step after every chemical transformation. For instance, after the initial reduction, the amino alcohol is recrystallized from an ethyl acetate and petroleum ether mixture, removing inorganic salts and boron residues. Similarly, the intermediate (S)-4-benzyl-2-oxazolidinone is purified via crystallization before nitration, ensuring that the nitration reaction occurs on a pure substrate, which minimizes the formation of ortho-nitro isomers or dinitro byproducts. This disciplined approach to purification means that the final hydrogenation step starts with a high-quality nitro precursor, resulting in a final amine product that requires no further purification before being used in the next stage of drug synthesis. For quality assurance teams, this translates to a much narrower impurity profile and simplified analytical testing, reducing the time and cost associated with release testing and stability studies.
How to Synthesize (S)-4-(4-Aminobenzyl)-2-Oxazolidinone Efficiently
The implementation of this synthetic route requires careful attention to temperature control and stoichiometry, particularly during the exothermic reduction and nitration phases. The process is designed to be robust, utilizing common solvents like THF, methanol, and ethyl acetate, which are easily recovered and recycled in a standard chemical plant. The use of vinyl chloroformate instead of phosgene simplifies the engineering controls needed, allowing the reaction to be performed in standard glass-lined or stainless steel reactors without the need for specialized gas scrubbing systems required for phosgene. Each step is optimized for maximum recovery, with specific solvent ratios (e.g., ethyl acetate to petroleum ether at 1:1 to 1:2) defined to ensure optimal crystal growth and purity. The detailed standardized operating procedures for scaling this chemistry from laboratory to pilot plant are outlined below, providing a clear roadmap for technical teams looking to adopt this superior manufacturing process.
- Reduction of L-Phenylalanine to L-phenylpropyl amino alcohol using sodium borohydride and sulfuric acid in THF.
- Cyclization with vinyl chloroformate and sodium methylate to form (S)-4-benzyl-2-oxazolidinone.
- Nitration using 95% nitric acid followed by catalytic hydrogenation to obtain the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers substantial strategic advantages beyond mere technical superiority. The primary benefit is the drastic simplification of the supply chain risk profile by removing the dependency on phosgene, a reagent that is heavily regulated and often subject to supply disruptions due to safety incidents or transport restrictions. By switching to vinyl chloroformate and standard mineral acids, the manufacturing process becomes more resilient and easier to source, ensuring reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the significant improvement in overall yield directly correlates to a lower cost of goods sold (COGS). When a process improves from a 16% total yield to a multi-step sequence with individual yields exceeding 80-90%, the amount of raw material required to produce one kilogram of final product drops precipitously. This material efficiency translates into significant cost savings that can be passed down the supply chain or retained as margin. Additionally, the environmental footprint of the process is markedly reduced; the elimination of toxic gas emissions and the reduction of waste volume align with increasingly stringent global environmental regulations, reducing the risk of regulatory fines or shutdowns. This makes the supplier a more stable and compliant partner for long-term contracts.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive safety infrastructure required for phosgene handling and the dramatic increase in material throughput. By avoiding the 84% material loss associated with the old 16% yield route, manufacturers can achieve a much higher output per batch, effectively lowering the unit cost of the intermediate. The use of recyclable solvents and the ability to recover reagents further contributes to a leaner cost structure, making this a highly competitive option for price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Sourcing L-Phenylalanine is straightforward as it is a commodity fermentation product with a stable global supply, unlike specialized or hazardous reagents that may have single-source suppliers. The robustness of the four-step sequence, with its built-in purification checkpoints, ensures consistent batch-to-batch quality, reducing the likelihood of supply interruptions caused by failed quality control tests. This reliability is crucial for pharmaceutical companies managing tight production schedules for finished dosage forms, as it guarantees a steady flow of critical intermediates without unexpected delays.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids unit operations that are difficult to enlarge, such as gas-liquid reactions with toxic gases. The liquid-phase reactions and crystallizations are standard unit operations that scale linearly from kilograms to tons. Moreover, the 'green' nature of the process, characterized by the absence of persistent toxic pollutants and lower waste generation, facilitates easier permitting and operation in regions with strict environmental oversight. This ensures long-term operational continuity and protects the buyer's reputation by associating their supply chain with sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this key pharmaceutical intermediate. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing production workflows and assess the potential for process optimization.
Q: Why is this new synthetic route preferred over the traditional phosgene method?
A: The new route eliminates the use of highly toxic phosgene gas, significantly improving operational safety and environmental compliance while maintaining high product purity through intermediate crystallization.
Q: What is the overall yield advantage of this four-step process?
A: Unlike the conventional five-step route which reports a total yield of only 16%, this optimized four-step sequence achieves significantly higher individual step yields, ranging from 76% to 95%, resulting in superior material efficiency.
Q: How does the process ensure high purity for downstream Zomitriptan synthesis?
A: The protocol mandates a crystallization purification step after every reaction stage, preventing the accumulation of impurities common in 'one-pot' methods and ensuring the final intermediate meets stringent quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-(4-Aminobenzyl)-2-Oxazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient and safer synthetic route is critical for maintaining competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel four-step synthesis are fully realized at an industrial level. We are committed to delivering stringent purity specifications for every batch, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity, assay, and impurity profiles. Our facility is equipped to handle the specific requirements of this chemistry, including the safe handling of nitration reactions and the precise temperature control needed for the reduction steps, guaranteeing a supply of high-quality intermediate that meets the exacting standards of the Zomitriptan manufacturing process.
We invite you to collaborate with us to optimize your supply chain for this critical migraine medication intermediate. Our technical sales team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to our manufacturing process. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By partnering with us, you secure not just a chemical supplier, but a strategic ally dedicated to enhancing your production efficiency and product quality through superior chemical engineering.
