Advanced Catalytic Reduction for High-Purity Zolmitriptan Intermediates and Commercial Scale-Up
Advanced Catalytic Reduction for High-Purity Zolmitriptan Intermediates and Commercial Scale-Up
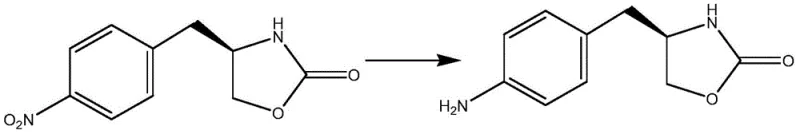
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for synthesizing critical active pharmaceutical ingredient (API) precursors. Patent CN110003129B introduces a significant technological advancement in the preparation of zolmitriptan intermediates, specifically focusing on the reduction of (S)-4-(4-nitrobenzyl)-2-oxazolidinone to its corresponding amine derivative. This innovation addresses long-standing challenges in process chemistry by replacing hazardous reagents with a greener catalytic system utilizing ammonium formate and palladium carbon. For R&D directors and procurement specialists, this shift represents not merely a chemical modification but a strategic improvement in supply chain reliability and operational safety. The method ensures high conversion rates while simplifying post-reaction processing, which is essential for maintaining cost-efficiency in large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-4-(4-aminobenzyl)-2-oxazolidinone relied heavily on reduction protocols involving ferric chloride hexahydrate and hydrazine hydrate. These traditional methodologies present severe operational drawbacks that complicate industrial scale-up and increase environmental liabilities. The use of iron salts inevitably generates substantial quantities of iron mud, a viscous waste byproduct that is notoriously difficult to separate via centrifugation, leading to significant solid waste disposal issues. Furthermore, the reliance on hydrazine hydrate introduces critical safety risks; spent activated carbon contaminated with residual hydrazine poses a spontaneous combustion hazard when exposed to air, necessitating specialized handling and storage protocols. To maintain catalytic activity, processes often require a large excess of hydrazine to compensate for self-decomposition, driving up raw material costs and complicating purification steps due to the presence of nitrogenous impurities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a transfer hydrogenation strategy employing ammonium formate as a hydrogen donor and palladium carbon as a heterogeneous catalyst. This methodology operates under mild thermal conditions, typically between 50°C and 60°C, significantly reducing energy consumption compared to more vigorous reduction techniques. The reaction mechanism avoids the formation of inorganic sludge entirely, resulting in a much cleaner reaction profile that facilitates easier downstream processing. By eliminating the need for hazardous hydrazine derivatives, the process inherently improves workplace safety and reduces the regulatory burden associated with handling toxic reagents. The simplicity of the workup, involving mere filtration and crystallization, allows for a streamlined production workflow that is highly attractive for commercial manufacturing.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthetic breakthrough lies in the efficient transfer hydrogenation mechanism facilitated by the palladium catalyst. In this system, ammonium formate serves as the source of hydrogen, decomposing on the surface of the palladium carbon to generate reactive hydrogen species in situ. This nascent hydrogen immediately reduces the nitro group on the benzyl ring to an amino group without affecting the sensitive oxazolidinone ring structure. The efficiency of this reduction is heavily dependent on the adsorption characteristics of ammonium formate onto the catalyst surface, ensuring that hydrogen transfer occurs selectively at the nitro functionality. This selectivity is paramount for preserving the chiral integrity of the (S)-enantiomer, preventing racemization which could compromise the biological activity of the final zolmitriptan drug product.
From an impurity control perspective, the absence of transition metal salts like iron chloride drastically simplifies the purification landscape. Traditional iron-based reductions often leave trace metal residues that require complex chelation or washing steps to meet pharmaceutical grade standards. The heterogeneous nature of the palladium carbon catalyst allows for its complete removal via simple filtration at the end of the reaction, leaving the filtrate virtually free of heavy metal contaminants. Additionally, the controlled addition of ammonium formate in batches prevents localized overheating and excessive gas evolution, which minimizes the formation of side products such as azo-compounds or hydroxylamines. This precise control over reaction kinetics ensures that the final crystalline product achieves purity levels exceeding 98%, satisfying the rigorous quality requirements of global regulatory bodies.
How to Synthesize (S)-4-(4-aminobenzyl)-2-oxazolidinone Efficiently
Implementing this synthesis route requires careful attention to solvent selection and reagent stoichiometry to maximize yield and reproducibility. The patent specifies the use of polar aprotic solvents such as DMF or dioxane, which provide excellent solubility for the starting nitro compound while maintaining stability under reaction conditions. The process involves dissolving the substrate, adding the catalyst, and carefully managing the exothermic addition of the hydrogen donor to maintain the optimal temperature window. Detailed standard operating procedures regarding mixing speeds, addition rates, and crystallization temperatures are critical for transferring this laboratory success to pilot and production scales. The following guide outlines the fundamental steps derived from the patented examples to assist process engineers in establishing a robust manufacturing protocol.
- Dissolve (S)-4-(4-nitrobenzyl)-2-oxazolidinone in DMF or dioxane solvent system.
- Add 10% palladium carbon catalyst and heat the mixture to 50-60°C under stirring.
- Add ammonium formate in batches to maintain controlled hydrogen release and reaction temperature.
- Filter to recover catalyst, then add water to filtrate at 0-10°C to crystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this ammonium formate-based reduction pathway offers tangible economic and logistical benefits that extend beyond simple yield improvements. The elimination of hazardous hydrazine hydrate removes a significant bottleneck in raw material sourcing and storage, as hydrazine often requires special licensing and segregated warehousing due to its toxicity and instability. By switching to ammonium formate, a stable and widely available commodity chemical, facilities can reduce their inventory holding costs and simplify their safety compliance audits. Furthermore, the ability to recover and reuse the palladium catalyst multiple times without significant loss of activity translates directly into reduced consumption of precious metals, providing a substantial cost advantage over single-use catalytic systems or stoichiometric metal reductions.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the recyclability of the palladium carbon catalyst, which can be filtered, washed, and reintroduced into subsequent batches. This circular usage pattern significantly lowers the variable cost per kilogram of the intermediate compared to methods requiring fresh catalyst loads for every run. Additionally, the avoidance of iron sludge generation eliminates the costs associated with hazardous waste treatment and disposal, which can be a major expense in chemical manufacturing. The simplified workup procedure also reduces labor hours and utility consumption, as extensive washing and filtration steps required for iron removal are no longer necessary.
- Enhanced Supply Chain Reliability: Relying on stable, non-hazardous reagents like ammonium formate enhances the resilience of the supply chain against regulatory disruptions or transportation restrictions often placed on dangerous goods. The robustness of the reaction conditions, tolerating slight variations in temperature and addition rates, ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. This reliability allows for more accurate demand forecasting and inventory planning, ensuring that downstream API synthesis lines remain fully stocked without interruption. The high yield consistency reported in the patent data further guarantees that material throughput remains predictable and efficient.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method align perfectly with modern environmental, social, and governance (ESG) goals, making it easier to obtain permits for capacity expansion. The absence of heavy metal waste streams simplifies wastewater treatment requirements, allowing facilities to operate with a smaller environmental footprint. The process is inherently scalable because the heat management during ammonium formate decomposition is manageable through batched addition, preventing thermal runaway scenarios common in large-scale hydrazine reductions. This scalability ensures that the method remains viable whether producing hundreds of kilograms for clinical trials or tons for commercial market supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zolmitriptan intermediate synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity for potential partners. Understanding these nuances is crucial for evaluating the feasibility of adopting this technology within existing manufacturing frameworks. We encourage technical teams to review these points closely to appreciate the full scope of the process advantages.
Q: What are the advantages of using ammonium formate over hydrazine hydrate?
A: Ammonium formate eliminates the safety hazards associated with hydrazine hydrate, such as spontaneous combustion of spent carbon, and avoids the generation of difficult-to-remove iron sludge found in traditional ferric chloride reduction methods.
Q: Can the palladium catalyst be reused in this process?
A: Yes, the heterogeneous nature of the palladium carbon catalyst allows for simple filtration and recovery, enabling multiple reuse cycles without significant loss of catalytic activity, thereby reducing raw material costs.
Q: What purity levels can be achieved with this synthetic route?
A: The patented method consistently achieves product purity exceeding 98% and yields greater than 95%, meeting stringent specifications required for downstream pharmaceutical API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-(4-aminobenzyl)-2-oxazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving migraine medications like zolmitriptan. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of (S)-4-(4-aminobenzyl)-2-oxazolidinone meets the highest international standards. Our facility is equipped to handle the specific solvent systems and catalytic requirements of this patented process, delivering consistent quality that supports your regulatory filings and commercial launch timelines.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success. Let us be your partner in delivering reliable, cost-effective, and high-purity pharmaceutical intermediates.
