Advanced Chemical Resolution of Branched-Chain Amino Acids for Industrial Scale Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the production of high-purity chiral intermediates, particularly branched-chain amino acids which serve as critical building blocks for therapeutic agents and nutritional supplements. Patent CN101659623A introduces a significant advancement in this domain by detailing a novel chemical resolution method for DL-leucine and DL-isoleucine. This technology leverages the stereoselective properties of dibenzoyl-tartaric acid (DBTA) isomers to efficiently separate racemic mixtures into their respective D and L enantiomers. Unlike traditional biological fermentation which often suffers from long production cycles and complex downstream processing, this chemical approach offers a streamlined pathway that integrates high yield with exceptional optical purity. The strategic implementation of this resolution technique allows manufacturers to bypass the limitations of enzymatic specificity while maintaining rigorous quality standards required for active pharmaceutical ingredients (APIs) and high-value food additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of L-leucine and L-isoleucine has relied heavily on biological fermentation or protein hydrolysis, methods that present substantial operational bottlenecks for modern supply chains. Biological fermentation processes are notoriously time-consuming, often requiring extended incubation periods that delay time-to-market and tie up significant bioreactor capacity. Furthermore, the downstream purification of fermentation broths is chemically intensive, generating large volumes of waste liquid that pose environmental compliance challenges and increase disposal costs. Alternatively, protein hydrolysis methods utilizing organic sulfonic acids for precipitation introduce toxic reagents into the workflow, complicating safety protocols and necessitating expensive removal steps to meet residual solvent limits. Ion exchange techniques, while common, frequently struggle with low throughput and inconsistent product purity, leading to variable batch quality that is unacceptable for regulated pharmaceutical manufacturing. These legacy methods collectively contribute to higher production costs and reduced agility in responding to market demand fluctuations.
The Novel Approach
The methodology outlined in the patent data represents a paradigm shift by employing a direct chemical resolution strategy that circumvents the biological constraints of fermentation. By utilizing chiral resolving agents such as dibenzoyl-L-tartaric acid (L-DBTA) or dibenzoyl-D-tartaric acid (D-DBTA), the process creates diastereomeric salts with distinct solubility profiles, enabling precise physical separation through crystallization. This approach operates under relatively mild thermal conditions, typically between 60°C and 100°C, which reduces energy consumption compared to high-temperature hydrolysis. Crucially, the process design incorporates an inherent mechanism for the recovery and reuse of the expensive chiral resolving agent, addressing one of the primary cost drivers in classical resolution chemistry. The ability to simultaneously harvest both D and L enantiomers from a single racemic starting material effectively doubles the theoretical yield potential compared to asymmetric synthesis routes that discard the unwanted isomer, thereby maximizing raw material efficiency and economic viability.
Mechanistic Insights into DBTA-Mediated Chiral Resolution
The core of this technology lies in the formation of diastereomeric salts between the racemic amino acid and the chiral tartaric acid derivative. When DL-amino acid is dissolved in a dilute acid solvent and treated with a specific enantiomer of DBTA, the resulting interaction is stereospecific. For instance, reacting DL-leucine with D-DBTA preferentially forms a less soluble D-Leu·D-DBTA salt complex, which precipitates out of the solution upon cooling. This precipitation is driven by the lattice energy differences between the homochiral salt complex and the heterochiral counterpart remaining in the mother liquor. The reaction parameters, including the molar ratio of amino acid to resolving agent (optimized between 1:0.5 and 1:2.5) and the reaction temperature, are critical for maximizing the diastereomeric excess. Following filtration, the isolated salt is subjected to acidolysis using dilute hydrochloric acid, which protonates the amino acid and liberates the free DBTA resolving agent due to solubility differences in the acidic medium. This step is pivotal as it allows the DBTA to be filtered off and recycled, while the amino acid remains in the aqueous phase as a hydrochloride salt ready for final isolation.
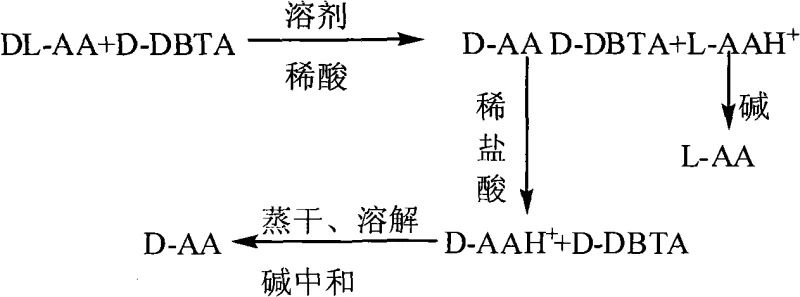
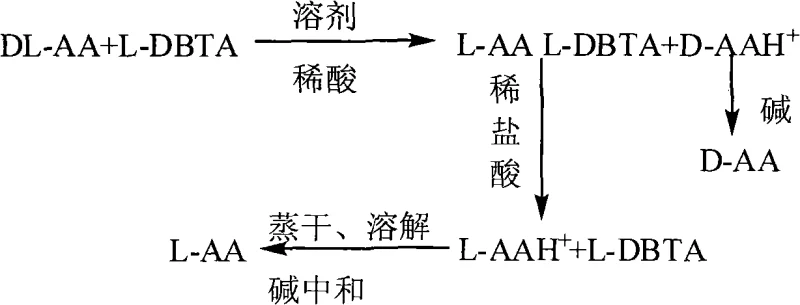
Impurity control is inherently managed through the crystallization dynamics of the diastereomeric salts. The patent specifies that cooling the reaction mixture to room temperature or lower promotes the selective crystallization of the target salt, leaving impurities and the undesired enantiomer in the supernatant. Subsequent washing of the filter cake with dilute acid further purifies the solid phase by removing adhering mother liquor containing the opposite enantiomer. The final neutralization step, adjusting the pH to a narrow range of 5.5 to 6.5 using bases such as ammonia or sodium hydroxide, ensures the precipitation of the zwitterionic amino acid form while keeping inorganic salts in solution. This precise pH control is essential for achieving the reported optical purity of greater than 98% and chemical content exceeding 98.5%. The robustness of this mechanism ensures that even with variations in raw material quality, the final product specifications remain consistent, a key requirement for reliable pharmaceutical intermediate supplier operations.
How to Synthesize High-Purity Leucine Efficiently
Implementing this resolution protocol requires careful attention to solvent selection and thermal management to ensure optimal crystal growth and agent recovery. The process begins with the dissolution of the racemic substrate in a dilute strong acid, followed by the controlled addition of the chiral selector. Maintaining the reaction temperature between 60°C and 100°C for a duration of 1 to 4 hours is essential to reach thermodynamic equilibrium before inducing crystallization through cooling. The subsequent acid treatment step not only isolates the product but also regenerates the valuable resolving agent, creating a semi-closed loop system that enhances sustainability. Detailed standard operating procedures regarding stirring rates, cooling gradients, and filtration techniques are critical for translating this laboratory-scale success to commercial production environments. The following guide outlines the standardized synthesis steps derived from the patent examples to facilitate technology transfer and process validation.
- Dissolve racemic amino acid in dilute acid solvent and add chiral resolving agent (D-DBTA or L-DBTA) at a molar ratio of 1: 0.5 to 1:2.5.
- React the mixture at 60-100°C for 1-4 hours, then cool to room temperature to precipitate the diastereomeric salt.
- Treat the filtered salt with dilute hydrochloric acid to recover the resolving agent, then neutralize the filtrate to isolate the pure amino acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chemical resolution technology offers tangible strategic benefits that extend beyond simple unit cost metrics. The primary advantage lies in the significant reduction of raw material waste through the efficient recovery of the chiral resolving agent. In traditional resolution processes, the resolving agent is often consumed or lost in mother liquors, representing a substantial portion of the cost of goods sold. By implementing the acidolysis recovery step described in the patent, manufacturers can retain a high percentage of the DBTA for reuse, drastically lowering the recurring cost of chiral auxiliaries. This efficiency translates directly into improved margin stability and protection against volatility in the pricing of specialty chemicals. Furthermore, the reliance on commodity reagents such as hydrochloric acid, ethanol, and ammonia simplifies the procurement landscape, reducing dependency on exotic or single-source suppliers that pose supply chain risks.
- Cost Reduction in Manufacturing: The elimination of expensive organic sulfonic acid precipitants and the avoidance of complex ion exchange resins result in a leaner cost structure. The ability to recycle the dibenzoyl-tartaric acid resolving agent means that the effective cost per kilogram of resolved amino acid decreases significantly over multiple batches. Additionally, the process avoids the high energy costs associated with long-duration fermentation cycles and the extensive wastewater treatment required for biological broths. By consolidating the production of both D and L isomers from a single racemic feedstock, the facility maximizes asset utilization and reduces the inventory burden of managing separate synthetic routes for each enantiomer.
- Enhanced Supply Chain Reliability: Sourcing racemic amino acids is generally more straightforward and cost-effective than sourcing pure enantiomers, as racemates are often available in bulk from diverse global suppliers. This flexibility allows procurement teams to negotiate better terms and switch vendors without compromising the final product quality, as the resolution step acts as a purification barrier. The use of standard industrial solvents and equipment (reactors capable of heating to 100°C and standard filtration units) ensures that production can be scaled or shifted between facilities with minimal requalification effort. This adaptability is crucial for maintaining continuity of supply in the face of geopolitical disruptions or logistical bottlenecks affecting specialized reagent shipments.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to protein hydrolysis methods that utilize toxic precipitants. The aqueous waste streams are primarily composed of salts and residual alcohols, which are easier to treat and dispose of in compliance with modern environmental regulations. The simplicity of the unit operations—dissolution, crystallization, filtration, and neutralization—makes the process highly scalable from pilot plant to multi-ton commercial production without the nonlinear scale-up issues often seen in bioprocessing. This scalability ensures that the supply chain can rapidly respond to surges in demand for high-purity amino acid intermediates used in parenteral nutrition and peptide synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. They are derived from the specific operational parameters and beneficial effects documented in the patent literature to provide clarity for potential partners and technical evaluators. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing manufacturing portfolios.
Q: What is the optical purity achievable with this resolution method?
A: According to patent CN101659623A, this method consistently achieves an optical purity greater than 98% for both D and L enantiomers of leucine and isoleucine.
Q: Can the chiral resolving agent be recycled in this process?
A: Yes, the process includes a specific step using dilute hydrochloric acid to treat the amino acid-resolving agent salt, allowing for the pre-recovery of DBTA with a recovery rate of 75-90%.
Q: Is this method suitable for large-scale industrial production?
A: The method utilizes common solvents like dilute hydrochloric acid and ethanol, operates at moderate temperatures (60-100°C), and avoids expensive precipitation agents, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Leucine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chiral intermediate manufacturing, leveraging advanced resolution technologies like the one described in CN101659623A to deliver superior value to our global clientele. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral HPLC and polarimetry instruments to guarantee that every batch of Leucine or Isoleucine meets the exacting standards of the pharmaceutical industry. Our commitment to process optimization allows us to offer high-purity chiral amino acids that are essential for the synthesis of next-generation therapeutics and high-performance nutritional products.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized resolution processes can enhance your supply chain resilience. Whether you require D-isoleucine for antimicrobial peptide synthesis or L-leucine for nutritional formulations, our dedicated experts are ready to collaborate on developing a supply strategy that balances cost, quality, and delivery performance effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
