Advanced Synthesis of Optical Ornidazole Enantiomers: A Scalable Solution for Global API Manufacturing
Advanced Synthesis of Optical Ornidazole Enantiomers: A Scalable Solution for Global API Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective methodologies for the production of chiral active pharmaceutical ingredients (APIs) and their intermediates. Patent CN100338039C represents a significant technological breakthrough in the synthesis of Ornidazole optical antimers, specifically S-(-)-Ornidazole and R-(+)-Ornidazole. This intellectual property outlines a sophisticated chemical pathway that bypasses the traditional and economically burdensome enzymatic resolution of racemic mixtures. By leveraging a direct asymmetric alkylation strategy using optically active epichlorohydrin, this method delivers high-purity intermediates essential for the formulation of third-generation nitroimidazole derivatives. For R&D directors and procurement specialists, understanding the nuances of this patent is critical, as it offers a viable route to reduce manufacturing complexity while ensuring the stringent stereochemical purity required for modern anti-anaerobic and antiprotozoal therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer Ornidazole has relied heavily on the resolution of racemic Ornidazole, often utilizing enzymatic methods as referenced in prior art such as CN 1400312A. While enzymatic resolution can theoretically separate enantiomers, it introduces substantial operational inefficiencies and economic bottlenecks for large-scale manufacturing. The reliance on biological catalysts often necessitates strict control over pH, temperature, and substrate concentration, leading to prolonged reaction times and inconsistent batch-to-batch reproducibility. Furthermore, the theoretical maximum yield of such resolution processes is inherently capped at 50% unless dynamic kinetic resolution is employed, which adds further layers of chemical complexity and cost. The disposal of the unwanted enantiomer and the recovery of enzymes contribute to a higher environmental footprint and increased waste treatment costs, making this conventional approach less attractive for cost-sensitive generic drug manufacturing and bulk API production.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN100338039C employs a direct condensation reaction between 2-methyl-5-nitroimidazole and optically active epichlorohydrin, either S-(+)-epichlorohydrin or R-(-)-epichlorohydrin. This strategic shift from resolving a racemate to synthesizing the chiral center directly from a chiral starting material fundamentally alters the economic equation of production. The process utilizes a Lewis acid catalyst, preferably Aluminum Chloride (AlCl3), to facilitate the nucleophilic attack of the imidazole nitrogen on the epoxide ring. This approach not only circumvents the 50% yield ceiling associated with resolution but also streamlines the downstream processing by eliminating the need for complex enzymatic separation columns or extensive chromatographic purification. The result is a streamlined workflow that transitions smoothly from condensation to hydrolysis and final crystallization, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
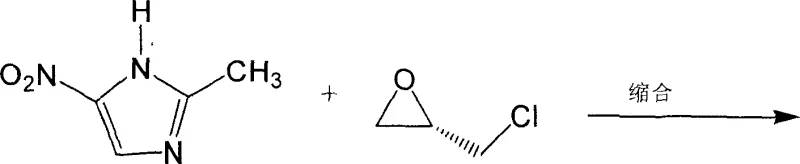
Mechanistic Insights into Lewis Acid-Catalyzed Chiral Alkylation
The core of this innovative synthesis lies in the precise activation of the epoxide ring by the Lewis acid catalyst. When Aluminum Chloride is introduced into the reaction system containing ethyl acetate and 2-methyl-5-nitroimidazole, it forms a coordinate complex that significantly enhances the electrophilicity of the epichlorohydrin. The reaction is meticulously controlled at low temperatures, typically between -10°C and 20°C, to prevent non-selective ring opening or polymerization of the epoxide. The nitrogen atom at the 1-position of the imidazole ring acts as a nucleophile, attacking the less hindered carbon of the activated epoxide. This regioselective ring opening is crucial for establishing the correct connectivity of the 3-chloro-2-hydroxypropyl side chain. The stereochemical information inherent in the starting S-(+)- or R-(-)-epichlorohydrin is effectively transferred to the final product, ensuring that the resulting Ornidazole possesses the desired optical rotation without the need for subsequent chiral separation steps.
Following the condensation phase, the reaction mixture undergoes a critical hydrolysis step which serves a dual purpose: it quenches the reactive Lewis acid species and facilitates the release of the organic product from the aluminum complex. The addition of water or ice water at controlled temperatures (0-40°C) breaks down the intermediate aluminum alkoxide complexes, liberating the free hydroxyl group on the propyl side chain. Subsequent acidification and neutralization steps are employed to partition the product into the organic phase while leaving inorganic salts and residual catalyst in the aqueous layer. This workup strategy is particularly effective for impurity control, as it ensures that metal residues are reduced to trace levels, thereby simplifying the final purification. The crude product obtained after solvent removal is then subjected to recrystallization, typically using toluene or ethanol, which further elevates the chemical purity to exceed 99.5%, effectively removing any minor regioisomers or unreacted starting materials.
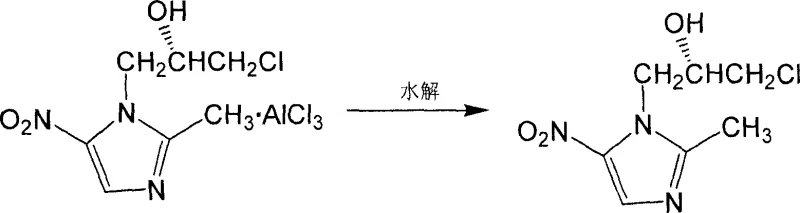
How to Synthesize S-(-)-Ornidazole Efficiently
The synthesis of high-purity S-(-)-Ornidazole requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure optimal yield and stereochemical integrity. The process begins with the preparation of a slurry of 2-methyl-5-nitroimidazole in ethyl acetate, followed by the careful addition of the Lewis acid catalyst under anhydrous conditions to prevent premature deactivation. The dropwise addition of S-(+)-epichlorohydrin must be managed to maintain the exotherm within safe limits, preserving the selectivity of the alkylation. Once the condensation is complete, the hydrolysis and extraction sequence must be executed rapidly to minimize product degradation. For a comprehensive understanding of the specific operational parameters, including exact molar ratios, stirring speeds, and crystallization cooling rates, please refer to the standardized protocol below.
- Condensation Reaction: Dissolve 2-methyl-5-nitroimidazole in ethyl acetate, cool to 0-10°C, add Aluminum Chloride (AlCl3), and dropwise add S-(+)-epichlorohydrin while maintaining temperature below 10°C.
- Hydrolysis and Workup: Add ice water to the reaction mixture to hydrolyze the aluminum complex, separate the organic phase, acidify the aqueous layer, and then neutralize with weak base to isolate the crude product.
- Purification: Recrystallize the crude S-(-)-Ornidazole from toluene or ethanol to achieve purity levels exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN100338039C offers transformative advantages regarding cost structure and supply reliability. The elimination of enzymatic resolution steps removes a significant variable cost component associated with biocatalysts and specialized filtration equipment. Furthermore, the starting materials, specifically 2-methyl-5-nitroimidazole and optically active epichlorohydrin, are commodity chemicals available from multiple global suppliers, which mitigates the risk of raw material shortages. The robustness of the chemical synthesis allows for flexible production scheduling and easier scale-up from pilot plants to multi-ton commercial reactors without the need for bespoke biological containment facilities. This flexibility translates directly into enhanced supply chain resilience and the ability to respond quickly to market demand fluctuations for Ornidazole APIs.
- Cost Reduction in Manufacturing: The direct chiral synthesis route fundamentally lowers the cost of goods sold (COGS) by maximizing atomic economy and yield efficiency. Unlike resolution methods that discard half of the material, this process utilizes the chirality of the starting epichlorohydrin to drive the formation of the desired product, effectively doubling the theoretical yield per unit of imidazole starting material. Additionally, the use of inexpensive Lewis acids like Aluminum Chloride instead of precious metal catalysts or expensive enzymes drastically reduces catalyst costs. The simplified workup procedure involving standard acid-base extraction reduces solvent consumption and energy usage during distillation, contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Dependence on single-source enzymatic providers can create bottlenecks in the supply chain; however, this chemical synthesis route relies on widely available petrochemical derivatives. The process conditions are compatible with standard glass-lined or enamel reactors found in most fine chemical manufacturing facilities, meaning that production can be easily outsourced or duplicated across different geographic regions without technology transfer barriers. This decentralization potential ensures continuous supply continuity even in the face of regional disruptions. Moreover, the stability of the intermediates allows for safer storage and transportation, further securing the logistics network for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvents like ethyl acetate and toluene which have well-established recovery and recycling protocols. The absence of biological waste streams simplifies wastewater treatment requirements, aligning with increasingly stringent environmental regulations. The high purity of the crude product prior to recrystallization minimizes the volume of mother liquor generated, reducing the load on solvent recovery units. This efficient resource utilization supports sustainable manufacturing practices and facilitates the commercial scale-up of complex pharmaceutical intermediates while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ornidazole synthesis technology. These insights are derived directly from the experimental data and claims within patent CN100338039C, providing clarity on purity specifications, process safety, and regulatory compliance. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of this synthesis method over enzymatic resolution?
A: Unlike enzymatic resolution of racemates which is costly and complex, this direct chiral alkylation method uses optically active epichlorohydrin to directly synthesize the target enantiomer, significantly reducing production costs and simplifying the process for industrial scale-up.
Q: What level of optical purity can be achieved with this process?
A: The patent data indicates that through optimized recrystallization steps, the final S-(-)-Ornidazole or R-(+)-Ornidazole products can achieve a chemical purity of over 99.5% with high optical rotation values, meeting stringent pharmaceutical standards.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents like ethyl acetate and toluene, and standard Lewis acid catalysts like Aluminum Chloride. The reaction conditions (0-20°C) are easily manageable in standard enamel reactors, making it highly suitable for metric-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ornidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation antimicrobial therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering Ornidazole enantiomers that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced chiral HPLC and polarimetry capabilities. Our facility is optimized for the handling of Lewis acid catalyzed reactions, ensuring safety and consistency in every batch produced for our global partners.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations for S-(-)-Ornidazole and R-(+)-Ornidazole. Let us help you optimize your API production costs while securing a reliable supply of critical nitroimidazole intermediates.
