Revolutionizing 5-Nitroimidazole Production: A Green Organocatalytic Approach for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for the production of critical anti-infective agents, particularly within the 5-nitroimidazole class which includes vital drugs like Ornidazole and Secnidazole. Patent CN111471017A introduces a transformative process that utilizes organic micromolecule catalysis to synthesize these essential medications, marking a significant departure from traditional, harsh synthetic methods. This innovation addresses long-standing challenges in the field, such as the reliance on corrosive Lewis acids and the generation of substantial hazardous waste, by employing a bifunctional thiourea catalyst that operates under remarkably mild conditions. For R&D directors and process chemists, this patent represents a pivotal shift towards greener chemistry, offering a route that not only enhances reaction selectivity but also simplifies the purification process, thereby ensuring a higher quality final product with reduced impurity profiles. The ability to achieve high yields at near-ambient temperatures suggests a robust methodology that is highly attractive for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-nitroimidazole derivatives has been plagued by inefficient and environmentally taxing processes that pose significant risks to both equipment integrity and operational safety. Traditional routes often involve the use of bis(3-chloro-hydroxypropyl) sulfate, which requires dangerous sulfuryl chloride and results in unstable intermediates, or rely heavily on formic acid solutions that necessitate excessive amounts of corrosive reagents and prolonged reaction times of up to 72 hours with yields as low as 42%. Furthermore, the widespread use of Lewis acids like anhydrous aluminum trichloride creates a massive burden on waste management systems, generating tons of aluminum-containing sludge that is difficult and costly to treat, while also causing severe corrosion to reactor vessels. These legacy methods not only drive up the cost of goods sold due to low atom economy and high energy consumption but also introduce complex supply chain vulnerabilities related to the handling and disposal of hazardous chemicals.
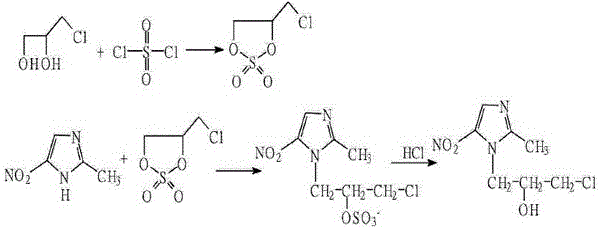
The Novel Approach
In stark contrast to these archaic methodologies, the novel organocatalytic process described in the patent utilizes a sophisticated bifunctional thiourea catalyst to facilitate the ring-opening reaction of epoxides with 2-methyl-5-nitroimidazole. This approach allows the reaction to proceed efficiently in solvents like ethyl acetate at temperatures ranging from 20°C to 35°C, drastically reducing energy requirements and eliminating the need for aggressive acidic or basic conditions. The new synthetic pathway achieves impressive yields of 80% to 85% with exceptional regioselectivity, ensuring that the desired therapeutic isomer is produced predominantly while minimizing the formation of difficult-to-remove byproducts. By replacing stoichiometric amounts of corrosive catalysts with a catalytic amount of a recoverable organic molecule, this method fundamentally alters the economic and environmental landscape of 5-nitroimidazole manufacturing.
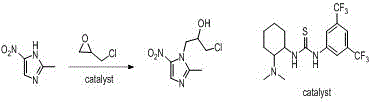
Mechanistic Insights into Bifunctional Thiourea Catalysis
The core of this technological advancement lies in the unique dual-activation mechanism provided by the catalyst, 1-(3,5-bis(trifluoromethyl)phenyl)-3-(2-dimethylamino-cyclohexyl)-thiourea, which acts as a molecular machine to orchestrate the reaction with high precision. The tertiary amine moiety within the catalyst structure serves to activate the 2-methyl-5-nitroimidazole substrate by interacting with the active hydrogen on the nitrogen atom, thereby enhancing its nucleophilicity and readiness to attack the electrophile. Simultaneously, the thiourea unit forms a double-hydrogen bond six-membered ring transition state with the oxygen atom of the epichlorohydrin or propylene oxide, effectively polarizing the epoxide ring and making it more susceptible to nucleophilic attack. This synergistic activation lowers the activation energy of the reaction significantly, allowing it to proceed rapidly under mild thermal conditions without the need for external heating or harsh promoters.
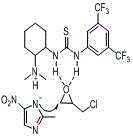
Beyond merely accelerating the reaction rate, this specific catalytic architecture plays a critical role in controlling the regioselectivity of the ring-opening process, which is paramount for ensuring the biological efficacy of the final drug product. The steric hindrance provided by the bulky 3,5-bis(trifluoromethyl)phenyl group on the catalyst directs the nucleophilic attack preferentially to the less hindered carbon of the epoxide ring, effectively suppressing the formation of unwanted regioisomers. This high degree of selectivity, reported to be up to 98%, translates directly into a cleaner crude reaction mixture, which simplifies downstream processing and reduces the load on purification units such as chromatography columns or crystallization tanks. For quality control teams, this means a more consistent impurity profile and a higher probability of meeting stringent pharmacopeial standards for residual solvents and related substances.
How to Synthesize Ornidazole Efficiently
The practical implementation of this synthesis involves a straightforward procedure where 2-methyl-5-nitroimidazole is first mixed with a solvent such as ethyl acetate and the specific thiourea catalyst under controlled stirring conditions. Once the mixture is homogenized, the epoxide reagent, either epichlorohydrin for Ornidazole or propylene oxide for Secnidazole, is added to the system, and the reaction is allowed to proceed at ambient temperatures for a duration of 4 to 10 hours. Following the completion of the reaction, a simple aqueous workup involving pH adjustment allows for the separation of the organic product phase from the aqueous catalyst phase, facilitating the recovery and reuse of the valuable catalyst while the product is isolated through standard crystallization techniques. Detailed standardized synthesis steps are provided in the guide below.
- Mix 2-methyl-5-nitroimidazole with a solvent (ethyl acetate) and the bifunctional thiourea catalyst.
- Add epichlorohydrin or propylene oxide to the mixture and react at 20-35°C for 4-10 hours.
- Perform aqueous workup to separate the organic phase, recover the catalyst from the water phase, and recrystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this organocatalytic technology offers profound strategic benefits that extend far beyond simple chemical efficiency, impacting the total cost of ownership and operational resilience of the manufacturing site. The elimination of corrosive reagents like concentrated hydrochloric acid and aluminum trichloride significantly extends the lifespan of reactor vessels and piping infrastructure, reducing capital expenditure on maintenance and replacement of corroded equipment. Furthermore, the ability to recover and recycle the organic catalyst multiple times without significant loss of activity creates a circular material flow that decouples production costs from the volatility of raw material markets, providing a stable and predictable cost structure for long-term supply agreements.
- Cost Reduction in Manufacturing: The shift from stoichiometric inorganic catalysts to a recyclable organic system drastically reduces the consumption of raw materials and the associated costs of waste disposal, leading to substantial overall savings in the cost of goods sold. By avoiding the generation of heavy metal sludge and acidic wastewater, the facility saves significantly on environmental compliance fees and the energy-intensive processes required for effluent treatment, directly improving the bottom line. Additionally, the milder reaction conditions reduce utility consumption for heating and cooling, further contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents ensures a robust supply chain that is less susceptible to disruptions compared to processes relying on hazardous or tightly regulated chemicals. The simplified workflow and reduced reaction times allow for faster batch turnover, enabling the manufacturer to respond more agilely to fluctuations in market demand and shorten lead times for customers. This reliability is crucial for maintaining continuous supply of critical antibiotics, ensuring that downstream pharmaceutical partners can meet their own production schedules without interruption.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against potential regulatory crackdowns on hazardous waste. The absence of toxic heavy metals and the minimization of solvent usage make the scale-up from pilot plant to commercial tonnage much smoother, with fewer engineering hurdles related to heat dissipation and safety containment. This environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the supplier as a responsible partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic process, derived directly from the experimental data and beneficial effects outlined in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement specialists assessing the quality implications of the new method. The answers provided reflect the specific advantages of the bifunctional catalyst system in terms of yield, purity, and operational safety.
Q: What are the advantages of the organocatalytic route over traditional Lewis acid methods?
A: The organocatalytic route eliminates the use of corrosive Lewis acids like AlCl3, significantly reducing hazardous waste generation and equipment corrosion while improving regioselectivity and yield up to 85%.
Q: Is the bifunctional thiourea catalyst reusable?
A: Yes, the catalyst can be recovered from the aqueous phase by pH adjustment, filtered, dried, and reused directly in subsequent batches, lowering overall production costs.
Q: What represents the key technical breakthrough in Patent CN111471017A?
A: The breakthrough lies in using a dual-activation mechanism where the catalyst simultaneously activates the nucleophile (imidazole) and the electrophile (epoxide) via hydrogen bonding, enabling mild reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ornidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the organocatalytic synthesis route described in Patent CN111471017A and are fully equipped to leverage this technology for the benefit of our global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle sensitive organocatalytic reactions with precision, supported by rigorous QC labs that guarantee stringent purity specifications for every batch of 5-nitroimidazole intermediates we produce.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their supply chains and reduce their environmental footprint through the adoption of this advanced manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments to validate the commercial viability of this green synthesis method. Let us help you secure a sustainable and cost-effective supply of high-quality Ornidazole and Secnidazole intermediates for your critical drug formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
