Advanced Direct Resolution of D-Serine: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry's demand for high-purity chiral intermediates continues to escalate, particularly for neurological therapeutics targeting N-methyl-D-aspartate (NMDA) receptors. Patent CN103755582A introduces a transformative methodology for the preparation of D-serine, a critical active pharmaceutical ingredient (API) intermediate used in the treatment of schizophrenia and other cognitive disorders. This technical disclosure outlines a novel direct resolution strategy that bypasses the cumbersome esterification steps traditionally required in chemical separation processes. By leveraging specific L-cyclic phosphoric acid derivatives as resolving agents, the inventors have achieved a breakthrough in both operational simplicity and economic efficiency. The process utilizes a half-equivalent stoichiometry of the chiral auxiliary, which fundamentally alters the cost structure of production while maintaining exceptional optical purity levels exceeding 99% o.p. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this technology represents a significant leap forward in process chemistry, offering a robust pathway from laboratory discovery to commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of D-serine has been plagued by significant technical and economic hurdles associated with established synthetic routes. Asymmetric synthesis methods, such as those employing fluorine-containing chiral ligands with nickel complexes, often result in moderate yields around 65% and require expensive, specialized reagents that are difficult to source at scale. Biological fermentation and enzymatic catalysis routes present their own set of challenges, including extremely low substrate concentrations that necessitate large reactor volumes and generate substantial volumes of wastewater, creating immense pressure on environmental compliance and waste treatment infrastructure. Furthermore, traditional chemical resolution techniques typically mandate a preliminary esterification of DL-serine to facilitate separation, adding multiple unit operations such as protection, resolution, hydrolysis, and purification. These additional steps not only extend the production lead time but also accumulate yield losses at each stage, rendering the final product cost-prohibitive for large-scale applications. The reliance on 1:1 molar ratios of resolving agents in classical resolution further exacerbates raw material costs, as half of the expensive chiral auxiliary remains tied up in the unwanted enantiomer salt unless complex recycling schemes are implemented.
The Novel Approach
In stark contrast to these legacy methodologies, the patented process introduces a streamlined direct resolution technique that eliminates the need for esterification entirely. The core innovation lies in the utilization of specific L-cyclic phosphoric acid derivatives, namely (-)-4-phenyl, (-)-4-(2-chlorophenyl), and (-)-4-(2-methoxyphenyl) variants, which exhibit superior selectivity for D-serine in an acidic alcoholic medium. 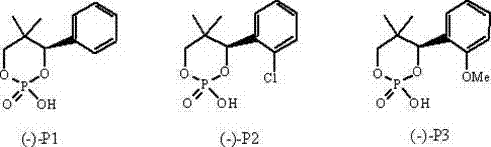 . By operating with a half-equivalent (0.5:1) molar ratio of the resolving agent to the racemic substrate, the process maximizes the effective utilization rate of the chiral auxiliary, effectively doubling the theoretical throughput per unit of resolving agent compared to standard protocols. The reaction conditions are remarkably mild, involving simple reflux in an aqueous alcohol solution followed by cooling crystallization, which facilitates the selective precipitation of the D-serine resolving agent salt. This approach not only simplifies the equipment requirements but also significantly reduces the energy consumption and solvent usage associated with multi-step synthesis, positioning it as a highly attractive option for cost reduction in API manufacturing.
. By operating with a half-equivalent (0.5:1) molar ratio of the resolving agent to the racemic substrate, the process maximizes the effective utilization rate of the chiral auxiliary, effectively doubling the theoretical throughput per unit of resolving agent compared to standard protocols. The reaction conditions are remarkably mild, involving simple reflux in an aqueous alcohol solution followed by cooling crystallization, which facilitates the selective precipitation of the D-serine resolving agent salt. This approach not only simplifies the equipment requirements but also significantly reduces the energy consumption and solvent usage associated with multi-step synthesis, positioning it as a highly attractive option for cost reduction in API manufacturing.
Mechanistic Insights into Chiral Resolution via Cyclic Phosphates
The success of this resolution strategy is rooted in the precise control of diastereomeric salt solubility within a tailored solvent environment. When racemic DL-serine interacts with the chiral L-cyclic phosphoric acid in the presence of an inorganic protonic acid, four potential diastereomeric salt isomers can theoretically form. However, the patent elucidates that by carefully modulating the solvent composition—specifically the ratio of alcohol to water—and the acidity of the medium, the solubility of the target D-serine·L-cyclic phosphate salt is drastically reduced relative to its counterparts. This differential solubility drives the selective crystallization of the desired diastereomer from the solution, leaving the unwanted L-serine species predominantly in the mother liquor. The use of inorganic acids such as hydrochloric, sulfuric, or phosphoric acid plays a critical role in protonating the amino group of serine, thereby enhancing the ionic interaction with the phosphate anion and stabilizing the crystal lattice. The mechanism ensures that the crystallization process is thermodynamically driven towards the formation of the less soluble D-isomer salt, achieving high optical purity without the need for repetitive recrystallization steps that typically erode overall yield.
Furthermore, the dissociation mechanism employed to liberate the free D-serine from the diastereomeric salt is engineered for maximum recovery and minimal impurity carryover. The process involves dissolving the isolated salt and adjusting the pH to a specific range of 5.0 to 6.0 using an alkali solution, which triggers the precipitation of free D-serine while keeping the resolving agent in solution or allowing for its separate recovery under strong acidic conditions. This pH-swing technique exploits the isoelectric point of serine to ensure high recovery rates, consistently demonstrating yields between 93% and 94% across various solvent systems including methanol, ethanol, and isopropanol. The robustness of this mechanistic approach allows for tight control over the impurity profile, ensuring that the final product meets stringent pharmaceutical specifications. Additionally, the ability to recover the resolving agent with efficiencies reaching 98% means that the chiral pool is effectively closed-loop, minimizing waste generation and aligning with green chemistry principles that are increasingly mandated by global regulatory bodies.
How to Synthesize D-Serine Efficiently
The synthesis of D-serine via this direct resolution method offers a practical and scalable route for manufacturing teams aiming to optimize their production workflows. The protocol is designed to be adaptable to various solvent systems, providing flexibility in sourcing raw materials and managing solvent recovery streams. Detailed below is the standardized operational framework derived from the patent examples, which guides the operator through the critical stages of salt formation, isolation, and dissociation. Adhering to these parameters ensures consistent optical purity and yield, mitigating the risks associated with batch-to-batch variability. For a comprehensive understanding of the specific temperature profiles and stirring rates, please refer to the detailed standard operating procedures provided in the technical documentation below.
- Combine racemic DL-serine with a half-equivalent of L-cyclic phosphoric acid resolving agent in an alcoholic aqueous solution containing an inorganic protonic acid.
- Heat the mixture to reflux until clear, then cool slowly to induce selective crystallization of the D-serine resolving agent salt.
- Separate the precipitate and dissociate the salt by pH adjustment to isolate high-purity D-serine, while recovering the resolving agent for recycling.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this technology addresses several critical pain points associated with the supply of high-value chiral intermediates. The elimination of the esterification step removes an entire category of reagents and processing time from the value chain, resulting in a drastically simplified manufacturing workflow. This simplification translates directly into reduced capital expenditure on equipment and lower operational overheads, as fewer reactors and separation units are required to achieve the same output volume. Moreover, the high recovery rate of the resolving agent creates a sustainable economic model where the cost of the chiral auxiliary is amortized over multiple cycles, leading to substantial cost savings in the long term. For supply chain heads, the robustness of the crystallization process ensures predictable batch cycles and reliable delivery schedules, reducing the risk of stockouts that can disrupt downstream drug formulation activities.
- Cost Reduction in Manufacturing: The implementation of a half-equivalent resolving agent strategy fundamentally shifts the cost dynamics of chiral separation. By requiring only 0.5 moles of the expensive chiral phosphate per mole of racemic substrate, the raw material intensity is significantly lowered compared to conventional 1:1 resolution methods. Additionally, the high recovery efficiency of the resolving agent allows for its repeated reuse, effectively decoupling production volume from the continuous purchase of fresh chiral reagents. This circular usage model, combined with the removal of esterification and hydrolysis steps, results in a leaner process with fewer unit operations, thereby reducing utility consumption, labor costs, and waste disposal fees associated with complex synthetic routes.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as racemic DL-serine and common inorganic acids ensures a stable and resilient supply chain不受制于 scarce specialty reagents. Unlike enzymatic processes that may depend on specific biocatalysts with limited shelf-life or availability, this chemical resolution utilizes stable small molecules that can be sourced from multiple global suppliers. The simplicity of the process also reduces the likelihood of technical failures or batch rejections due to complex biological variables, ensuring a consistent flow of material to meet production targets. This reliability is crucial for maintaining continuity in the manufacture of downstream neurological medications where D-serine is a key component.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations like reflux, filtration, and crystallization that are well-understood in chemical engineering. The avoidance of heavy metal catalysts found in asymmetric synthesis and the reduction of wastewater volume compared to fermentation methods significantly ease the burden on environmental treatment facilities. The ability to recycle the L-serine byproduct via thermal racemization further enhances the atom economy of the process, minimizing the generation of organic waste. These factors collectively facilitate easier regulatory approval for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to expand capacity from pilot scale to multi-ton production with confidence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity on process capabilities and limitations. Understanding these aspects is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs. The answers reflect the proven performance of the system under the described conditions.
Q: What is the primary advantage of using half-equivalent resolving agents in this D-serine process?
A: Using a 0.5:1 molar ratio of the chiral resolving agent to the racemic substrate significantly reduces raw material costs compared to traditional 1:1 resolution methods. Furthermore, the patent demonstrates that the resolving agent can be recovered with high efficiency (up to 98%) and recycled, drastically lowering the overall production cost per kilogram.
Q: How does this method compare to enzymatic or asymmetric synthesis routes for D-serine?
A: Unlike enzymatic methods which often suffer from low substrate concentrations and significant wastewater generation, this chemical resolution operates at higher concentrations with simpler workup procedures. Compared to asymmetric synthesis which requires expensive fluorinated ligands and yields around 65%, this resolution method achieves yields of 93-94% with optical purity exceeding 99% o.p., making it far more suitable for industrial scale-up.
Q: Can the L-serine byproduct be utilized in this process?
A: Yes, the process is designed for maximum atom economy. The L-serine remaining in the mother liquor after the initial crystallization can be isolated, subjected to thermal racemization, and recycled back into the process as DL-serine feedstock, effectively minimizing waste and maximizing overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Serine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of D-serine as a high-value pharmaceutical intermediate in the development of next-generation neurological therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify optical purity and impurity profiles. Our expertise in chiral resolution technologies allows us to offer customized solutions that align with your specific cost and quality targets, making us a trusted partner for global pharmaceutical companies.
We invite you to engage with our technical procurement team to discuss how this advanced resolution process can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this direct resolution method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your development timelines are met with the highest standards of quality and reliability.
