Advanced Dynamic Kinetic Resolution for High-Purity D-Serine Manufacturing
Introduction to Patent CN101735085A
The pharmaceutical industry constantly seeks more efficient pathways for producing chiral amino acids, which serve as critical building blocks for various therapeutic agents and biochemical reagents. Patent CN101735085A introduces a groundbreaking method for preparing D-serine through a sophisticated dynamic kinetic resolution process. Unlike traditional methods that are inherently limited by stoichiometry, this innovation leverages a specific racemization catalyst to continuously convert the undesired enantiomer back into the reactive pool. This technical breakthrough addresses the long-standing challenge of low yields in chiral resolution, offering a robust solution for manufacturers aiming to optimize their production lines. By integrating solid acid catalysis for esterification followed by a dynamic resolution step, the process achieves theoretical conversion rates nearing 100%, marking a significant leap forward in amino acid synthesis technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure D-serine relied heavily on classical static resolution techniques using resolving agents like L-dibenzoyltartaric acid (L-DBTA). While effective in separating enantiomers, these traditional processes suffer from a fundamental thermodynamic limitation: they can theoretically isolate only half of the starting racemic material. In practice, as noted in prior art such as literature from Southeast University, the resolution yield often stagnates around 48.8%, meaning more than half of the valuable raw material is left behind as the unwanted L-isomer or requires complex recycling loops. This inefficiency not only inflates the cost of goods sold due to wasted feedstock but also generates substantial chemical waste, creating environmental burdens and complicating downstream purification efforts for pharmaceutical grade intermediates.
The Novel Approach
The methodology disclosed in the patent fundamentally alters this paradigm by introducing a racemization catalyst into the resolution system. Instead of allowing the L-serine methyl ester to remain inert or be discarded, the added catalyst facilitates its rapid equilibration back into the DL-mixture under the reaction conditions. This dynamic process ensures that as the D-isomer is selectively crystallized as the L-DBTA salt, the L-isomer is continuously replenished and converted, driving the reaction towards completion. Consequently, the resolution yield is dramatically enhanced to a range of 78-88%, with optical purities consistently exceeding 99%. This approach transforms a wasteful separation process into a highly efficient conversion, stabilizing product quality and making the technology exceptionally suitable for large-scale industrial applications where material efficiency is paramount.
Mechanistic Insights into Racemization-Catalyzed Resolution
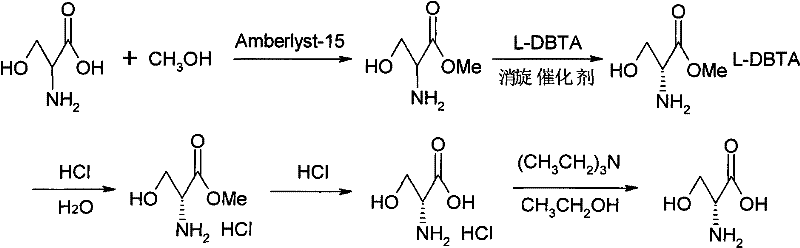
The core of this synthetic strategy lies in the synergistic interaction between the resolving agent and the racemization catalyst. The process begins with the esterification of DL-serine using methanol catalyzed by Amberlyst-15, a macroreticular strong acid ion exchange resin. This solid acid catalyst offers distinct advantages over homogeneous mineral acids, including ease of separation and reduced corrosion. Following esterification, the crude DL-serine methyl ester undergoes dynamic kinetic resolution. Here, the choice of racemization catalyst is critical; the patent highlights pyridine aldehydes, particularly 4-pyridinecarboxaldehyde, as highly effective agents. These catalysts likely operate through a Schiff base mechanism, temporarily binding to the amino group of the serine ester to facilitate proton abstraction and reprotonation at the chiral center, thereby scrambling the stereochemistry of the unreacted L-isomer.
This mechanistic feature ensures that the equilibrium constantly shifts to favor the formation of the less soluble diastereomeric salt, D-serine methyl ester·L-DBTA. Once this salt is isolated, it is subjected to acid dissociation using hydrochloric acid, which liberates the free amine and removes the resolving agent. The final step involves hydrolysis of the methyl ester under acidic conditions to yield the target D-serine. The rigorous control of reaction parameters, such as maintaining temperatures between 35-65°C during resolution and 60-100°C during hydrolysis, is essential to prevent epimerization of the product while ensuring complete conversion. The result is a high-purity amino acid with a melting point consistent with literature values (approx. 220°C) and specific rotation values confirming high enantiomeric excess.
How to Synthesize D-Serine Efficiently
The synthesis of D-serine via this patented route involves a streamlined three-step sequence that balances high yield with operational simplicity. The process initiates with the esterification of DL-serine in anhydrous methanol using Amberlyst-15 resin, followed by a dynamic kinetic resolution step where a pyridine-based racemization catalyst is employed alongside L-DBTA. The final stage involves acid hydrolysis to retrieve the free amino acid. This protocol is designed to maximize the utilization of the racemic starting material while minimizing the generation of hazardous waste. For detailed laboratory protocols and precise reaction conditions tailored to your specific scale, please refer to the standardized synthesis steps outlined below.
- Esterify DL-serine with methanol using Amberlyst-15 resin catalyst at 20-70°C to form DL-serine methyl ester.
- Perform dynamic kinetic resolution on the ester using L-DBTA and a pyridine-based racemization catalyst at 35-65°C.
- Dissociate the resulting salt with hydrochloric acid and hydrolyze at 60-100°C to obtain the final D-Serine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this dynamic kinetic resolution technology presents a compelling value proposition centered on cost efficiency and supply security. Traditional resolution methods inherently waste nearly half of the purchased raw material, creating a volatile cost structure dependent on the price of DL-serine. By contrast, this novel approach effectively utilizes the entire racemic input, drastically reducing the raw material cost per kilogram of finished D-serine. Furthermore, the use of heterogeneous catalysts like Amberlyst-15 simplifies the workup process, eliminating the need for extensive neutralization and salt disposal steps, which translates directly into lower utility and waste treatment costs for the manufacturing facility.
- Cost Reduction in Manufacturing: The implementation of a racemization catalyst fundamentally changes the economics of chiral amino acid production by breaking the 50% yield ceiling. In traditional static resolution, the theoretical maximum yield is capped, forcing manufacturers to purchase double the amount of starting material required for the final product mass. By enabling the continuous conversion of the unwanted isomer, this process significantly lowers the effective consumption of DL-serine. Additionally, the ability to recover and reuse the resolving agent L-DBTA and the solid acid catalyst further contributes to substantial operational expenditure savings, making the final product more price-competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain stability is often threatened by the inefficiencies of low-yield processes that require larger volumes of raw materials to meet production targets. This high-yield methodology mitigates such risks by maximizing output from existing feedstock inventories. The robustness of the reaction conditions, which tolerate standard industrial solvents like methanol and ethanol, ensures that the process is not overly sensitive to minor fluctuations in reagent quality. This reliability allows for more accurate forecasting and inventory management, ensuring that downstream customers in the pharmaceutical and biochemical sectors receive consistent deliveries of high-purity D-serine without unexpected interruptions caused by production bottlenecks.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden complexities, particularly regarding heat transfer and waste management. This patented route is inherently scalable due to the use of solid catalysts that can be easily filtered and regenerated, avoiding the handling of large volumes of corrosive liquid acids. The reduction in chemical waste, stemming from the higher atom economy of the dynamic resolution, aligns with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental footprint of the manufacturing site but also simplifies the regulatory approval process for new drug filings that require detailed documentation of impurity profiles and synthesis routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of D-serine using dynamic kinetic resolution. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on yield expectations, purity profiles, and process scalability. Understanding these details is crucial for R&D teams evaluating this technology for integration into their existing manufacturing portfolios.
Q: What is the primary advantage of using a racemization catalyst in D-Serine production?
A: The use of a racemization catalyst allows for the continuous conversion of the unwanted L-isomer back into the racemic mixture during resolution. This breaks the theoretical 50% yield barrier of traditional static resolution, enabling theoretical conversion rates approaching 100% and significantly improving overall process efficiency.
Q: How does this method improve the optical purity of the final D-Serine?
A: By employing dynamic kinetic resolution with specific resolving agents like L-DBTA and optimized racemization catalysts such as 4-pyridinecarboxaldehyde, the process ensures that the crystallized salt is highly enriched in the D-configuration. Subsequent hydrolysis yields D-Serine with optical purity exceeding 99%, meeting stringent pharmaceutical standards.
Q: Is the Amberlyst-15 catalyst reusable in this synthesis route?
A: Yes, Amberlyst-15 is a solid acid ion exchange resin that can be easily separated from the reaction mixture via filtration. This heterogeneity not only simplifies the workup procedure by avoiding neutralization steps associated with liquid acids but also allows for the potential recovery and reuse of the catalyst, contributing to lower operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Serine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering D-serine and other specialized amino acids with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify optical purity and chemical composition. Our infrastructure is designed to support the complex requirements of dynamic kinetic resolution, guaranteeing a stable supply of materials that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to this high-yield route can optimize your budget. Please contact us to request specific COA data for our current batches or to discuss route feasibility assessments for your upcoming projects. Let us be your partner in achieving both scientific excellence and commercial success.
