Optimizing Meropenem Production: A Novel Allyl-Protection Strategy for Commercial Scale-Up
Introduction to Advanced Carbapenem Synthesis
The landscape of beta-lactam antibiotic manufacturing is continuously evolving, driven by the urgent need for more efficient and cost-effective production methods for critical drugs like Meropenem. Patent CN101962383A introduces a significant technological breakthrough in the synthesis of this broad-spectrum carbapenem antibiotic, addressing long-standing challenges in yield optimization and environmental safety. By strategically replacing the traditional p-nitrobenzyl protecting group with an allyl moiety, this novel route streamlines the synthetic pathway, eliminating the need for hazardous hydrogen gas in certain embodiments and avoiding expensive transition metals. This innovation represents a pivotal shift towards greener chemistry in the pharmaceutical sector, offering a robust alternative for the large-scale production of high-purity active pharmaceutical ingredients.
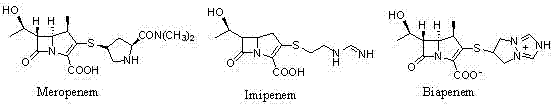
Understanding the structural nuances of carbapenems is essential for appreciating the complexity of their synthesis. As illustrated in the reference structures, Meropenem shares the core beta-lactam fused ring system with Imipenem and Biapenem, yet its specific side chain configuration demands precise stereochemical control. The patented method focuses on constructing this intricate architecture through a series of highly selective reactions, ensuring that the final product meets the rigorous purity standards required for treating severe infections. For global procurement teams, adopting such advanced methodologies translates directly into enhanced supply security and reduced dependency on legacy processes that are often plagued by low efficiency and high waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Meropenem has relied heavily on routes that involve the separate preparation of the side chain and the parent nucleus, typically designated as MAP, followed by a condensation step. As depicted in earlier prior art, these conventional pathways often necessitate the use of 4-acetoxyazetidinone (4AA) as a starting material, undergoing multiple transformation steps to reach the key intermediate. A critical bottleneck in these traditional methods is the reliance on precious metal catalysts, specifically Rhodium, during the formation of the bicyclic core. This dependency not only inflates the raw material costs significantly but also introduces complex purification challenges to remove trace metal residues, which is a major concern for regulatory compliance in API manufacturing.
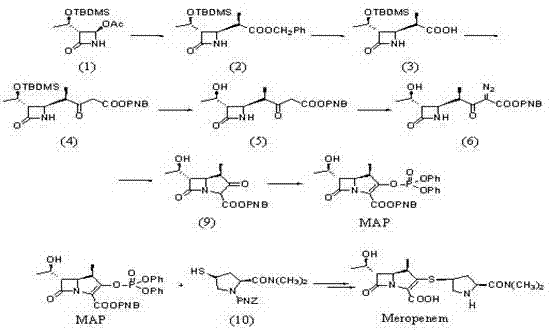
Furthermore, the use of p-nitrobenzyl as a protecting group in classical routes presents inherent steric and electronic drawbacks. The bulkiness of the nitrobenzyl group can hinder the efficiency of subsequent cyclization reactions, often resulting in lower overall yields and requiring harsher reaction conditions to drive the conversion to completion. Additionally, the removal of the nitrobenzyl group typically requires catalytic hydrogenation, which involves handling high-pressure hydrogen gas, posing significant safety risks and requiring specialized infrastructure. These cumulative inefficiencies create a fragile supply chain where minor deviations in reaction parameters can lead to substantial batch failures, impacting the reliability of supply for downstream drug manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the synthesis method disclosed in CN101962383A leverages an allyl-protection strategy that fundamentally alters the reaction dynamics. By utilizing allyl esters and carbamates instead of nitrobenzyl derivatives, the new route capitalizes on the smaller steric profile of the allyl group. This structural modification facilitates the crucial Wittig ring-closure reaction, allowing it to proceed with greater ease and higher selectivity. The process flow, as detailed in the patent, involves the condensation of specific azetidinone precursors followed by a streamlined cyclization and deprotection sequence that bypasses the need for Rhodium catalysts entirely.
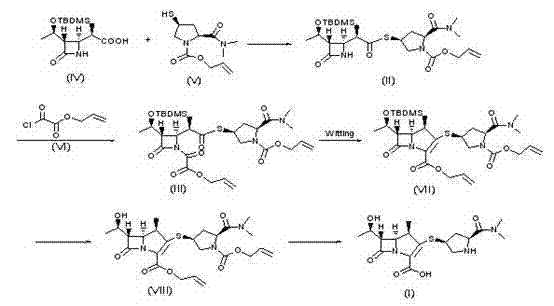
The operational simplicity of this novel approach is another defining feature. The reaction conditions are notably milder, often proceeding at or near room temperature, which reduces energy consumption and minimizes the formation of thermal degradation byproducts. Moreover, the final deprotection step can be achieved using palladium catalysts in conjunction with chemical scavengers like dimedone or tributyltin hydride, effectively removing the allyl groups without the need for gaseous hydrogen. This capability not only enhances plant safety but also simplifies the reactor setup, making the process highly adaptable for commercial scale-up in diverse manufacturing facilities. For supply chain managers, this translates to a more resilient production model with fewer technical barriers to entry.
Mechanistic Insights into Wittig Cyclization and Deprotection
The core of this synthetic innovation lies in the mechanistic advantages provided by the allyl protecting group during the formation of the carbapenem skeleton. In the traditional nitrobenzyl route, the steric bulk around the reaction center can impede the approach of the phosphorus ylide or phosphite reagent required for ring closure. By substituting this with an allyl group, the spatial congestion around the azetidinone nitrogen is significantly reduced. This reduction in steric hindrance allows the organophosphorus reagent, such as triphenyl phosphite or trimethyl phosphite, to interact more effectively with the carbonyl functionality, promoting a smoother intramolecular cyclization to form the bicyclic [3.2.0] heptene system. The result is a cleaner reaction profile with fewer side products, which simplifies downstream purification.
Equally critical is the mechanism of the final deprotection step, which utilizes a palladium-catalyzed allyl transfer reaction. In this process, the palladium catalyst coordinates with the allyl double bond, forming a pi-allyl palladium complex that is susceptible to nucleophilic attack. Unlike hydrogenolysis which requires H2 gas, this method employs a chemical scavenger to trap the liberated allyl group, driving the equilibrium towards the free amine and carboxylic acid forms of Meropenem. This mechanism is highly specific and operates under mild conditions, preserving the sensitive beta-lactam ring from hydrolysis or epimerization. The ability to control this deprotection precisely ensures that the stereochemistry at the C-1 and C-6 positions remains intact, which is vital for the biological activity of the final antibiotic.
How to Synthesize Meropenem Efficiently
The execution of this synthesis requires careful attention to stoichiometry and reaction monitoring to maximize the benefits of the new route. The process begins with the condensation of the protected azetidinone acid and the thiol-containing pyrrolidine derivative, followed by activation with an oxalyl chloride derivative. The subsequent cyclization and deprotection steps must be managed to prevent the accumulation of impurities that could complicate crystallization. While the general pathway is robust, the specific choice of scavengers and solvents plays a pivotal role in determining the final crystal quality and yield. For a detailed breakdown of the operational parameters, please refer to the standardized synthesis guide below.
- Condense compound (IV) and compound (V) in organic solvent with a condensing agent to form the key intermediate (II).
- React intermediate (II) with compound (VI) under alkaline conditions to generate the activated azetidinone (III).
- Perform Wittig cyclization on compound (III) using organophosphorus reagents, followed by hydrolysis and palladium-catalyzed deprotection to yield Meropenem.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement directors and supply chain heads, the adoption of this patented synthesis method offers compelling strategic advantages that extend beyond mere technical novelty. The primary value proposition lies in the substantial reduction of raw material costs associated with catalyst usage. By eliminating the requirement for Rhodium, a precious metal subject to volatile market pricing and supply constraints, manufacturers can achieve significant cost stabilization. Furthermore, the replacement of high-pressure hydrogenation with ambient pressure chemical deprotection reduces the capital expenditure required for specialized safety equipment, thereby lowering the overall barrier to production and enhancing the economic viability of the project.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts like Rhodium directly impacts the bill of materials, leading to a more favorable cost structure for the final API. Additionally, the milder reaction conditions reduce energy consumption for heating and cooling, while the simplified workup procedures minimize solvent usage and waste disposal costs. These cumulative efficiencies allow for a more competitive pricing model without compromising on the quality or purity of the Meropenem produced, ensuring long-term margin protection for stakeholders.
- Enhanced Supply Chain Reliability: Relying on readily available organophosphorus reagents and common palladium catalysts mitigates the risk of supply disruptions often associated with specialty metals. The robustness of the allyl-protection chemistry ensures consistent batch-to-batch performance, reducing the likelihood of production delays caused by failed reactions or difficult purifications. This reliability is crucial for maintaining continuous supply to downstream formulation partners, especially in a market where demand for broad-spectrum antibiotics can fluctuate rapidly due to seasonal infection rates.
- Scalability and Environmental Compliance: The brevity of the synthetic route, combined with the avoidance of hazardous hydrogen gas, makes this process inherently safer and easier to scale from pilot plant to multi-ton commercial production. The reduced toxicity of reagents and the potential for solvent recycling align with increasingly stringent environmental regulations, minimizing the ecological footprint of the manufacturing process. This alignment with green chemistry principles not only future-proofs the production facility against regulatory changes but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific improvements and data points highlighted in the patent documentation, providing clarity on how this method compares to established industry standards. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value of this supply partnership.
Q: How does the allyl protection strategy improve yield compared to traditional nitrobenzyl methods?
A: The allyl group possesses significantly less steric hindrance compared to the bulky p-nitrobenzyl group. This structural advantage facilitates the critical Wittig ring-closure reaction, leading to improved conversion rates and higher overall yields without requiring harsh conditions.
Q: Does this synthesis method require expensive noble metal catalysts like Rhodium?
A: No. A major advantage of this patented process is the elimination of precious Rhodium catalysts traditionally used in the cyclization step. Instead, it utilizes more accessible organophosphorus reagents and Palladium catalysts for deprotection, substantially reducing raw material costs.
Q: Is hydrogen gas required for the final deprotection step?
A: Not necessarily. The process offers flexibility by allowing deprotection using chemical scavengers such as dimedone, tributyltin hydride, or N,N-dimethyl barbituric acid in the presence of a palladium catalyst. This avoids the safety hazards and infrastructure costs associated with high-pressure hydrogenation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meropenem Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the allyl-protection strategy are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Meropenem intermediate or API meets the highest international standards, providing our clients with the confidence needed to navigate complex regulatory filings.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative synthesis technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized Meropenem production capabilities can drive value and security for your organization.
