Advanced Manufacturing of High-Purity Dexmedetomidine Hydrochloride via Novel Friedel-Crafts Alkylation
Advanced Manufacturing of High-Purity Dexmedetomidine Hydrochloride via Novel Friedel-Crafts Alkylation
The pharmaceutical landscape for sedative agents continues to evolve, driven by the demand for safer, more efficient synthetic routes for critical active pharmaceutical ingredients (APIs). Patent CN112979552A, published in June 2021, introduces a transformative preparation method for high-purity Dexmedetomidine Hydrochloride, a potent alpha2-adrenoceptor agonist widely used in intensive care sedation. This intellectual property discloses a robust chemical strategy that bypasses the complexities of traditional multi-step syntheses, leveraging a direct Friedel-Crafts alkylation between 1-(1-chloroethyl)-2,3-dimethylbenzene and N-trimethylsilylimidazole. The structural integrity of the target molecule, characterized by its specific imidazole and dimethylphenyl moieties, is preserved through mild Lewis acid catalysis, setting a new benchmark for process chemistry in this therapeutic class.
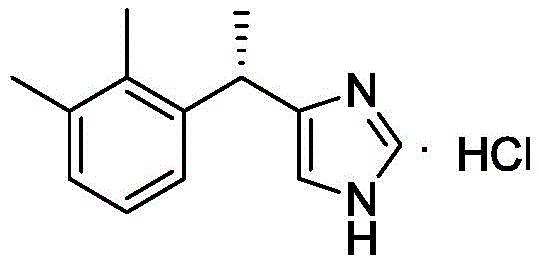
For R&D directors and process chemists, the significance of this patent lies in its ability to deliver a product with exceptional purity profiles. The disclosed method achieves both chemical and optical purity levels reaching 100 percent, a metric that is often elusive in chiral drug manufacturing. By integrating a specific recrystallization step for the medetomidine racemate prior to chiral resolution, the process effectively strips away regio-isomers and oligomeric byproducts that typically plague earlier synthetic attempts. This pre-purification strategy not only simplifies the downstream resolution but also ensures that the final API meets the stringent regulatory requirements for parenteral administration without the need for excessive refining cycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Dexmedetomidine Hydrochloride has been fraught with significant technical and economic hurdles. As illustrated in prior art such as US4910214, traditional routes often commence with N-trityl-4-acetylimidazole, necessitating a cumbersome sequence involving two distinct Grignard reactions, oxidation with manganese dioxide, elimination, and hydrogenation. These legacy processes are inherently problematic for modern manufacturing; the use of Grignard reagents introduces severe safety risks due to their pyrophoric nature, complicating scale-up operations in large reactors. Furthermore, the reliance on manganese dioxide for oxidation generates substantial quantities of solid waste, creating environmental disposal challenges and increasing the overall cost of goods sold (COGS).
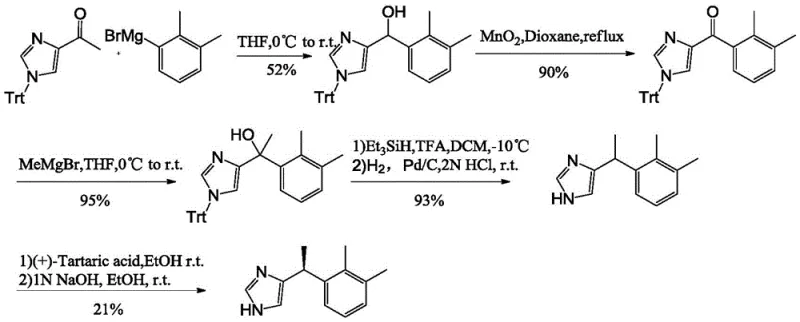
Alternative pathways, such as those described in EP1918282, attempt to mitigate some risks but introduce others, primarily centered around raw material costs and yield efficiency. These methods frequently utilize expensive starting materials like 4-iodo-1-trityl-1H-imidazole, which significantly inflates the material cost basis. Additionally, the hydrogenation reduction steps in these older protocols are prone to racemization, leading to a loss of optical purity that requires costly corrective measures. The cumulative effect of these inefficiencies—low yields, hazardous reagents, and expensive precursors—renders conventional methods suboptimal for a reliable agrochemical intermediate supplier or pharma partner seeking to optimize their supply chain for high-volume production.
The Novel Approach
In stark contrast to the convoluted legacy pathways, the novel approach detailed in CN112979552A streamlines the synthesis into a highly efficient, three-stage operation. The core innovation is the direct coupling of the aromatic and heterocyclic fragments via a Lewis acid-catalyzed Friedel-Crafts alkylation. This reaction utilizes 1-(1-chloroethyl)-2,3-dimethylbenzene and N-trimethylsilylimidazole, both of which are commercially available commodity chemicals, thereby drastically reducing raw material procurement costs. The reaction proceeds smoothly in aprotic organic solvents such as dichloromethane or chloroform, catalyzed by titanium tetrachloride or boron trifluoride, avoiding the extreme conditions and safety hazards associated with organometallic reagents.
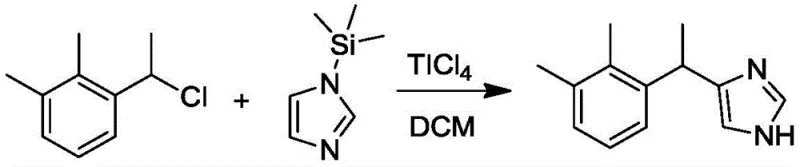
Furthermore, the novel approach incorporates a critical purification checkpoint that was absent in previous methodologies. Instead of carrying the crude reaction mixture directly into resolution, the process isolates the medetomidine racemate as a solid through a specialized recrystallization technique using solvents like methyl tert-butyl ether and petroleum ether. This step elevates the purity of the intermediate to over 99.5 percent, effectively removing trace impurities that could interfere with chiral separation. Consequently, the subsequent resolution with L-tartaric acid becomes highly selective and efficient, culminating in a final product with 100 percent optical purity and a total molar yield exceeding 20 percent, representing a substantial improvement in process mass intensity.
Mechanistic Insights into Lewis Acid-Catalyzed Friedel-Crafts Alkylation
The mechanistic elegance of this synthesis rests on the activation of the benzylic chloride by a strong Lewis acid, such as Titanium Tetrachloride (TiCl4). In this catalytic cycle, the Lewis acid coordinates with the chlorine atom of the 1-(1-chloroethyl)-2,3-dimethylbenzene, facilitating the heterolytic cleavage of the carbon-chlorine bond to generate a stabilized benzylic carbocation. This electrophilic species is then attacked by the nucleophilic nitrogen of the N-trimethylsilylimidazole. The trimethylsilyl (TMS) group serves a dual purpose: it enhances the nucleophilicity of the imidazole ring and acts as a leaving group that is easily removed during the aqueous workup, driving the equilibrium toward the formation of the desired C-N bond. This mechanism avoids the formation of unstable intermediates common in other routes, ensuring a cleaner reaction profile.
Impurity control is meticulously managed through the thermodynamic properties of the recrystallization step. The crude reaction mixture typically contains regio-isomers where the alkylation might occur at the alternative nitrogen of the imidazole ring, or oligomeric byproducts resulting from over-alkylation. By carefully selecting a solvent system with specific polarity characteristics, such as a mixture of toluene and petroleum ether, the process exploits the differential solubility of the target medetomidine versus these impurities. The target molecule crystallizes out of the solution with high fidelity, while the impurities remain in the mother liquor. This physical separation is far more robust than chromatographic methods for large-scale manufacturing, providing a scalable solution for producing high-purity pharmaceutical intermediates without the need for expensive silica gel columns.
How to Synthesize Dexmedetomidine Hydrochloride Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the reaction vessel under anhydrous conditions, followed by the sequential addition of the aprotic solvent and the silylated imidazole. The Lewis acid catalyst is introduced at controlled temperatures (0°C to 45°C) to manage the exotherm and prevent side reactions. Following the alkylation, the workup involves a careful quench with ice water and pH adjustment to isolate the free base. The critical recrystallization step must be performed with strict adherence to the solvent ratios and cooling profiles specified in the patent to ensure the removal of the 0.2% to 0.7% impurities typically found in crude batches. For the complete standardized operating procedure and safety guidelines, please refer to the detailed synthesis steps provided below.
- Perform Friedel-Crafts alkylation between 1-(1-chloroethyl)-2,3-dimethylbenzene and N-trimethylsilylimidazole using TiCl4 or BF3 in an aprotic solvent like DCM.
- Purify the resulting crude medetomidine oily substance via specific solvent recrystallization (e.g., MTBE/Petroleum Ether) to obtain high-purity racemate solid.
- Resolve the purified racemate using L-tartaric acid, followed by alkali dissociation and salt formation with hydrogen chloride to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical benefits. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive, specialty reagents like trityl-protected iodides with commodity chlorides and silanes, the method significantly reduces the direct material cost. This shift not only lowers the entry barrier for production but also mitigates the risk of supply disruptions, as the key starting materials are produced by multiple global vendors, ensuring a stable and continuous supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of hazardous Grignard reagents and heavy metal oxidants like manganese dioxide translates directly into lower operational expenditures. Without the need for specialized handling equipment for pyrophoric materials or extensive waste treatment facilities for heavy metal sludge, the overhead costs associated with safety compliance and environmental remediation are substantially decreased. Furthermore, the higher overall yield and the ability to recycle solvents like dichloromethane and toluene contribute to a more favorable cost structure, allowing for competitive pricing in the generic API market.
- Enhanced Supply Chain Reliability: The robustness of the Friedel-Crafts chemistry ensures consistent batch-to-batch quality, which is critical for maintaining regulatory filings and avoiding production delays. The process is less sensitive to minor fluctuations in reaction conditions compared to multi-step Grignard sequences, reducing the rate of batch failures. This reliability shortens the lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demand fluctuations and secure long-term contracts with major pharmaceutical companies.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The absence of stoichiometric heavy metal waste simplifies the effluent treatment process, aligning with increasingly stringent global environmental regulations. The process is inherently safer, reducing the risk of thermal runaways or fires associated with organometallic chemistry. These factors facilitate easier scale-up from pilot plant to commercial production scales (100 kgs to 100 MT), making it an ideal candidate for green chemistry initiatives and sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical advantages of this new methodology over established industry standards.
Q: How does the new Friedel-Crafts route improve upon traditional Grignard methods?
A: Traditional methods rely on hazardous Grignard reagents and expensive trityl-protected intermediates, generating significant solid waste (MnO2). The new route utilizes commodity chlorides and Lewis acids, eliminating heavy metal waste and reducing safety risks associated with pyrophoric reagents.
Q: What is the critical factor for achieving 100% optical purity in this process?
A: The key innovation is the intermediate recrystallization of the medetomidine racemate solid prior to chiral resolution. By removing regio-isomers and oligomeric impurities at the racemate stage (achieving >99.5% purity), the subsequent resolution with L-tartaric acid proceeds with maximum efficiency, yielding a final product with 100% chemical and optical purity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids difficult-to-control steps like low-temperature Grignard reactions and uses common industrial solvents (DCM, Toluene, Ethanol). The total molar yield exceeds 20%, and the starting materials are conventional industrial commodities, ensuring supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexmedetomidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the pathway described in CN112979552A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient route to life. We are committed to delivering Dexmedetomidine Hydrochloride with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the 100% chemical and optical purity benchmarks demanded by top-tier regulatory agencies.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can reduce your overall manufacturing expenses. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of this vital sedative agent.
