Revolutionizing 2-Chloronicotinic Acid Production: A Safe, Scalable, and Eco-Friendly Synthetic Route
Revolutionizing 2-Chloronicotinic Acid Production: A Safe, Scalable, and Eco-Friendly Synthetic Route
The global demand for high-value heterocyclic intermediates continues to surge, driven by the expanding pipelines of novel herbicides and pharmaceutical agents. At the forefront of this chemical evolution is 2-chloronicotinic acid, a critical building block for active ingredients such as Nicosulfuron, Niflumic Acid, and Nevirapine. However, traditional manufacturing methods have long been plagued by severe environmental liabilities and safety hazards. A groundbreaking technical disclosure, identified as Patent CN111393361A, introduces a paradigm-shifting synthesis method that replaces hazardous phosphorus oxychloride with solid phosgene (BTC). This innovation not only mitigates the risk of explosive exotherms but also drastically curtails the generation of phosphorus-laden wastewater, offering a sustainable pathway for the commercial scale-up of complex pharmaceutical intermediates. For industry leaders seeking a reliable agrochemical intermediate supplier, understanding this technological leap is essential for securing a resilient and compliant supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-chloronicotinic acid has relied heavily on the oxidation and chlorination of 3-cyanopyridine or nicotinic acid derivatives using phosphorus oxychloride (POCl3). While chemically effective, this legacy approach imposes unacceptable burdens on modern manufacturing facilities. The use of POCl3 necessitates strictly anhydrous conditions, requiring energy-intensive drying of all raw materials and solvents to prevent violent hydrolysis reactions. Furthermore, the stoichiometric consumption of POCl3 generates equimolar amounts of phosphoric acid and other phosphorus-containing byproducts, resulting in massive volumes of acidic wastewater that are notoriously difficult and expensive to treat. The inherent instability of the reaction mixture, combined with the corrosive nature of the reagents, creates a high-risk operational environment where temperature excursions can lead to flushing or even explosion, thereby threatening both personnel safety and production continuity.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN111393361A leverages the unique reactivity of solid phosgene (BTC) in conjunction with 3-cyanopyridine N-oxide to achieve chlorination under significantly milder conditions. This innovative route eliminates the stringent requirement for anhydrous solvents, as BTC exhibits a much lower sensitivity to moisture compared to POCl3, thereby simplifying raw material preparation and reducing energy consumption. The process integrates the chlorination and subsequent hydrolysis steps in a manner that allows for the direct recovery and reuse of the organic solvent, creating a closed-loop system that minimizes waste discharge. By shifting the chlorination mechanism away from phosphorus-based reagents, this approach fundamentally alters the impurity profile, avoiding the introduction of hard-to-remove phosphorus contaminants and facilitating a cleaner downstream purification process that aligns with rigorous green chemistry principles.
Mechanistic Insights into BTC-Mediated Chlorination and Hydrolysis
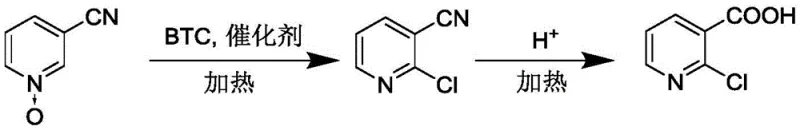
The core of this synthetic advancement lies in the activation of the pyridine N-oxide ring through a Vilsmeier-Haack-type mechanism facilitated by the BTC-DMF complex. Initially, the catalyst, typically a formamide derivative such as N,N-dimethylformamide (DMF), reacts with solid phosgene to generate a highly electrophilic chloroiminium species in situ. This activated complex selectively attacks the electron-rich positions on the 3-cyanopyridine N-oxide scaffold, specifically targeting the C2 position adjacent to the nitrogen atom. The presence of the N-oxide group serves as a powerful directing group, enhancing the nucleophilicity of the ring and ensuring high regioselectivity for the 2-chloro substitution. Following the chlorination event, the N-oxide functionality is reduced or eliminated during the workup, yielding 2-chloro-3-cyanopyridine as the key intermediate. This mechanistic pathway is distinct from radical chlorination methods, offering superior control over side reactions and ensuring that the cyano group remains intact for the subsequent transformation.
Following the chlorination stage, the process employs a clever tandem strategy where the aqueous workup serves a dual purpose. Upon the addition of water, any unreacted BTC is safely hydrolyzed, generating HCl which subsequently catalyzes the hydrolysis of the nitrile group to the carboxylic acid. Crucially, this hydrolysis step is performed in the presence of a decolorizing agent, typically activated carbon, at elevated temperatures ranging from 50°C to 110°C. This integration allows for the simultaneous removal of colored polymeric impurities and the conversion of the cyano moiety, effectively merging two unit operations into one. The resulting acidic aqueous solution is then subjected to hot filtration to remove the spent carbon, followed by cooling to induce crystallization of the final product. This streamlined sequence not only reduces the total processing time but also minimizes the number of isolation steps, thereby maximizing overall yield and operational efficiency.
How to Synthesize 2-Chloronicotinic Acid Efficiently
Implementing this robust synthetic route requires precise control over reaction parameters to ensure optimal yield and purity. The process begins with the suspension of 3-cyanopyridine N-oxide in a suitable organic solvent such as toluene, xylene, or ethyl acetate, followed by the sequential addition of solid phosgene and the formamide catalyst. The mixture is then heated to reflux, maintaining the temperature for a duration sufficient to drive the chlorination to completion, typically between 1 to 10 hours depending on the specific solvent system employed. After cooling, water is introduced to quench the reaction and extract the product into the aqueous phase, where the pH is naturally lowered by the generated HCl. For a comprehensive breakdown of the specific mass ratios, temperature profiles, and workup procedures validated by experimental data, please refer to the standardized protocol below.
- Charge 3-cyanopyridine N-oxide and an organic solvent (e.g., toluene or xylene) into a reactor, then add solid phosgene (BTC) and a catalytic amount of DMF derivative under stirring.
- Heat the mixture to reflux for chlorination (1-10 hours), cool to room temperature, and carefully add water to quench excess BTC and separate layers.
- Transfer the aqueous layer, add a decolorizing agent (activated carbon), heat to 50-110°C for simultaneous cyano hydrolysis and decolorization, then filter and crystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this BTC-based synthesis offers profound strategic benefits that extend far beyond simple chemical yield. The elimination of phosphorus oxychloride removes a major bottleneck related to hazardous material handling and waste disposal compliance. Traditional POCl3 routes often require specialized infrastructure for neutralizing and treating phosphorus-rich effluent, which represents a significant fixed cost overhead. By adopting this cleaner technology, manufacturers can bypass these expensive tertiary treatment requirements, leading to substantial cost savings in fine chemical intermediates manufacturing. Furthermore, the ability to recover and recycle the organic solvent directly from the reaction mixture reduces the consumption of fresh solvents, lowering both material costs and the logistical burden of solvent procurement and storage.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily derived from the simplification of the waste management workflow. Since the method avoids the generation of phosphorus-containing wastewater, facilities can operate with reduced environmental compliance expenditures and lower fees associated with hazardous waste disposal. Additionally, the mild reaction conditions reduce the energy load required for maintaining strict anhydrous environments, further driving down utility costs. The integration of the decolorization and hydrolysis steps also reduces labor hours and equipment occupancy time, enhancing the overall throughput of the production line without requiring additional capital investment in new reactors.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of solid phosgene, which is easier to transport and store safely compared to liquid phosphorus oxychloride. The reduced sensitivity to moisture means that raw material specifications can be relaxed, allowing for a broader base of qualified suppliers and reducing the risk of production stoppages due to off-spec reagents. Moreover, the high purity of the crude product (>99.0% in optimized examples) minimizes the need for extensive recrystallization or chromatographic purification, ensuring faster turnaround times from batch initiation to final packaging. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates in a just-in-time manufacturing environment.
- Scalability and Environmental Compliance: From a scalability perspective, the exothermic profile of the BTC-mediated reaction is far more manageable than that of POCl3, making the scale-up from pilot plant to commercial tonnage safer and more predictable. The process generates significantly less hazardous waste, aligning perfectly with increasingly stringent global environmental regulations such as REACH and local EPA standards. This forward-looking compliance ensures long-term operational viability and protects the brand reputation of downstream partners who prioritize sustainable sourcing. The robustness of the method allows for consistent quality across large batches, ensuring that commercial scale-up of complex agrochemical intermediates can be achieved without compromising on safety or environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what stakeholders can expect when adopting this technology. Understanding these nuances is vital for R&D teams evaluating process feasibility and for procurement officers negotiating supply contracts based on technical merit.
Q: Why is solid phosgene (BTC) preferred over phosphorus oxychloride for this synthesis?
A: Solid phosgene eliminates the generation of massive amounts of phosphorus-containing wastewater associated with POCl3, significantly reducing environmental compliance costs and explosion risks while allowing for milder reaction conditions that do not require strictly anhydrous solvents.
Q: What is the typical purity achievable with this novel synthetic route?
A: The patented process consistently yields 2-chloronicotinic acid with a content exceeding 99.0%, as demonstrated in multiple examples where HPLC detection confirmed high purity levels suitable for pharmaceutical and agrochemical applications.
Q: How does this method improve production efficiency compared to traditional routes?
A: By combining the hydrolysis and decolorization steps into a single operation within the aqueous phase and enabling solvent recovery from the organic layer for reuse, the process shortens the overall production cycle and minimizes waste generation at the source.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloronicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of next-generation synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of 2-chloronicotinic acid meets the exacting standards required for pharmaceutical and agrochemical applications. Our facility is equipped to handle the specific safety protocols associated with solid phosgene chemistry, guaranteeing a secure and compliant production environment for our global clientele.
We invite you to collaborate with us to leverage this superior synthetic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot runs and conduct detailed route feasibility assessments to demonstrate how this eco-friendly method can enhance your product portfolio while optimizing your operational expenditures.
