Advanced Aerobic Oxidation of 2-Chloronicotinic Acid: A Scalable Route for Global Pharma and Agrochemical Manufacturing
Advanced Aerobic Oxidation of 2-Chloronicotinic Acid: A Scalable Route for Global Pharma and Agrochemical Manufacturing
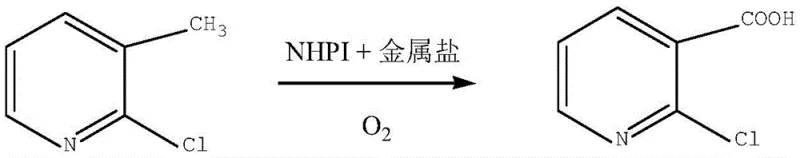
The chemical synthesis landscape for critical heterocyclic intermediates is undergoing a significant transformation driven by the dual imperatives of environmental sustainability and cost efficiency. A pivotal development in this domain is documented in patent CN111153853B, which discloses a highly efficient preparation method for 2-chloronicotinic acid, a cornerstone building block for numerous high-value pharmaceuticals and agrochemicals. This technology leverages a biomimetic catalytic system comprising N-hydroxyphthalimide (NHPI) and transition metal salts to facilitate the direct aerobic oxidation of 2-chloro-3-methylpyridine. Unlike traditional stoichiometric oxidation methods that generate substantial hazardous waste, this novel approach utilizes molecular oxygen as the terminal oxidant, aligning perfectly with modern green chemistry principles. For R&D directors and procurement strategists, this patent represents a viable pathway to secure a reliable agrochemical intermediate supplier status by mitigating supply chain risks associated with heavy metal waste disposal and volatile raw material costs. The process operates under relatively mild conditions, utilizing solvents such as acetonitrile or ethyl acetate, and demonstrates exceptional scalability potential for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-chloronicotinic acid has been plagued by significant technical and environmental bottlenecks that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. One prevalent legacy method involves the hydrolysis of 2-chloro-3-trichloromethyl pyridine using large quantities of strong inorganic acids; however, the precursor material is prohibitively expensive and difficult to source reliably on a global scale. Another common route entails the oxidation of nicotinic acid followed by chlorination using phosphorus oxychloride or phosphorus pentachloride. This multi-step sequence not only suffers from low overall yields but also generates copious amounts of acidic wastewater containing chlorine and phosphorus, imposing severe regulatory burdens and escalating three-waste treatment costs. Furthermore, alternative oxidation strategies employing potassium permanganate, while chemically straightforward, produce massive quantities of solid manganese dioxide sludge, creating a logistical nightmare for waste management and significantly inflating the cost of goods sold. Even greener alternatives like ozone oxidation face barriers due to the high capital expenditure required for specialized ozone generation equipment, rendering them less attractive for standard batch processing facilities.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the technology outlined in CN111153853B introduces a streamlined, one-step oxidative transformation that fundamentally alters the economic equation for producing this vital intermediate. By employing 2-chloro-3-methylpyridine as a readily available starting material and utilizing a catalytic amount of NHPI combined with cobalt or manganese salts, the process achieves direct conversion to the carboxylic acid using pressurized oxygen. This methodology eliminates the need for stoichiometric oxidants like permanganate or hazardous chlorinating agents, thereby drastically simplifying the downstream purification workflow. The reaction proceeds with high atom economy, where oxygen serves as the clean oxidant and water is the primary byproduct, effectively decoupling production volume from hazardous waste generation. Moreover, the flexibility in solvent selection, particularly the preference for acetonitrile which offers superior mass transfer properties, ensures consistent reaction kinetics and facilitates easy solvent recovery through distillation. This robust protocol delivers high-purity 2-chloronicotinic acid with minimal impurity profiles, specifically avoiding the troublesome 6-chloro isomer often found in electrophilic substitution routes.
Mechanistic Insights into NHPI-Catalyzed Aerobic Oxidation
The efficacy of this synthetic route relies on a sophisticated radical chain mechanism mediated by the synergistic interaction between the organic initiator NHPI and the transition metal catalyst. Under thermal conditions, the N-hydroxyphthalimide undergoes hydrogen abstraction, typically facilitated by the metal species, to generate the phthalimide-N-oxyl (PINO) radical. This stable nitroxyl radical is the active species responsible for abstracting a hydrogen atom from the benzylic methyl group of the 2-chloro-3-methylpyridine substrate, generating a carbon-centered radical. This carbon radical subsequently reacts with molecular oxygen to form a peroxy radical, which propagates the chain reaction by abstracting hydrogen from another molecule of NHPI or the substrate, eventually leading to the formation of the aldehyde and finally the carboxylic acid functionality. The metal salt, whether cobalt acetate, cobalt acetylacetonate, or their manganese counterparts, plays a crucial role in decomposing intermediate hydroperoxides and regenerating the active PINO radical, thus maintaining the catalytic cycle without being consumed. This mechanistic pathway ensures high selectivity for the methyl group oxidation while leaving the sensitive chloro-substituent and the pyridine ring intact, a feat that is often challenging with harsher chemical oxidants.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic reaction pathways. Traditional chlorination methods often suffer from poor regioselectivity, leading to the formation of 6-chloronicotinic acid as a persistent structural impurity that is difficult to separate due to similar physical properties. However, the radical oxidation described here targets the specific C-H bond of the methyl group, inherently minimizing the risk of ring substitution or halogen scrambling. The use of acetonitrile as a solvent further enhances this selectivity by providing a polar environment that stabilizes the transition states involved in the radical propagation steps without participating in side reactions. Additionally, the mild reaction temperatures, typically optimized between 50°C and 120°C, prevent thermal degradation of the product or the formation of polymeric tars that can complicate filtration. The result is a crude product profile that is exceptionally clean, often requiring only a simple acid-base workup to achieve purity levels exceeding 98.5%, thereby reducing the burden on analytical quality control laboratories and accelerating batch release times.
How to Synthesize 2-Chloronicotinic Acid Efficiently
Implementing this advanced oxidation protocol requires precise control over reaction parameters to maximize yield and ensure operational safety, particularly when handling pressurized oxygen systems. The process begins with the careful charging of the autoclave with the substrate, solvent, and catalytic components, followed by a controlled pressurization and heating phase to initiate the radical chain. Detailed standard operating procedures regarding the specific molar ratios of NHPI to substrate and the optimal pressure ramping rates are critical for reproducibility on a multi-ton scale. For a comprehensive breakdown of the exact experimental conditions, reagent grades, and workup parameters validated in the patent examples, please refer to the standardized synthesis guide below.
- Charge an autoclave with 2-chloro-3-methylpyridine, acetonitrile solvent, N-hydroxyphthalimide (NHPI), and a cobalt or manganese salt catalyst.
- Pressurize the reactor with oxygen to 0.2-2 MPa and heat the mixture to 50-120°C while stirring for 3-24 hours to complete the oxidation.
- Cool the reaction, filter the solid, dissolve in alkali, acidify with hydrochloric acid to pH 1-2, and dry to obtain the final white crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this NHPI-catalyzed aerobic oxidation route translates into tangible strategic advantages that extend far beyond simple yield metrics. The primary value driver is the substantial reduction in raw material complexity and waste disposal liabilities. By replacing expensive and hazardous stoichiometric oxidants like potassium permanganate or phosphorus halides with cheap molecular oxygen, the variable cost of production is significantly lowered. Furthermore, the elimination of heavy metal sludge (manganese dioxide) and acidic phosphorus wastewater removes a major bottleneck in environmental compliance, allowing manufacturing sites to operate with greater flexibility and reduced regulatory risk. This streamlined process also enhances supply chain resilience by relying on commodity chemicals (2-chloro-3-methylpyridine, acetonitrile, oxygen) that are widely available globally, reducing the risk of supply disruptions associated with specialty reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental shift from stoichiometric reagents to catalytic turnover. In traditional methods, the cost of the oxidant often equals or exceeds the cost of the substrate, and the subsequent waste treatment adds a hidden tax to every kilogram produced. By utilizing oxygen from the air and a reusable metal catalyst, the direct material costs are drastically minimized. Additionally, the ability to recover and recycle the acetonitrile solvent through distillation further compounds these savings, creating a closed-loop system that maximizes resource efficiency. The high selectivity of the reaction also means less product is lost to side reactions or difficult purification steps, effectively increasing the throughput of existing manufacturing assets without the need for capital expansion.
- Enhanced Supply Chain Reliability: Securing a consistent supply of critical intermediates like 2-chloronicotinic acid is paramount for downstream manufacturers of herbicides and antiviral drugs. This synthetic route mitigates supply risk by decoupling production from the availability of niche precursors like 2-chloro-3-trichloromethyl pyridine. The starting material, 2-chloro-3-methylpyridine, is a mature commodity chemical with a robust global supply base. Furthermore, the simplicity of the reaction setup—requiring standard pressure reactors rather than specialized ozone generators or corrosion-resistant glass-lined vessels for strong acids—means that more contract manufacturing organizations (CMOs) are capable of executing this chemistry. This broadens the potential supplier base and reduces the likelihood of single-source bottlenecks affecting the broader supply chain.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions tighten globally, the environmental profile of a chemical process becomes a key determinant of its long-term viability. This aerobic oxidation method generates water as the primary byproduct, virtually eliminating the liquid effluent load associated with acidic hydrolysis or chlorination routes. The absence of solid manganese waste simplifies the solid waste stream, reducing the need for hazardous waste landfills. From a scalability standpoint, the exothermic nature of oxidation reactions is well-managed in this protocol through controlled oxygen feeding and temperature regulation, making it safe to scale from pilot plants to hundred-ton commercial reactors. This alignment with green chemistry principles future-proofs the supply chain against evolving environmental regulations and carbon taxation schemes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHPI-catalyzed oxidation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational feasibility and quality advantages of this method for potential partners and licensees. Understanding these nuances is essential for evaluating the technology transfer potential and integration into existing manufacturing portfolios.
Q: Why is the NHPI/Metal Salt system superior to traditional KMnO4 oxidation for 2-chloronicotinic acid?
A: Traditional potassium permanganate oxidation generates massive amounts of solid manganese dioxide waste, creating significant disposal costs and environmental burdens. The NHPI/metal salt catalytic system uses molecular oxygen as the terminal oxidant, producing water as the only byproduct, which drastically simplifies waste treatment and improves the overall green chemistry profile of the manufacturing process.
Q: What is the expected purity and yield profile for this aerobic oxidation method?
A: According to patent data, this method consistently achieves product purity exceeding 98.5% with yields ranging from 75% to over 94% depending on optimization. This high selectivity minimizes the formation of difficult-to-remove isomers like 6-chloronicotinic acid, which is a common impurity in chlorination-based routes.
Q: Can the solvent system be recovered to reduce operational costs?
A: Yes, the preferred solvent, acetonitrile, can be efficiently recovered from the reaction mixture through simple distillation after the product precipitation. This recyclability significantly lowers the variable cost per kilogram compared to processes using stoichiometric acids or non-recoverable solvent systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloronicotinic Acid Supplier
The technological breakthroughs detailed in patent CN111153853B underscore the immense potential for optimizing the production of key heterocyclic intermediates through green catalytic oxidation. At NINGBO INNO PHARMCHEM, we recognize that translating such laboratory innovations into commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this NHPI-catalyzed route are fully realized in large-scale batches. Our state-of-the-art facilities are equipped to handle pressurized oxidation reactions safely, and our stringent purity specifications guarantee that every batch of 2-chloronicotinic acid meets the exacting standards required for pharmaceutical and agrochemical applications. With our rigorous QC labs and commitment to process safety, we provide a secure foundation for your supply chain.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this advanced synthetic route for their specific project needs. Whether you require custom synthesis of novel derivatives or bulk supply of the parent acid, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only cost-effective and reliable but also aligned with the highest standards of environmental stewardship and scientific innovation.
