Advanced Manufacturing of 1-Aryltriazolinones for High-Efficiency Herbicide Production
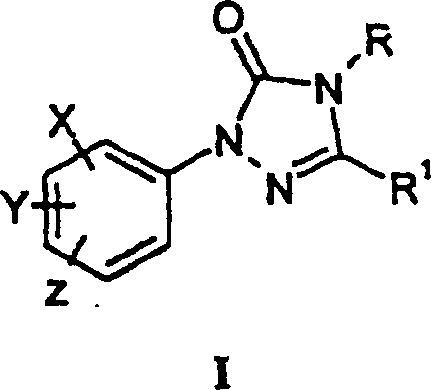
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical heterocyclic intermediates. Patent CN100349879C introduces a groundbreaking methodology for the preparation of 1-aryltriazolinones, specifically compounds of Formula I, which serve as pivotal building blocks in the manufacture of commercially significant agrochemicals. This technology addresses long-standing challenges in process chemistry by offering streamlined routes that bypass the inefficiencies of traditional multi-step syntheses. By leveraging either direct carbonylation of amidrazones or condensation of hydrazone derivatives, this process achieves remarkably high yields and purity profiles essential for downstream pharmaceutical and agricultural applications. For R&D directors and procurement specialists, understanding the mechanistic nuances of this patent is crucial for optimizing supply chains and reducing the total cost of ownership for herbicide active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-aryltriazolinones has been plagued by inefficient reaction pathways that necessitate the formation of a saturated 1-aryltriazolidinone ring as a discrete intermediate. This conventional requirement imposes a significant burden on manufacturing processes, as it inherently adds an extra chemical transformation step that must be isolated, purified, and subsequently converted into the desired unsaturated triazolinone product. Such multi-step sequences not only increase the consumption of raw materials and solvents but also amplify the potential for yield loss at each stage of the operation. Furthermore, the conversion of the triazolidinone to the triazolinone often generates substantial quantities of difficult-to-remove by-products, complicating the purification process and potentially compromising the final purity specifications required for high-value agrochemical formulations. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for suppliers aiming to meet the rigorous demands of the global herbicide market.
The Novel Approach
In stark contrast to legacy methodologies, the process disclosed in patent CN100349879C offers a direct and elegant solution by enabling the one-step formation of the 1-aryltriazolinone ring system from readily accessible precursors. This novel approach utilizes either the carbonylation of amidrazones (Formula A where W is -NHR) or the condensation of hydrazone derivatives (Formula A where W is a leaving group) with specific ring-forming agents. By eliminating the need for the intermediate triazolidinone species, the process drastically simplifies the synthetic workflow, reducing the number of unit operations and minimizing waste generation. The ability to achieve high conversion rates and exceptional purity in a single reactor setup represents a paradigm shift in process economics, allowing manufacturers to scale production with greater confidence and efficiency. This technological leap not only enhances the viability of existing herbicide portfolios but also opens new avenues for the cost-effective development of next-generation crop protection agents.
Mechanistic Insights into Carbonylation and Cyclization Chemistry
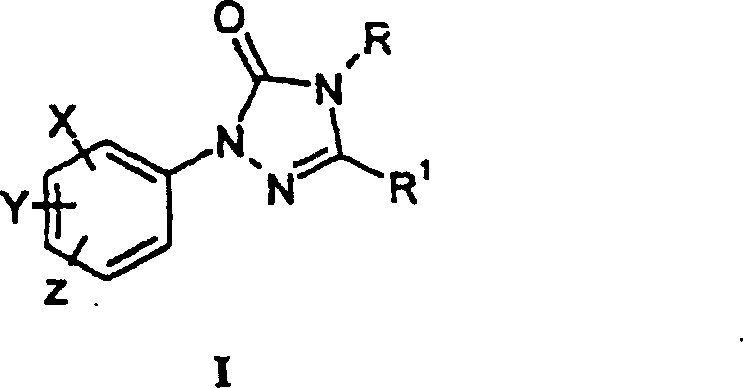
The core of this innovative synthesis lies in the precise manipulation of nitrogen-containing heterocycles through two distinct yet complementary mechanistic pathways. In the carbonylation route, the amidrazone intermediate undergoes a nucleophilic attack on a carbonylating agent, such as diphenyl carbonate or diethyl carbonate, facilitated by the presence of a catalytic amount of a base like DMAP or an acid like boron trifluoride. This reaction proceeds through a tetrahedral intermediate that collapses to expel the leaving group (e.g., phenol), resulting in the closure of the five-membered triazolinone ring with the concomitant formation of the carbonyl functionality at the 5-position. The choice of solvent plays a critical role in this mechanism; polar aprotic solvents like DMAC or non-polar solvents like toluene are selected to optimize the solubility of the reactants while minimizing solvolysis side reactions, ensuring that the reaction kinetics favor the desired cyclization over competing degradation pathways.
Alternatively, the condensation pathway relies on the reactivity of hydrazone derivatives, particularly those bearing a good leaving group such as chlorine at the imine carbon. When treated with a ring-forming agent like potassium cyanate or sodium cyanate in the presence of a catalyst such as potassium fluoride, the hydrazone nitrogen attacks the electrophilic carbon of the cyanate. This triggers an intramolecular cyclization that constructs the triazolinone core with high regioselectivity. The inclusion of catalytic amounts of water in glyme solvents has been observed to further accelerate this reaction, likely by enhancing the dissolution of the inorganic cyclizing agent and facilitating ion pair separation. This mechanistic flexibility allows process chemists to tailor the reaction conditions to the specific electronic and steric properties of the substituents on the aryl ring, ensuring robust performance across a wide range of substrate variations.
How to Synthesize 1-Aryltriazolinones Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal management to maximize the benefits of the novel pathways. The process begins with the preparation of the key amidrazone or hydrazone intermediates from substituted phenylhydrazines, followed by the critical ring-closing step which defines the efficiency of the overall sequence. Operators must select the appropriate solvent system—whether it be toluene for carbonylation or DMAC for condensation—to ensure optimal reaction homogeneity and heat transfer. While the general principles are outlined here, the specific parameters for temperature ramps, addition rates, and workup procedures are critical for reproducibility. For a comprehensive understanding of the standardized operating procedures and safety protocols required for commercial implementation, please refer to the detailed synthesis guide below.
- Preparation of Amidrazone or Hydrazone Derivatives: Synthesize the key intermediate (Formula A) by reacting substituted phenylhydrazines with ethyl acetimidate or acetic anhydride followed by chlorination.
- Ring Closure via Carbonylation: React the amidrazone intermediate with a carbonylating agent like diphenyl carbonate in toluene with a DMAP catalyst to form the triazolinone ring.
- Alternative Ring Closure via Condensation: Condense the hydrazone derivative with a ring-forming agent such as potassium cyanate in DMAC or glyme solvents, utilizingKF catalysis for cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented synthesis route offers compelling advantages that extend far beyond simple yield improvements. By fundamentally restructuring the synthetic pathway to eliminate redundant steps, manufacturers can achieve a significant reduction in the overall consumption of raw materials, solvents, and energy resources. This streamlining of the process directly translates to a lower cost of goods sold (COGS), providing a competitive edge in price-sensitive markets such as generic herbicide production. Furthermore, the robustness of the reaction conditions, which tolerate a broad range of temperatures and solvent qualities, reduces the risk of batch failures and ensures a more predictable production schedule, thereby enhancing supply chain reliability for downstream customers.
- Cost Reduction in Manufacturing: The elimination of the intermediate triazolidinone formation step removes an entire unit operation from the manufacturing train, including the associated costs of isolation, drying, and re-dissolution. This reduction in processing time and equipment usage leads to substantial operational savings without compromising product quality. Additionally, the use of inexpensive and widely available reagents such as potassium cyanate and diphenyl carbonate, coupled with recoverable solvents like toluene, further drives down the variable costs associated with large-scale production, making the process economically viable for high-volume commodity chemical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials mitigates the risks associated with sourcing specialized or unstable intermediates that often plague complex organic syntheses. The process's tolerance for varied reaction conditions means that production can be maintained even if minor fluctuations in utility supplies or raw material grades occur, ensuring continuous availability of the critical 1-aryltriazolinone intermediate. This resilience is vital for maintaining uninterrupted supply lines to formulation plants, preventing costly downtime and ensuring that end-users receive their agrochemical products on time.
- Scalability and Environmental Compliance: The simplified workflow inherently generates less waste, as fewer purification steps result in reduced solvent discharge and lower volumes of solid by-products. The ability to run reactions in standard industrial solvents like toluene and DMAC facilitates easier solvent recovery and recycling, aligning with modern green chemistry principles and regulatory requirements. This environmental efficiency not only lowers waste disposal costs but also simplifies the permitting process for facility expansions, allowing for seamless scale-up from pilot plant quantities to multi-ton commercial production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing frameworks.
Q: What are the primary advantages of the novel 1-aryltriazolinone synthesis process over conventional methods?
A: The novel process described in patent CN100349879C eliminates the need for forming a 1-aryltriazolidinone ring as a separate intermediate step, which is required in conventional methods. This direct formation of the triazolinone ring significantly reduces the number of unit operations, minimizes by-product formation, and results in surprisingly high yields and purity levels suitable for commercial herbicide production.
Q: Which solvents and catalysts are preferred for the carbonylation route of amidrazones?
A: For the carbonylation of amidrazones, preferred organic solvents include toluene, DMF, DMAC, and glymes, with toluene being particularly preferred for its balance of cost and solubility. The reaction is effectively accelerated by acid or base catalysts, with 4-dimethylaminopyridine (DMAP), boron trifluoride, and scandium triflate identified as particularly effective catalysts for driving the reaction to completion under mild thermal conditions.
Q: How does this process ensure supply chain reliability for agrochemical manufacturers?
A: The process utilizes readily available and cost-effective reagents such as diphenyl carbonate, potassium cyanate, and common industrial solvents like toluene and DMAC. By avoiding exotic or highly unstable intermediates and operating under robust conditions that tolerate a range of temperatures and concentrations, the method ensures consistent batch-to-batch quality and reduces the risk of supply disruptions associated with complex multi-step syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aryltriazolinones Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final agrochemical products. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN100349879C are fully realized in practice. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-aryltriazolinones meets the exacting standards required for herbicide registration and performance. Our commitment to technical excellence ensures that you receive a product that is not only cost-effective but also consistently reliable for your formulation needs.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your supply chain efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the agrochemical sector.
